- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Differentiation of Human Embryonic Stem Cells into Cone Photoreceptors
Published: Vol 6, Iss 14, Jul 20, 2016 DOI: 10.21769/BioProtoc.1870 Views: 11147
Reviewed by: Agnieszka PastulaNicoletta CordaniAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
In vitro Time-lapse Imaging of Primary Cilium in Migrating Neuroblasts
Masato Sawada [...] Kazunobu Sawamoto
Nov 20, 2020 4902 Views
Generation of Mouse Primitive Endoderm Stem Cells
Yasuhide Ohinata [...] Haruhiko Koseki
Nov 20, 2023 2471 Views
An Efficient Method for Immortalizing Mouse Embryonic Fibroblasts by CRISPR-mediated Deletion of the Tp53 Gene
Srisathya Srinivasan and Hsin-Yi Henry Ho
Jan 20, 2025 2800 Views
Abstract
Photoreceptors are specialized retinal neurons able to respond to light in order to generate visual information. Among photoreceptors, cones are involved in colors discrimination and high-resolution central vision and are selectively depleted in macular degenerations and cone dystrophies. A possible therapeutic solution for these disorders is to replace degenerating cells with functional cones. Here, we describe a simple protocol for the rapid production of large amount of cone photoreceptors from human pluripotent stem cells. The differentiation protocol is based on the “default pathway” of neural induction using the BMP, TGFβ and WNT antagonist COCO.
Keywords: Cone photoreceptorMaterials and Reagents
- Tissue culture 6-well plates (Thermo Fischer Scientific, catalog number: 130184 )
- Pipets (5 ml, 10 ml)
- 50 ml conical tubes (Thermo Fischer Scientific, catalog number: 339653CS )
- Micro cover glass (VWR International, catalog number: 89015-724 )
- Cell scraper (Thermo Fischer Scientific, catalog number: 179693 )
- 0.22 μm sterilize filter Filtropur (Sarstedt, catalog number: 83.1826.001 )
- Human embryonic stem (hES) cell line HUES9 (Harvard stem cell institute) or H9 cells (WiCell, catalog number: WA09 )
- hES cell media (Peprotech, catalog number: BM-HESC-500 )
- N2 supplement (Gibco, catalog number: 17502-048 )
- B27 supplement (Gibco, catalog number: 17504-044 )
- DMEM/F12 (Gibco, catalog number: 11320-033 )
- NEAA (Gibco, catalog number: 11140-050 )
- Human FGF-basic (Peprotech, catalog number: 100-18B-50UG )
- Heparin (Sigma-Aldrich, catalog number: H3149-10KU )
- Human IGF-I (Peprotech, catalog number: 100-11 )
- Human recombinant COCO (R&D Systems, catalog number: 3047-CC )
- Accutase solution (100 ml) (Sigma-Aldrich, catalog number: A6964 )
- Rock inhibitor Y-27632 (hydrochloride) (Cayman chemical, catalog number: 10005583 )
- Reduced growth factor matrigel (Corning, catalog number: 354230 )
- hES cell qualified matrigel (Corning, catalog number: 354277 )
- Trizol reagent (Thermo Fisher Scientific, AmbionTM, catalog number: 15596026 )
- Protease inhibitor cocktail (Sigma-Aldrich, catalog number: 04693116001 )
- Na2HPO4·7H2O (FisherBiotech, catalog number: BP332-500 )
- NaCl (Amresco, catalog number: 0241 )
- KCl (EMD Millipore, catalog number: PX1405-1 )
- KH2PO4 (FisherBiotech, catalog number: BP362-1 )
- Crystalline PFA (Sigma-Aldrich, catalog number: P6148 )
- 1x PBS
- Antibodies against CRX, S-opsin, ABCA4 and cone arrestin
- CI media (see Recipes)
- 10x phosphate-buffer saline (PBS) (see Recipes)
- 4% paraformaldehyde (PFA) (see Recipes)
- hES-qualified matrigel plate coating (see Recipes)
Equipment
- 37 °C, 5% CO2 water jacketed incubator (Thermo Fisher Scientific, catalog number: 3110 )
- P1000, P200 pipets
- Microscope (Leica, model: DM IL )
- Refrigerated Tabletop Centrifuge for 50 ml conical tubes (Thermo Fisher Scientific, Centra GP8R, 031220F )
Procedure
- Preparation and plating of human Embryonic Stem (hES) cells-Time: day -6 to day 0
- Culture hES cell line HUES9 on matrigel-coated plates (see hES qualified Matrigel in Recipes) until they form large colonies (~1 x 106 cells) using hES cell media (see Materials and Reagents) (Figure 1A).
- Dilute reduced growth factor matrigel in ice cold DMEM/F12 using pre-chilled 5 ml pipets and 200 μl pipet tips: mix 100 μl of matrigel on ice and 6 ml of ice cold DMEM/F12.
- Add 1 ml of freshly prepared matrigel per well of a 6-well plate.
Note: Place a sterile coverslip before adding the matrigel for immunofluorescence analysis. - Evenly distribute the matrigel and incubate for 30 min at 37 °C into a water jacketed incubator.
- During matrigel incubation, discard hES cell media and wash with 1 ml of 1x PBS pre-warmed at 37 °C.
Note: This protocol is adapted to the use of 3 well of a 6-well plate with large hES cell colonies as starting material. - Discard PBS and add 1 ml of Accutase pre-warmed at room temperature.
- Incubate the hES cell plate until the cells start to detach (round shaped cells) at 37 °C (1-2 min).
- Stop the reaction with 1 ml of hES cell media.
- Collect the cells into a 50 ml conical tube with a P1000 pipet tip by pipetting up and down.
- Add 6 ml of warmed hES cell media.
- Add ROCK inhibitor Y27632 to reach the final concentration of 10 μM.
- Centrifuge at 1,300 rpm (400 x g) for 5 min at room temperature.
- Discard the supernatant and resuspend the cells into 12 ml of warm hES cell media.
- Add ROCK inhibitor Y27632 to reach the final concentration of 10 μM.
- Take the matrigel-coated plate from the incubator and discard the DMEM/F12 remaining on each well.
- Distribute 2 ml of hES cells suspension in each well of the matrigel-coated plate.
- Swirl the plate and incubate into an incubator at 37 °C.
- Change culture media every day until the cells reach full confluency (100%).
- Culture hES cell line HUES9 on matrigel-coated plates (see hES qualified Matrigel in Recipes) until they form large colonies (~1 x 106 cells) using hES cell media (see Materials and Reagents) (Figure 1A).
- Differentiation-Time: day 0 to day 21
- Prepare and sterile filter CI media needed for two weeks.
- Upon full confluency, remove hES cell media from the plate and add 2 ml of freshly prepared CI media (Figure 1B).
- Change media every day by slowly dispensing warmed CI media on the side of the well for at least 21 days (Figure 1C).
- Prepare and sterile filter CI media needed for two weeks.
- Cell collection and analysis
- For gene expression analysis
- Discard the media, wash with 1 ml of 1x PBS and add 1 ml of Trizol reagent.
- Pipet up and down with a P1000 pipet tip and collect all the cells.
- Store at -80 °C or proceed to RNA extraction.
- Differentiation efficiency was confirmed by real-time PCR using primers against SIX6, CRX, S-opsin (OPN1SW), cone arrestin [also known as arrestin 3 (ARR3)], cone transducing (GNAT2), phosphodiesterase 6H (PDE6H) and phosphodiesterase 6C (PDE6C). Primers sequences are: SIX6 (F; CCTGCAGGATCCATACCCTA and R; ACCTGCTGCTGGAGTCTGTT), CRX (F; CCTTCTGACAGCTCGGTGTT and R; CCACTTTCTGAAGCCTGGAG), S-opsin (F; TGTGCCTCTCTCCCTCATCT and R; GGCACGTAGCAGACACAGAA), Cone Arrestin (F; CCCAGAGCTTTGCAGTAACC and R; CACAGGACACCATCAGGTTG), PdE6h (F; TACTCTGCCTGCTCCAGCTT and R; GAGAGTGGCAGAACCTCTGG), PdE6c (F; TTGGGAACAAGGAGATCTGG and R; GGCTCCTCCTTCTTGCTTTT) (Zhou et al., 2015).
- For protein analysis
Detach the cells in the culture media using a cell scraper. Centrifuge at 1,300 rpm for 5 min and discard the supernatant. Resuspend the pellet in 200 μl of protease inhibitor and store at -20 °C or proceed to protein extraction. Protein expression can be analyzed by western blotting using antibodies against CRX, S-opsin, cone arrestin and cone transducing (Zhou et al., 2015). - For immunofluorescence analysis
Discard the media, wash with 1 ml of 1x PBS and add 1 ml of fresh PFA at 4%. Incubate 10 min at room temperature. Discard PFA and proceed to immunofluorescence using antibodies against CRX, S-opsin, ABCA4 and cone arrestin. Coverslips can be mounted on slides after secondary antibody incubation (Figure 1D) (Zhou et al., 2015).
Representative data
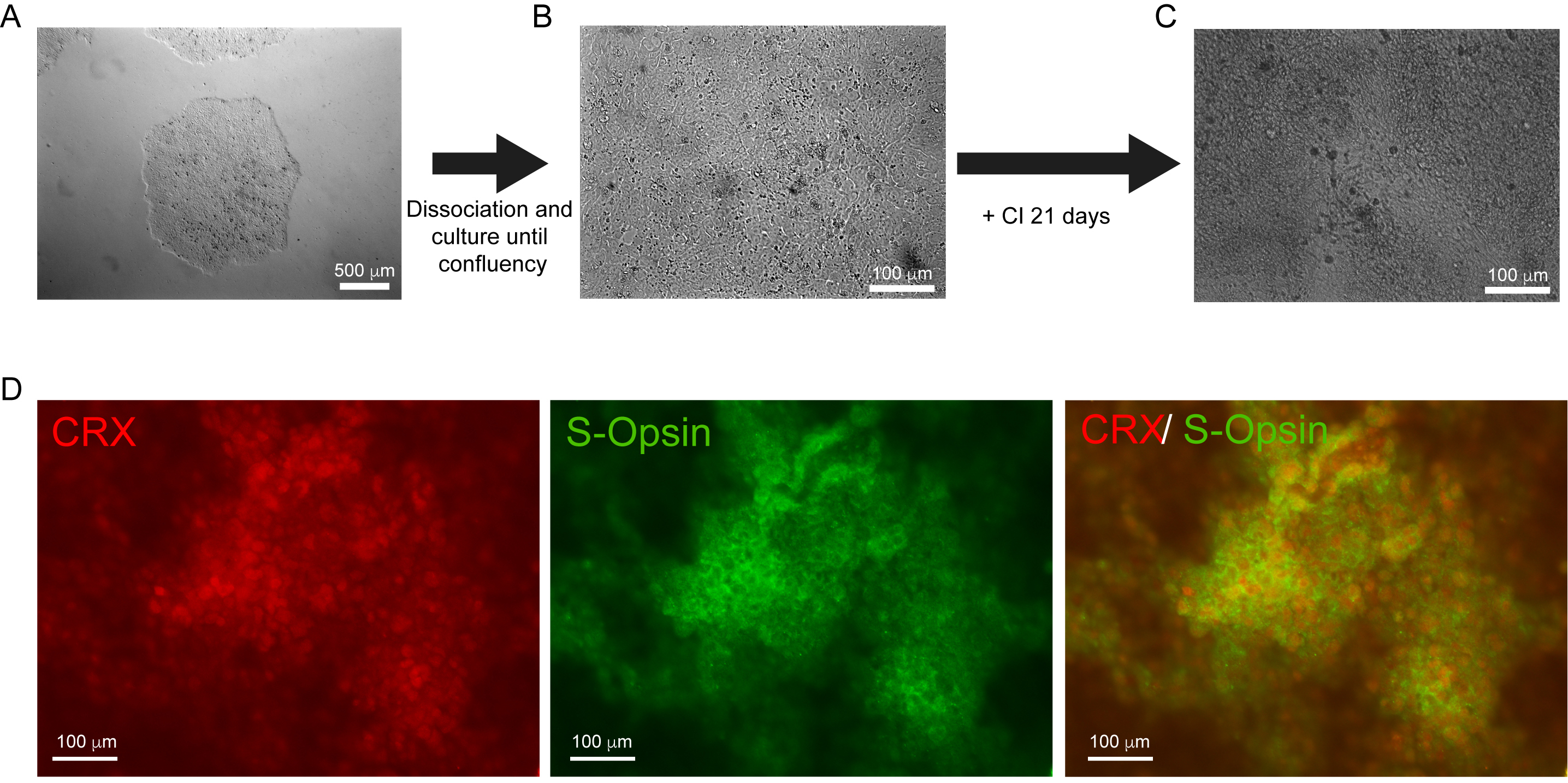
Figure 1. Representative images of hES cells at different time points of the protocol. A. Large hES cell colony ready for passaging. Scale bar: 500 μm B. Confluent layer of hES cells ready to start the differentiation (100% confluency). Scale bar: 100 μm C. Cells obtained after 21 days of differentiation. Scale bar: 100 μm D. Differentiated cells after 21 days of differentiation labelled with the photoreceptor markers CRX and S-Opsin. Scale bar: 100 μm.
Notes
The quality of the starting hES cells is essential to obtain a good efficiency of differentiation. Therefore, it is recommended to assess pluripotency of the cells by detecting hES cell markers such as Tra1-60, Oct3/4, SSEA4 and Nanog. This protocol has been tested using either HUES9 cell line or H9 cells and differentiation efficiency was confirmed by the detection of CRX, S-Opsin, Recoverin and ABCA4.
The use of Accutase is essential to obtain a good cell viability after re-plating at the opposite of Trypsin.
Recipes
- 10x Phosphate-Buffer Saline (PBS)
Mix 25.6 g Na2HPO4·7H2O, 80 g NaCl, 2 g KCl, 2 g KH2PO4
Bring to 1 liter with H2O.
Autoclave for 40 min at 121 °C. - CI media
Add all of these components:
48 ml DMEM/F12
0.5 ml N2 supplement (100x)
1 ml B27 supplement (100x)
0.5 ml NEAA (100x)
12.5 μl human FGF-basic (10 ng/μl)
5 μl Heparin (10 μg/μl)
5 μl human IGF-I (100 ng/μl)
7.5 μl human recombinant COCO (200 μg/ml)
Filter sterilize (0.22 μm) and store at 4 °C.
Use within two weeks. - 4% PFA
Weigh 16 g of crystalline PFA in a beaker and add 400 ml of 1x PBS. Cover and heat the solution until the powder is fully dissolved (50 °C). Aliquot and store at -20 °C. - hES-qualified matrigel plate coating
Dilute hES-qualified matrigel in ice cold DMEM/F12 using pre-chilled 10 ml pipets and P1000 pipet tips: mix a volume of matrigel on ice corresponding to the dilution factor (see certificate) and 25 ml of ice cold DMEM/F12. Dispend 1 ml of diluted matrigel per well of a 6-well plate and incubate for 30 min at 37 °C into a water jacketed incubator. Discard the excess of DMEM/F12 before use.
Acknowledgments
We are grateful to D. Melton and Harvard Stem Cell Institute for the HUES9 cell line. This work was supported by grants from the Foundation Fighting Blindness Canada, Turmel Family Foundation for Macular Degeneration Research, Canadian Stem Cell Network, C. Durand Foundation, the GO Foundation, and Natural Science and Engineering Research Council of Canada (Grant #250970-2012). A.F. is supported by fellowships from the Réseau Vision du Québec and from University of Montreal Molecular Biology Program. G.B is supported by the Fonds de Recherche en Santé du Québec.
References
- Zhou, S., Flamier, A., Abdouh, M., Tetreault, N., Barabino, A., Wadhwa, S. and Bernier, G. (2015). Differentiation of human embryonic stem cells into cone photoreceptors through simultaneous inhibition of BMP, TGFbeta and Wnt signaling. Development 142(19): 3294-3306.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Flamier, A., Barabino, A. and Bernier, G. (2016). Differentiation of Human Embryonic Stem Cells into Cone Photoreceptors. Bio-protocol 6(14): e1870. DOI: 10.21769/BioProtoc.1870.
Category
Developmental Biology > Cell growth and fate > Neuron
Stem Cell > Embryonic stem cell > Maintenance and differentiation
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link












