- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Telomere Dysfunction Induced Foci (TIF) Analysis
Published: Vol 5, Iss 22, Nov 20, 2015 DOI: 10.21769/BioProtoc.1656 Views: 13175
Reviewed by: HongLok LungVanesa Olivares-IllanaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Telomerase Repeated Amplification Protocol (TRAP)
Ilgen Mender and Jerry W. Shay
Nov 20, 2015 23240 Views

Telomere Restriction Fragment (TRF) Analysis
Ilgen Mender and Jerry W. Shay
Nov 20, 2015 22174 Views
Abstract
Telomerase maintains telomeric DNA in eukaryotes during early developments, ~90% of cancer cells and some proliferative stem like cells. Telomeric repeats at the end of chromosomes are associated with the shelterin complex. This complex consists of TRF1, TRF2, Rap1, TIN2, TPP1, POT1 which protect DNA from being recognized as DNA double-stranded breaks. Critically short telomeres or impaired shelterin proteins can cause telomere dysfunction, which eventually induces DNA damage responses at the telomeres. DNA damage responses can be identified by antibodies to 53BP1, gammaH2AX, Rad17, ATM, and Mre11. DNA damage foci at uncapped telomeres are referred to as Telomere dysfunction-Induced Foci (TIFs) (de Lange, 2005; Takai et al., 2003).
The TIF assay is based on the co-localization detection of DNA damage by an antibody against DNA damage markers, such as gamma-H2AX, and telomeres using an antibody against one of the shelterin proteins such as TRF2 (Takai et al., 2003; de Lange, 2002; Karlseder et al., 1999). The method we describe here can be used in normal human and cancer cells.
Other commonly used methods-Telomere Restriction Fragment (TRF) Analysis (Mender and Shay, 2015b) and Telomere Repeat Amplification Protocol (TRAP) (Mender and Shay, 2015a)- in telomere biology can be found by clicking on the indicated links.
Materials and Reagents
- Lab-Tek II Chamber slides (Thomas Scientific, catalog number: 154461 )
- Cancer or normal human cells
- Triton X-100 (J.T. Baker Chemical Co., catalog number: 7-x198 )
Octyl Phenol Ethoxylate (Avantor Performance Materials, J.T. Baker, catalog number: X198-07 ) - Paraformaldehyde (PFA) (Sigma-Aldrich, catalog number: P6148 )
- Nonidet-P40 (Fluka BioChemika, catalog number: 74385 )
Note: Currently, it is “Sigma-Aldrich, catalog number: 74385”. - Fish gelatin blocking buffer, 10% (Amresco, catalog number: M319 )
- Bovine Serum Albumin (BSA), Fraction V (Gemini Bio-Products, catalog number: 700-106P )
- Anti-phospho-histone H2A.X (Ser139, mouse) Antibody (Merck Millipore Corporation, catalog number: 05-636 )
- Anti-TRF2 Antibody [EPR3517 (Ouellette et al., 2000)] (rabbit) (Abcam, catalog number: ab108997 )
- Donkey anti-Rabbit IgG (H+L) Secondary Antibody, Alexa Fluor® 488 conjugate (Invitrogen, catalog number: A-21206 )
Note: Currently, it is “Thermo Fisher Scientific, NovexTM, catalog number: A-21206”. - Goat anti-Mouse IgG (H+L) Secondary Antibody, Alexa Fluor® 568 conjugate (Invitrogen, catalog number: A-11004 )
Note: Currently, it is “Thermo Fisher Scientific, NovexTM, catalog number: A-11004”. - Vectashield® Mounting Medium with DAPI (Vector Laboratories, catalog number: H-1200 )
- Deltavision® Immersion Oil N=1.518 (GEHC, part no: 290291717 )
- Phosphate-buffered saline (PBS)
- MilliQ® water
- 10x PBS buffer-phosphate buffer saline (see Recipes)
- 1x PBST buffer (see Recipes)
Equipment
- Personal DeltaVision wide-field fluorescent microscope (GE Healthcare, model: PD11435 )
- Lamp (Xenon)
- Camera (Photometrics)
- 60x/1.42 N.A. (numerical aperture) objective (Olympus)
Software
- DeltaVision® SoftWoRx software (GE Healthcare)
- Autoquant® Software (Media Cybernetics)
- Imaris® Software (Bitplane Imaris)
Procedure
- Sample preparation
- Seed cells on chamber slides and incubate them until they are at the desired confluency (~70% confluency). However, It is important to seed them well separated to analyze individual cells. Avoid overlapping cells.
Note: The amount described here is for a two well chamber slide. It can be adjusted to differing sizes of chamber slides. Shaking is not required for washing steps. - Wash cells with 1 ml 1x PBS for 5 min.
- Fix cells with 4% paraformaldehyde (600 μl) for 10 min.
- Wash cells twice with 1 ml 1x PBS for 5 min.
- Permeabilize cells with 0.5% Nonidet-P40 (600 μl) for 10 min at room temperature.
- Wash cells three times with 1 ml 1x PBS for 5 min.
- Incubate cells with blocking solution (0.2% fish gelatin and 0.5% BSA) (600 μl) for 30 min at room temperature to reduce nonspecific binding.
Note: Fish gelatin needs to be warmed up to room temperature before use. - Incubate cells with primary antibodies (450 μl/well) diluted in blocking solution for either 1 h at room temperature or overnight at 4 °C in humidified chamber.
Primary antibodies: gamma-H2AX: 1:1,000 dilution
TRF2: 1:250 dilution - Wash cells three times with 1x PBST for 5 min.
- Wash cells three times with 1x PBS for 5 min.
- Incubate cells with secondary antibodies (1:500 dilution for each) diluted in blocking solution for 40 min (450 μl/well) at room temperature.
- Wash cells six times with 1 ml 1x PBS for 5 min.
- Remove the chamber portion of slide.
- Counterstain with DAPI and seal the edges of the slides with nail polish.
Notes:- The bottle of mounting medium is supplied with a screw cap that has a drop dispenser pipet. One drop of mounting medium, approximately equal to 25 μl, is dispensed on the slide and then coverslipped.
- Coverslip should be carefully inverted to a drop of mounting medium on microscope slides to allow the mounting medium disperse over the slide.
- The bottle of mounting medium is supplied with a screw cap that has a drop dispenser pipet. One drop of mounting medium, approximately equal to 25 μl, is dispensed on the slide and then coverslipped.
- The slides can be viewed immediately after drying or stored at 4 °C up to a month/ -20 °C for a longer time.
- Image on a fluorescent microscope.
- Seed cells on chamber slides and incubate them until they are at the desired confluency (~70% confluency). However, It is important to seed them well separated to analyze individual cells. Avoid overlapping cells.
- Imaging with fluorescent microscope
Images can be acquired using a Personal DeltaVision® wide-field fluorescent microscope with an Olympus® 60x/1.42 N.A objective and a Coolsnap® HQ2 camera with an image size of 1,024 x 1,024. For best resolution, set the bin value for the camera to be 1 x 1 resulting in a pixel size of 0.1077 μm using the 60x/1.42 N.A objective. The optical section spacing between each z-stack should be approximately 0.15 μm. At least three channels can be sequentially captured with the TRITC (excitation: 555/28 nm, emission: 617/73 nm), FITC (excitation: 490/20 nm, emission: 528/38 nm) and DAPI (excitation: 360/40 nm, emission: 457/50 nm) filterset. Exposure for each of those channels is selected such that it was well below the saturation limit of 4,095 for the maximum intensity value in that image. - Image analysis
Before analyzing images for co-localization of two different antibodies, the resolution of the z-stacks can be improved by de-convolving using a blind de-convolution algorithm in AutoquantX3® software. De-convolution is a computational method to process images, which are captured in a microscope by using series of optical sections (z-stacks), in a better contrast and resolution. During de-convolution, these series of optical sections are combined in three dimensions and improves the image quality by removing the blurry effect of microscope (Goodwin, 2014). Then, a background subtraction filter is applied in Imaris® software to improve the quality of the images before running the co-localization analysis. It is useful to maintain the same background subtraction settings for all images. - Co-localization analysis
Co-localization analysis is performed using a Bitplane Imaris. Background subtraction filter to improve the quality of the images and baseline subtraction filter to subtract the estimated baseline from the data. Coloc algorithm with channel 1 (gamma-H2AX) and channel 2 (TRF2) is selected in Imaris® (Costes et al., 2004). A Region of Interest (ROI) is selected using channel 3 (DAPI). The ROI is thresholded allowing the investigator to select the signals that are inside of the nucleus. Any signal represented by dashed lines is considered as background signal (Costes et al., 2004) (Figure 1). The algorithm allows calculation of the threshold values for channel 1 (gamma-H2AX) and channel 2 (TRF2) according to automatic co-localization analysis thus removing user bias. After automatic thresholding of both channels, a new coloc channel, channel 4, is built with ‘Build Coloc Channel’ button in the coloc analysis algorithm. After creating a surface for each nucleus (channel 3, DAPI), Spot function is used in Imaris® to generate new spots for channel 1 (gamma-H2AX), channel 2 (TRF2), and channel 4 (coloc). For creating new spots in the previous step, we generally select 0.2 microns as the diameter for the spots. The surface that is created earlier for Channel 3 (DAPI) is chosen as the ROI and a Split Spots into Surface Objects function, which is a matlab extension in Imaris, is applied to generate new spots. Using the above steps, the number of spots for each protein in every nucleus is calculated individually.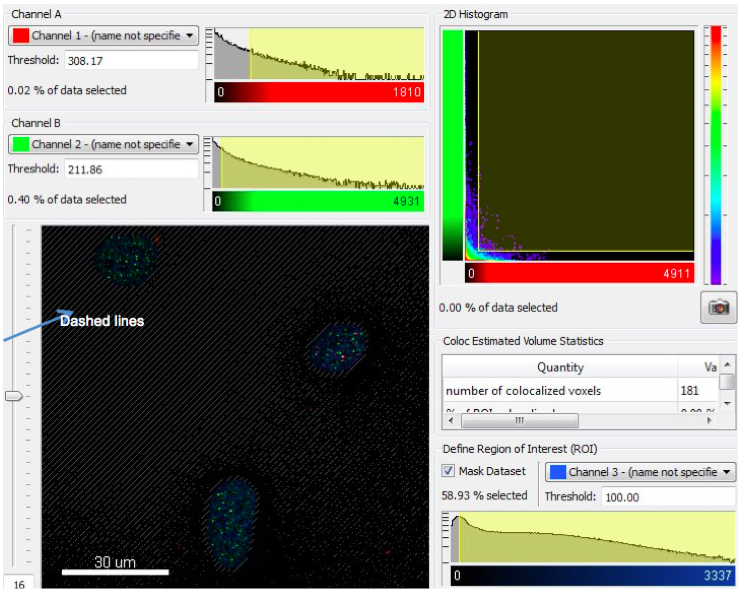
Figure 1. This is an example figure to show 2D Histogram and the Define Region of Interest (ROI) function. The ROI using Channel 3 (DAPI) is thresholded such that the signal outside the nucleus represented by dashed lines is not used for the co-localization analysis calculations. This image does not reflect actual biology and is only for illustrative purposes.
Steps for colocalization analysis in imaris software


Recipes
- 10x PBS buffer-phosphate buffer saline
Powder for 5 L of 10x is ready to use for preparation of 5 L of concentrated 10x phosphate-buffered saline (PBS)
Prepare 5 L milliQ® water and a PBS powder in it
Then add large stir bar and place on stirrer until solids are dissolved - 1x PBST buffer
1x PBS with 0.1% Triton
Acknowledgments
Some of these protocols were adapted from previously published studies. We thank Zeliha Gunnur Dikmen for her help in acquisition of TRAP gel and Abhijit Bugde from the Live Cell Imaging Facility at UT Southwestern for his assistance with the imaging and analysis part of Telomere dysfunction Induced Foci (TIF) analysis.
References
- Costes, S. V., Daelemans, D., Cho, E. H., Dobbin, Z., Pavlakis, G. and Lockett, S. (2004). Automatic and quantitative measurement of protein-protein colocalization in live cells. Biophys J 86(6): 3993-4003.
- de Lange, T. (2002). Protection of mammalian telomeres. Oncogene 21(4): 532-540.
- de Lange, T. (2005). Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev 19(18): 2100-2110.
- Goodwin, P. C. (2014). Quantitative deconvolution microscopy. Methods Cell Biol 123: 177-192.
- Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. and de Lange, T. (1999). p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283(5406): 1321-1325.
- Mender, I. and Shay, J. W. (2015a). Telomerase repeated amplification protocol (TRAP). Bio-protocol 5(22): e1657.
- Mender, I. and Shay, J. W. (2015b). Telomere restriction fragment (TRF) analysis. Bio-protocol 5(22): e1658.
- Takai, H., Smogorzewska, A. and de Lange, T. (2003). DNA damage foci at dysfunctional telomeres. Curr Biol 13(17): 1549-1556.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Mender, I. and Shay, J. W. (2015). Telomere Dysfunction Induced Foci (TIF) Analysis. Bio-protocol 5(22): e1656. DOI: 10.21769/BioProtoc.1656.
Category
Cancer Biology > Replicative immortality > Cell biology assays > Nucleic acid sequencing
Cancer Biology > Replicative immortality > Cell biology assays > Protein
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link













