- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Sensitive Assessment of Hippocampal Learning Using Temporally Dissociated Passive Avoidance Task
Published: Vol 6, Iss 11, Jun 5, 2016 DOI: 10.21769/BioProtoc.1821 Views: 8835
Reviewed by: Soyun KimAdler R. DillmanXi Feng

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Protocol to Study Spatial Subgoal Learning Using Escape Behavior in Mice
Philip Shamash and Tiago Branco
Jun 20, 2022 2619 Views

Conditioned Lick Suppression: Assessing Contextual, Cued, and Context-cue Compound Fear Responses Independently of Locomotor Activity in Mice
Youcef Bouchekioua [...] Yu Ohmura
Dec 5, 2022 1666 Views

A Protocol to Assess Time-of-Day-Dependent Learning and Memory in Mice Using the Novel Object Recognition Test
Jordan Mar [...] Isabella Farhy-Tselnicker
Sep 20, 2025 2658 Views
Abstract
The temporally dissociated passive avoidance (TDPA) paradigm is a variant of passive avoidance testing, and allows for more sensitive investigation of mild impairments in avoidance learning. Passive avoidance learning measures the latency to enter a “dark” context in which an aversive stimulus (foot shock) has been previously experienced using a light-dark box paradigm. Briefly, the animal is placed into the light side of the box and the time spent to cross into the dark side is measured. After entry into the dark chamber, the animal receives a mild (0.4-1.6 mA) footshock and is removed from the box. After a period of time, typically 24 h (note that this is entirely dependent on whether various levels of memory retention, e.g., short or long, are being measured), the animal is placed back into the box and cross-over latency is measured. Passive avoidance is learned after one trial and results in a robust increase in crossover latency. This behavior requires the association between a normally neutral environment and an aversive stimulus, and is dependent on hippocampal function (Stubley-Weatherly et al., 1996; Impey et al., 1998). TDPA extends this learning across multiple once-daily trials, producing a more graded and malleable latency score, and thus allows a more sensitive evaluation of changes in hippocampal function The task remains dependent on an intact hippocampus (Zhang et al., 2008), and subtle changes in hippocampal gene expression can result in robust alterations in TDPA latency scores (Eagle et al., 2015). We describe here a common method used to assess TDPA learning in mice.
Keywords: MemoryMaterials and Reagents
- Adult (7 weeks or older) mice (C57BL/6J) (the Jackson laboratory)
Note: C57BL/6J mice are typical, though alternate strains and ages of mice may also be used. Mice are housed singly or in groups of 4-5 per cage. Conditions should comply with the Guide for the Care and Use of Laboratory Animals, 8th ed. (https://grants.nih.gov/grants/olaw/Guide-for-the-Care-and-use-of-laboratory-animals.pdf). See Animals considerations in Notes for more details. - Ethanol (70% volume/volume) diluted in distilled water
Equipment
- Light-dark box (14 in. W x 7 in. D x 12 in. H) equipped with overhead houselight (light side only), guillotine-style door, drop pan, and shock-capable grid flooring (Coulbourn Instruments, model: H10-11M-PA )
- Precision animal shocker (Coulbourn Instruments, model: H13-15 ) and cable
- Video camera, IR-capable (Panasonic Corporation of North America, model: WV-CP304 ), mounted in front of box with side-view of light chamber and door
- Timer
- Sound-attenuating cubicle, as needed
Note: For example, Coulbourn Instruments provides a range of isolation cubicles in different sizes (H10-24 series) that are suitable for the needs of this experiment.
Procedure
- Set up equipment (Figure 1). Clean the light-dark chamber with 70% ethanol/water solution on all surface areas and wipe clean. Make sure doorway between light side and dark side is closed. Turn on houselight. Turn on shocker and adjust for pre-determined shock intensity (see below).

Figure 1. Example of a light-dark box - Place mouse in light chamber of the light-dark box. Wait approximately 60 sec for mouse to acclimate to light side. Mice will freely explore the entire light side. Before opening the door, ensure the mouse is facing away from the door (at least 45°) and at least 5 cm from the door. This prevents impulsive entry upon door movement. Note that the door will make an audible click upon opening.
- Raise the door separating the dark side from the light side of the light-dark box. Start the timer to measure cross-over latency, defined as the duration after door opens for mouse to enter the dark side of the light-dark box. Crossover is defined by full body entry (including tail). Stop the timer when the mouse enters the dark side of the chamber.
Note: Mice may enter halfway and dart back into the light side. This is more likely after repeated daily trials. Do not lower the door until the mouse has fully crossed over. - Once the mouse has crossed over, lower the door. After repeated trials, most non-experimental mice (e.g., wild-type C57BL/6J with no manipulation) will reach criterion. Criterion typically consists of 300-600 sec in the light side without any crossover. The determination of the criterion is entirely dependent on the experiment. Once an animal has reached criterion it is manually removed from the light side (no footshock is given). It no longer undergoes additional testing and crossover latency is considered maximal for remaining sessions.
- After a specific amount of time in the dark side (see below), apply a single foot-shock to the grid floor. The amount of time and the shock intensity will determine (affect) cross-over latency in subsequent trials (see Figure 2).
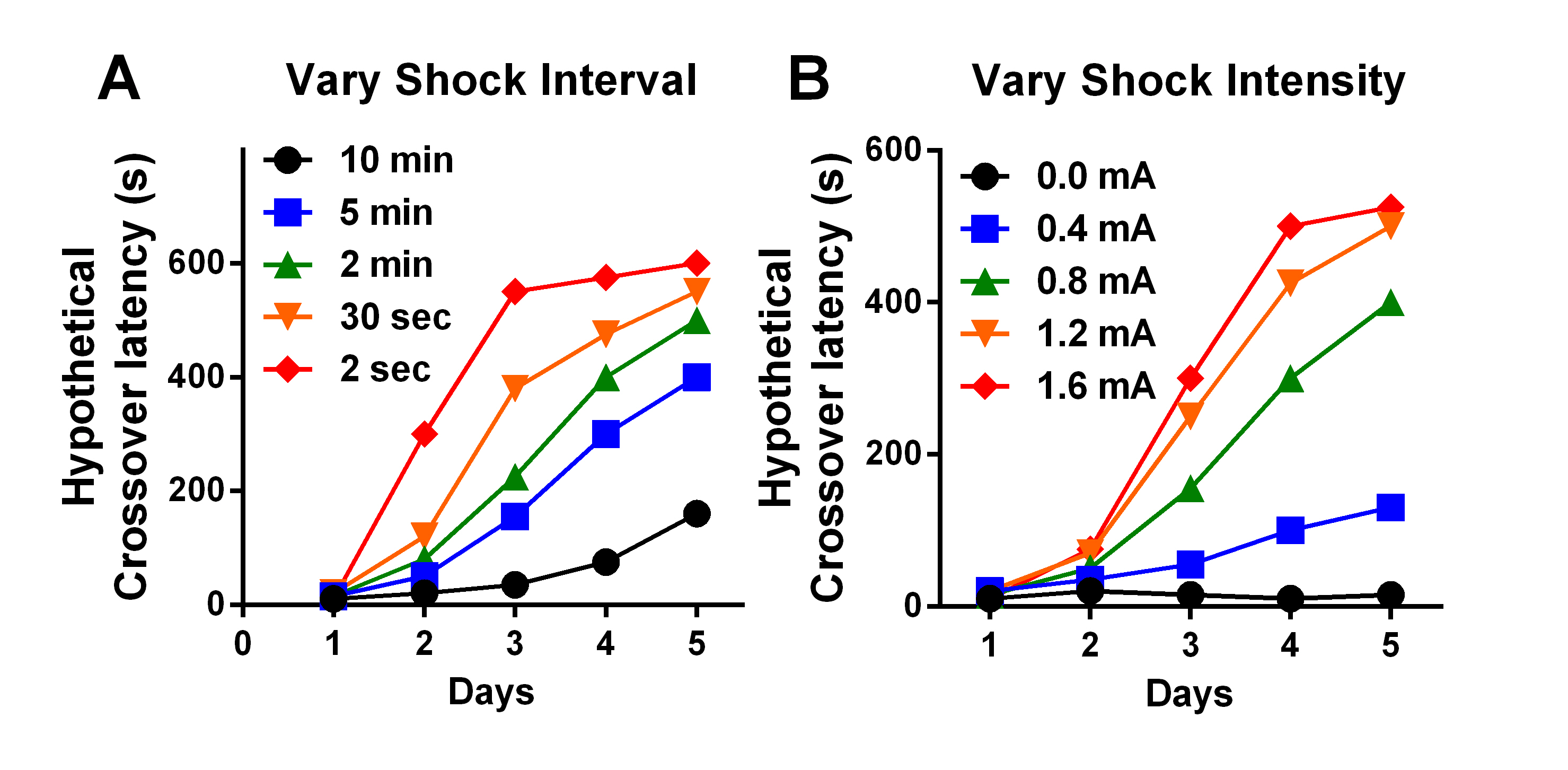
Figure 2. Hypothetical effects of varying delay to shock and shock intensity in the TDPA task. A. Increases in delay to shock (2 sec-10 min) reduce latency to crossover. B. Increases in shock intensity (0.0-1.6 mA) increase latency to crossover. - Allow the mouse to remain in the dark chamber for 30 sec after the foot shock to associate the environment with the aversive stimulus, then remove mouse to home cage.
- All the urine and fecal boli must be removed from both sides of the chamber, and the chamber cleaned entirely with 70% ethanol to remove any residual smell from the first mouse. Afterwards, the next mouse may be submitted to the test.
- Repeat procedure until all mice have been tested.
- Repeat procedure each day for 5 daily sessions.Video 1. Demonstration of temporally dissociated passive avoidance procedure in mice.
Note: This video was recorded with the room lighets on. Normally, room lights are turn off.
Notes
- Determining timing and intensity of shock
Increasing the time between entry into the dark chamber and foot shock and/or decreasing shock intensity can be used to achieve greater sensitivity for detection of mild learning impairments (Figure 2). For instance, if a robust reduction in hippocampal function resulting in an expected increase in crossover latency is hypothesized, a strong shock (1.2 mA) and/or short time before shock (30 sec) could be used to elicit maximal differences between groups. In contrast, if a mild reduction in hippocampal function is hypothesized, a moderate shock (0.4-0.8 mA) and time before shock (5-10 min) could be used to detect a modest reduction in crossover latency, particularly in later sessions. Across five days of TDPA, mild impairment in hippocampal function results in a rightward shift in the learning curve (Eagle et al., 2015). However, it should be expected that increasing time and decreasing intensity will also contribute to greater between-subject variability and across-trial variability. For extended (e.g., 5-day) learning, times ranging from 30 sec to 600 sec and intensity ranging between 0.4-1.2 mA are recommended. Other variables may also alter behavior, including time to criterion. While we provide suggested parameters, we strongly recommend conducting preliminary studies to address the needs of your own experiments. - Measurement/control
The protocol we present is based on a manual setup. However, many hardware and software suppliers allow for alternative methods of measurement and testing control. Measurements can be achieved by automated video tracking or photobeam systems. Typical measurements consist of crossover latency across trials (Figure 2) and survival graph depicting the % of animals (per experimental condition) reaching criterion across trials. No behavior is recorded during the shock because the mouse is in an opaque dark chamber. Crossover latency to enter the dark side (from the light side) is the only behavior that is assessed. - Environment and other factors
It is critical to control all aspects of the environment in order to achieve consistent, replicable results. Testing should always occur in a controlled environment with minimal extraneous sounds or visual and olfactory stimuli. This is best achieved by conducting testing in a quiet, dim room and may be further enhanced by placing the light-dark box in a sound attenuating cubicle. As circadian condition has been shown to affect learning and memory, we recommend that training and testing be performed with fixed time span for all sessions (e.g., 2-4 h into light cycle to determine behavior in the inactive phase or 2-4 h into dark cycle for the active phase). - Animal considerations
It is important to conduct preliminary studies in order to characterize the behavioral phenotype with particular equipment, environment, and animals. This is especially true in the case of varying mouse strains, either inbred or outbred, if mice have been surgically or behaviorally manipulated prior to TDPA testing, or for determining differences in juvenile mice or sex effects. If factors such as genetic or pharmacological alteration are involved, it is important to determine whether these factors affect sensitivity to foot shock and spontaneous behavior in the light-dark box. We recommend first running a 5-10 min light-dark box test to examine the number of transitions between the two chambers and the total time spent in the light chamber. In addition, some labs prefer to pre-handle mice for 5-7 days and singly house the animals prior to TDPA training/testing.
Acknowledgments
This behavioral procedure was adapted from previously published studies (Wang et al., 2004; Zhang et al., 2008; Wang et al., 2009) and was performed by our group as described (Eagle et al., 2015). This work was supported by the Whitehall Foundation (AJR; 2013-08-43), the Multidisciplinary Training in Environmental Toxicology training grant (ALE; T32-ES007255), a 2014 NARSAD Young Investigator Award from the Brain and Behavior Research Foundation (ALE), and NIH (HW, MH093445).
References
- Eagle, A. L., Gajewski, P. A., Yang, M., Kechner, M. E., Al Masraf, B. S., Kennedy, P. J., Wang, H., Mazei-Robison, M. S. and Robison, A. J. (2015). Experience-dependent induction of hippocampal DeltaFosB controls learning. J Neurosci 35(40): 13773-13783.
- Impey, S., Smith, D. M., Obrietan, K., Donahue, R., Wade, C. and Storm, D. R. (1998). Stimulation of cAMP response element (CRE)-mediated transcription during contextual learning. Nat Neurosci 1(7): 595-601.
- Institute of Laboratory Animal Resources (U.S.) (2011). Guide for the care and use of laboratory animals, 8th edition. Washington DC: National Academy of Sciences.
- Stubley-Weatherly, L., Harding, J. W. and Wright, J. W. (1996). Effects of discrete kainic acid-induced hippocampal lesions on spatial and contextual learning and memory in rats. Brain Res 716(1-2): 29-38.
- Wang, B., Hu, Q., Hearn, M. G., Shimizu, K., Ware, C. B., Liggitt, D. H., Jin, L. W., Cool, B. H., Storm, D. R. and Martin, G. M. (2004). Isoform-specific knockout of FE65 leads to impaired learning and memory. J Neurosci Res 75(1): 12-24.
- Wang, Y., Zhang, M., Moon, C., Hu, Q., Wang, B., Martin, G., Sun, Z. and Wang, H. (2009). The APP-interacting protein FE65 is required for hippocampus-dependent learning and long-term potentiation. Learn Mem 16(9): 537-544.
- Zhang, M., Moon, C., Chan, G. C., Yang, L., Zheng, F., Conti, A. C., Muglia, L., Muglia, L. J., Storm, D. R. and Wang, H. (2008). Ca-stimulated type 8 adenylyl cyclase is required for rapid acquisition of novel spatial information and for working/episodic-like memory. J Neurosci 28(18): 4736-4744.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Eagle, A. L., Wang, H. and Robison, A. J. (2016). Sensitive Assessment of Hippocampal Learning Using Temporally Dissociated Passive Avoidance Task. Bio-protocol 6(11): e1821. DOI: 10.21769/BioProtoc.1821.
-
Eagle, A. L., Gajewski, P. A., Yang, M., Kechner, M. E., Al Masraf, B. S., Kennedy, P. J., Wang, H., Mazei-Robison, M. S. and Robison, A. J. (2015). Experience-dependent induction of hippocampal DeltaFosB controls learning. J Neurosci 35(40): 13773-13783.
Category
Neuroscience > Behavioral neuroscience > Learning and memory
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link