- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Preparation of Synovial Mesenchymal Stem Cells from a Rat Knee Joint
Published: Vol 6, Iss 9, May 5, 2016 DOI: 10.21769/BioProtoc.1799 Views: 10209
Reviewed by: Hui ZhuAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
A Full Good Manufacturing Practice–Compliant Protocol for Corneal Stromal Stem Cell Cultivation
Mithun Santra [...] Gary H.F. Yam
Sep 20, 2024 2181 Views
Spheroid Sheets: A Scalable Platform for Producing Tissue Membrane Constructs
Quang Bach Le [...] Deepak Choudhury
Nov 20, 2025 1580 Views
A Protocol to Induce Brown and Beige Adipocyte Differentiation From Murine and Human Adipose-Derived SVF
Rohit Raj Yadav [...] Narendra Verma
Dec 5, 2025 1707 Views
Abstract
Mesenchymal stem cells (MSCs), first described in human bone marrow, are emerging as promising cell-based therapeutics for a wide range of diseases (Caplan and Correa, 2011). MSCs have been isolated from various organs in the body, and synovial MSCs were first reported by De Bari et al. (2001). We previously reported that synovial MSCs have superior proliferation and chondrogenic potentials as compared to bone marrow-, muscle-, and adipose- derived MSCs in humans (Sakaguchi et al., 2005) and rats (Yoshimura et al., 2007). In addition, administration of synovial MSCs for osteochondral defect promoted cartilage regeneration in a rabbit (Koga et al., 2008) and a pig model (Nakamura et al., 2012). In 2008, we started a clinical trial in human and obtained satisfactory results of symptoms and regenerated cartilage by Magnetic Resonance Imaging (Sekiya et al., 2015). We have also engaged in multiple research lines using synovial MSCs for meniscus regeneration in rats (Horie et al., 2009; Horie et al., 2012; Katagiri et al., 2013; Okuno et al., 2014; Ozeki et al., 2015). In this article, we demonstrated how to harvest the synovium including infrapatellar fat pad from a rat knee joint, and to describe the technique of isolation and culture of rat synovial MSCs.
Keywords: SynoviumMaterials and Reagents
- Culture dish (culture area: 56.7 cm2, diameter: 100 mm) (Thermo Fisher Scientific, catalog number: 150350 )
- Cell strainer (70 µm) (VWR International, Greiner Bio-One GmbH, catalog number: 89508-344 )
- Clip (Mitsuya, catalog number: GM-590 ) cut by pliers in a half size (Figure 1B)
- Styrene foam (RIKAKEN) (Figure 1C)
- 50 ml Falcon tube (Corning, catalog number: 352070 )
- 8-12 weeks old Lewis rat (Charles River Laboratories International)
- Collagenase V (Wako Pure Chemical Industries, catalog number: 038-17851 )
- α-minimal essential medium (α-MEM) (Thermo Fisher Scientific, catalog number: 12561-056 )
- Fetal bovine serum (FBS) (Thermo Fisher Scientific, catalog number: 12483-020 )
- Streptomycin, Penicillin, Amphotericin B (Antibiotic-Antimycotic, 100x) (Thermo Fisher Scientific, Gibco™, catalog number: 15240-062 )
- Phosphate buffer saline (PBS) (Thermo Fisher Scientific, Gibco™, catalog number: 14190-235 )
- α-MEM containing 10% FBS with antibiotics (see Recipes)
- 3 mg/ml Collagenase solution (for synovium from both knee joints) (see Recipes)
Equipment
- 37 degree, 5% CO2 forced-air incubator (ASTEC, catalog number: SCA-165DS )
- Centrifuge machine (KUBOTA Corporation, model: Model 8730 )
- Scalpel holder (Natsume, catalog number: No.3 D-11 ) with blade (Natsume, catalog number: No.11 D-13 ), scissors (Natsume, catalog number: B-12 ), and tweezers (Natsume, catalog number: A-6 ) (Figure 1A)
Procedure
- Preparation of the surgery (10 min)
- Sacrifice a rat by delivering CO2 in a vinyl bag.
- Remove the hair around the knee joints, and sterile clean the legs with 70% ethanol (Figure 1D).
- Put the rat on the styrene foam, and cover the sterile sheet on the rat, keeping the right leg out of the hole. Stabilize the ankle with clip on the styrene foam (Figure 1E).
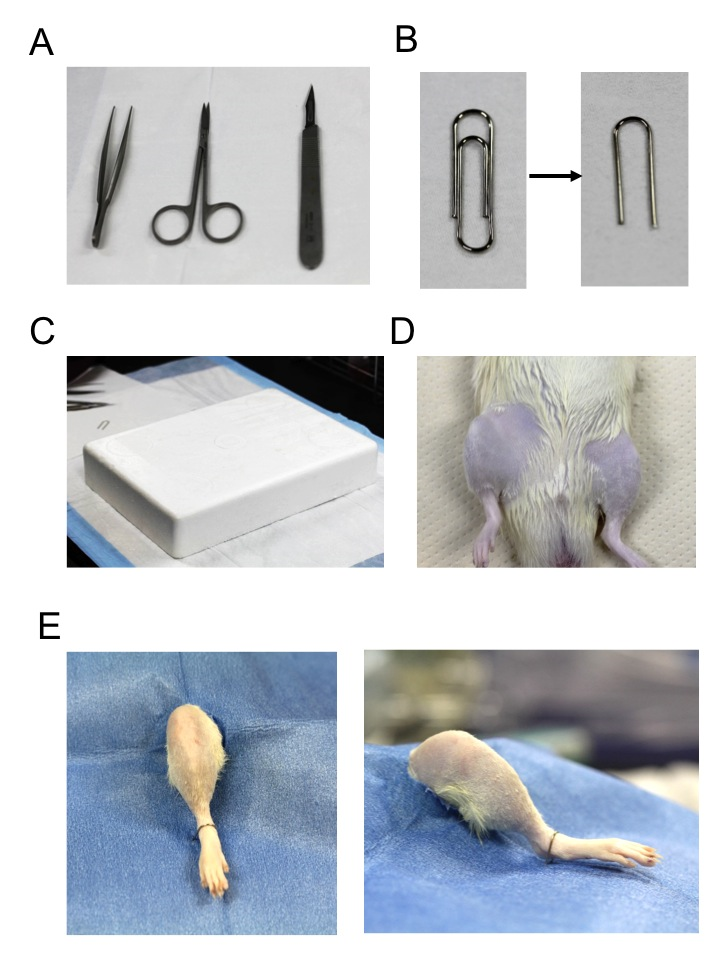
Figure 1. Preparation of the surgery. A. Surgical instruments; B. Clip for stabilization of legs; C. Styrene form; D. Removal of the hair; E. Settings of the legs.
- Sacrifice a rat by delivering CO2 in a vinyl bag.
- Harvesting the synovium from the knee joint (Video 1) (10 min for both knee joints)
- Expose the patellar tendon with straight skin incision.
- Cut the patellar tendon transversely, and peel the tendon upward and downward to expose the infrapatellar fat pad.
- Separate the infrapatellar fat pad from the femur and tibia.
- Put the synovium into PBS in a Falcon tube.Video 1. Harvesting the synovium including infrapatellar fat pad from the right knee
- Expose the patellar tendon with straight skin incision.
- Isolation and culture of synovial MSCs
- Mince the synovium with a scalpel into 2-3 mm pieces.
- Put the minced synovium in collagenase solution.
- Place the tube in the incubator for 2-3 h. Shake it 2-3 times per hour.
- Filter the digested solution through a cell strainer.
- Centrifuge the tube at 580 x g for 5 min.
- Remove the supernatant, and plate the cells on the 56.7 cm2 dish (about 1,000 cells/ cm2).
- Change the medium twice a week and culture for 2 weeks with α-MEM containing 10% FBS with antibiotics.
- Mince the synovium with a scalpel into 2-3 mm pieces.
Representative data
We can usually harvest 1 x 104 cells from one synovium (Figure 2). After 2 weeks, we can obtain 5 x 105-1 x 106 MSCs from one dish. The cells are heterogenous, and we usually use these cells of passage 2-4 in the experiments. We reported these data in our previous reports (Yoshimura et al., 2007).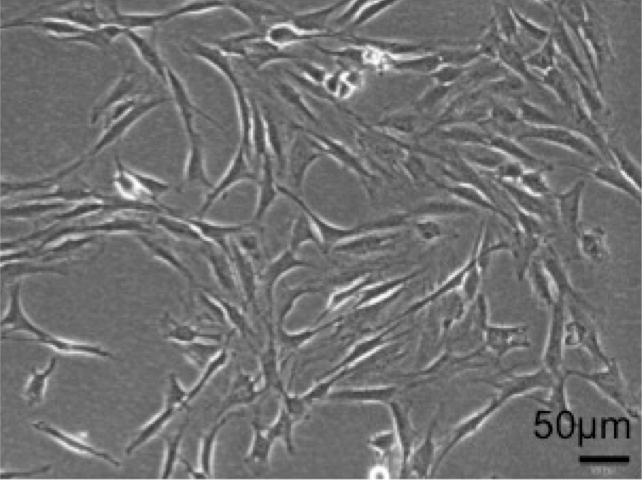
Figure 2. Representative morphology of synovial MSCs (passage 1, day 7). Scale bar = 50 µm
Recipes
- α-MEM containing 10% FBS with antibiotics
α-MEM 445 ml
FBS 50 ml
100x Antibiotic-Antimycotic 5 ml - Collagenase solution 3 mg/ml (for synovium from both knee joints)
α-MEM 3 ml
Collagenase V 9 mg
Acknowledgments
We have no conflict of interest to prepare this article. This study was supported by the Highway Program for Realization of Regenerative Medicine from the Japan Agency for Medical Research and Development (AMED) to IS.
References
- Caplan, A. I. and Correa, D. (2011). The MSC: an injury drugstore. Cell Stem Cell 9(1): 11-15.
- De Bari, C., Dell'Accio, F., Tylzanowski, P. and Luyten, F. P. (2001). Multipotent mesenchymal stem cells from adult human synovial membrane. Arthritis Rheum 44(8): 1928-1942.
- Horie, M., Sekiya, I., Muneta, T., Ichinose, S., Matsumoto, K., Saito, H., Murakami, T. and Kobayashi, E. (2009). Intra-articular Injected synovial stem cells differentiate into meniscal cells directly and promote meniscal regeneration without mobilization to distant organs in rat massive meniscal defect. Stem Cells 27(4): 878-887.
- Horie, M., Choi, H., Lee, R. H., Reger, R. L., Ylostalo, J., Muneta, T., Sekiya, I. and Prockop, D. J. (2012). Intra-articular injection of human mesenchymal stem cells (MSCs) promote rat meniscal regeneration by being activated to express Indian hedgehog that enhances expression of type II collagen. Osteoarthritis Cartilage 20(10): 1197-1207.
- Koga, H., Shimaya, M., Muneta, T., Nimura, A., Morito, T., Hayashi, M., Suzuki, S., Ju, Y. J., Mochizuki, T. and Sekiya, I. (2008). Local adherent technique for transplanting mesenchymal stem cells as a potential treatment of cartilage defect. Arthritis Res Ther 10(4): R84.
- Katagiri, H., Muneta, T., Tsuji, K., Horie, M., Koga, H., Ozeki, N., Kobayashi, E. and Sekiya, I. (2013). Transplantation of aggregates of synovial mesenchymal stem cells regenerates meniscus more effectively in a rat massive meniscal defect. Biochem Biophys Res Commun 435(4): 603-609.
- Nakamura, T., Sekiya, I., Muneta, T., Hatsushika, D., Horie, M., Tsuji, K., Kawarasaki, T., Watanabe, A., Hishikawa, S., Fujimoto, Y., Tanaka, H. and Kobayashi, E. (2012). Arthroscopic, histological and MRI analyses of cartilage repair after a minimally invasive method of transplantation of allogeneic synovial mesenchymal stromal cells into cartilage defects in pigs. Cytotherapy 14(3): 327-338.
- Okuno, M., Muneta, T., Koga, H., Ozeki, N., Nakagawa, Y., Tsuji, K., Yoshiya, S. and Sekiya, I. (2014). Meniscus regeneration by syngeneic, minor mismatched, and major mismatched transplantation of synovial mesenchymal stem cells in a rat model. J Orthop Res 32(7): 928-936.
- Ozeki, N., Muneta, T., Matsuta, S., Koga, H., Nakagawa, Y., Mizuno, M., Tsuji, K., Mabuchi, Y., Akazawa, C., Kobayashi, E., Saito, T. and Sekiya, I. (2015). Synovial mesenchymal stem cells promote meniscus regeneration augmented by an autologous Achilles tendon graft in a rat partial meniscus defect model. Stem Cells 33(6): 1927-1938.
- Sakaguchi, Y., Sekiya, I., Yagishita, K. and Muneta, T. (2005). Comparison of human stem cells derived from various mesenchymal tissues: superiority of synovium as a cell source. Arthritis Rheum 52(8): 2521-2529.
- Sekiya, I., Muneta, T., Horie, M. and Koga, H. (2015). Arthroscopic transplantation of synovial stem cells improves clinical outcomes in knees with cartilage defects. Clin Orthop Relat Res 473(7): 2316-2326.
- Yoshimura, H., Muneta, T., Nimura, A., Yokoyama, A., Koga, H. and Sekiya, I. (2007). Comparison of rat mesenchymal stem cells derived from bone marrow, synovium, periosteum, adipose tissue, and muscle. Cell Tissue Res 327(3): 449-462.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Ozeki, N., Muneta, T., Mizuno, M. and Sekiya, I. (2016). Preparation of Synovial Mesenchymal Stem Cells from a Rat Knee Joint. Bio-protocol 6(9): e1799. DOI: 10.21769/BioProtoc.1799.
Category
Stem Cell > Adult stem cell > Mesenchymal stem cell
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link