- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Isolation of Murine Adipose Tissue-derived Mesenchymal Stromal Cells (mASCs) and the Analysis of Their Proliferation in vitro
Published: Vol 5, Iss 21, Nov 5, 2015 DOI: 10.21769/BioProtoc.1642 Views: 11722
Reviewed by: Agnieszka PastulaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Assessing the Presence of Hematopoietic Stem and Progenitor Cells in Mouse Spleen
Isabelle J. Marié [...] David E. Levy
Jun 5, 2022 3810 Views
Thrombopoietin-independent Megakaryocyte Differentiation of Hematopoietic Progenitor Cells from Patients with Myeloproliferative Neoplasms
Chloe A. L. Thompson-Peach [...] Daniel Thomas
Jan 20, 2023 2384 Views
Isolation of Human Bone Marrow Non-hematopoietic Cells for Single-cell RNA Sequencing
Hongzhe Li [...] Stefan Scheding
Jun 20, 2024 2609 Views
Abstract
Mesenchymal stromal cells (MSCs) are non-hematopoietic, perivascular cells which support hematopoiesis and are thought to participate in tissue repair in vivo. MSCs can be isolated from various tissues and organs and are defined in vitro as plastic adherent cells expressing CD73, CD90, CD105 (human MSCs) or CD29, CD44, sca-1 (murine MSCs) which can differentiate into osteoblasts, adipocytes, chondroblasts and myocytes. MSCs possess potent immunomodulatory and trophic capacities in vitro and in vivo and have thus emerged as a promising treatment of inflammatory/autoimmune diseases. The use of MSCs for human disease relies on the injection of a large number of cells and much effort has been focused on acquiring MSCs with high proliferative capacity. Thus, establishing simple and accurate protocols for measuring MSC proliferation is of importance for both basic and applied research. The current protocol provides details on how to isolate and measure the proliferation of murine MSCs derived from inguinal and/or intraabdominal adipose tissue (mASCs) using the xCELLigence system and CellTiter-Blue reagent (Carrillo-Galvez et al., 2015; Anderson et al., 2013). The protocols described below can also be easily translated to human MSCs.
Materials and Reagents
- Scalpel blades No. 24 (VWR International, catalog number: 233-5487 )
- Cell strainers (100 μm and 70 μm) (BD Biosciences, catalog numbers: 352360 and 352350 )
Note: Currently, it is “Corning , catalog numbers: 352360 and 352350”. - Black 96-well plate (Greiner Bio-One GmbH, catalog number: 655076 )
- 16-well E-plate (E-plate 16) (ACEA BIO, catalog number: 06465382001 )
- Tissue culture treated Falcon 96-well plates (BD Biosciences, catalog number: 353072 )
Note: Currently, it is “Corning, catalog number: 353072”. - NuncTM Cell Culture Treated EasYFlasksTM (Thermo Fisher Scientific, catalog number: 156499 )
- BALB/c (or any strain of choice) 8-12 weeks old male or female mice (Charles River Laboratories)
- Hank’s balanced salt solution (HBSS) w/Calcium w/Magnesium (Biowest, catalog number: L0612-500 )
- Collagenase Type I (Sigma-Aldrich, catalog number: C0130-100MG )
- MesenCultTM Proliferation Kit with MesenPureTM (Mouse) (STEMCELL Technologies, catalog number: 05512 )
- Penicillin-Streptomycin 10,000 U/ml (Life Technologies, Gibco, catalog number: 15140-122 )
Note: Currently, it is “Thermo Fisher Scientific, GibcoTM catalog number: 15140-122”. - Stable Glutamine 200 mM (Biowest, catalog number: X0551-100 )
- DPBS w/o Calcium w/o Magnesium (Biowest, catalog number: L0615-500 )
- TrypLETM Express Enzyme (1x) (Life Technologies, catalog number: 12604021 )
Note: Currently, it is “Thermo Fisher Scientific, GibcoTM catalog number: 12604021”. - Fetal Bovine Serum (FBS) (Thermo Fisher Scientific, catalog number: 10309433 )
- Trypan blue (Sigma-Aldrich, catalog number: T8154-20 ml )
- CellTiter-Blue reagent (Promega Corporation, catalog number: G8081 )
- Collagenase Type I (see Recipes)
- Complete MesenCult medium (see Recipes)
Equipment
- Sterile stainless steel scissors and forceps
- Neubauer cell counting chamber (VWR International, catalog number: 631-1111 )
- Cell culture incubator (THERMO, model: HEPA class 100 )
- Phase contrast microscope (OLYMPUS, model: CKX41 )
- Centrifuge for cells (Eppendorf, model: 5810R )
- xCELLigence RTCA station and control unit (lap top) with the RTCA Software (ACEA BIO/ROCHE)
- Fluorometro [Glomax multidetection system (Promega Corporation) or equivalent]
Procedure
- Isolation of mASCs
Mesenchymal stromal cells are isolated from inguinal (subcutaneous) and gonadal (intraabdominal fat deposits attached to the epididymis/testes in male mice and ovaries/uterus in female mice) fat pads.- Sacrifice the mice by cervical dislocation and aseptically remove fat tissue and collect it in a 50 ml tube containing 10 ml of ice cold HBSS (Note 1).
- Wash the fat tissue twice in 10 ml HBSS containing 2% (v/v) of penicillin/streptomycin at room temperature (RT) and transfer the fat to a Petri dish. Remove as much liquid as possible and weigh the fat. Cut the fat tissue using sterile scissors or scalpel blades into small pieces (≤ 3 mm3).
- Resuspend the minced fat tissue in 2.5 ml HBSS containing 2 mg/ml collagenase Type I (equivalent to 1-5 FALGPA units and ≥ 625 collagen digestion units (CDU)/g of fat tissue) and transfer the mixture to a 15 ml tube (< 5 ml) or 50 ml tube (≥ 5 ml). Place the tube in a 37 °C water bath for 30 min; gently vortex tube for 10 sec every 5 min.
- Add 10 ml of HBSS to the digested fat solution and filter it using 100 μm cell strainers (Note 2).
- Spin down the released cells (300 x g, 7 min at 4 °C), aspirate the supernatant (Note 3) and resuspend the pellet in 10 ml HBSS.
- Filter the cell suspension using a 70 μm cell strainer and pellet the cells by centrifugation (300 x g, 7 min at 4 °C).
- Resuspend the pellet in 2 ml complete Mesencult medium and count cells using Trypan blue. We generally obtain 0.25 ± 0.03 g fat/mouse (mean ± SEM, n = 6) and 1.9 ± 0.4 x 106 cells/g fat (mean ± SEM, n = 11).
- Plate the released cells in tissue culture-treated Nunc EasYFlasks at 20-30,000 cells/cm2 in complete Mesencult medium.
- After 24 h, change the medium in order to remove non-attached cells and debris.
- Sacrifice the mice by cervical dislocation and aseptically remove fat tissue and collect it in a 50 ml tube containing 10 ml of ice cold HBSS (Note 1).
- Cell culture
Culture the adherent cells using complete Mesencult at 37 °C, 5% CO2, 21% O2, 90% relative humidity (RH) in 75 cm2 (T75) Nunc EasYFlask at 10,000 cells/cm2.- When cultures reach 80-90% confluency, wash the cells twice with 10 ml PBS.
- Add 2 ml TrypLE/T75 flask and incubate at 37 °C for 5 min. Tap firmly on the side of the bottle in order to detach the cells and then add 8 ml of PBS containing 2% FBS.
- Resuspend the cells gently until obtaining a homogenous cell suspension and spin down the cells at 300 x g for 5 min.
- Count the cells as described below and seed the cells at 10,000 cells/cm2 and expand the culture until it reaches 80-90% confluency. mASCs can be used at passage 3-7 (Note 4).
- The phenotype of passage 3 mASCs from Balb/c mice, as measured by flow cytometry, is CD11b- (0.6 ± 0.2%), CD31- (0%), CD45- (0.03 ± 0.06%), sca-1+ (80.3 ± 14.5%), CD29+ (99.9 ± 0.1%) and CD44+ (99.8 ± 0.2%). The expression levels are shown as % of the population expressing the marker relative to the corresponding isotype control staining (mean ± standard deviation, n = 3). The expression range can vary between mouse strains. Differentiation of murine MSCs into osteoblasts, adipocytes and chondroblasts (Anderson et al., 2013; Peister et al., 2004) can be performed using commercial differentiation kits from e.g., Stem Cell Technologies (http://www.stemcell.com) and Lonza (http://www.lonza.com).
- When cultures reach 80-90% confluency, wash the cells twice with 10 ml PBS.
- Proliferation (Note 5)
- Preparing the mASCs (passage 3-7) for the proliferation assays.
- Harvest the cells as described above.
- Resuspend the cell pellet gently but thoroughly in 2 ml Mesencult/confluent T75 flask to obtain a cell concentration between 5 x 105 and 1 x 106 cells/ml.
- Dilute the cell suspension 1:1 in Trypan blue (20 μl cell suspension + 20 μl Trypan blue).
- Count. Mix well and add 10 μl to a Neubauer counting chamber and count the four corner squares (Note 6).
- Prepare a working solution of 10,000 cells/ml in complete Mesencult for measuring cell proliferation using the xCELLigence or 6,600 cells/ml for measuring cell proliferation using the CellTiter-Blue reagent and keep the tube on ice (Note 7).
- Harvest the cells as described above.
- xCELLigence
The xCELLigence system can be used to measure cell proliferation in real time and uses specific cell culture plates in which the well bottoms are covered with electrodes (Figure 1). Attachment of cells to the electrodes increases their impedance which is displayed as an increase in cell index. The cell index gives information about cell number, viability and morphology in each well. The xCELLigence RTCA station should be placed in a cell incubator set at 37 °C, 21% O2, 5% CO2, 90% RH.
Figure 1. Components of the xCELLigence system. The left panel shows a 16-well E-plate used for measuring proliferation of adherent cells. The middle panel shows the DP RTCA station with its three integrated stations for E-plates. The right panel gives an example of how the RTCA Software visualizes cell proliferation in real time by plotting cell index vs. time.- In a sterile hood, add 100 μl/well of complete Mesencult in a 16-well E-plate, put on the lid and place it on the xCELLigence RTCA station. Check the connections and the cell index readings to make sure that the 16-well E-plate is correctly installed. Leave the 16-well E-plate on the xCELLigence RTCA station for 30-45 min and then measure the background impedance in the absence of cells.
- Remove the E-plate from the xCELLigence RTCA station and place it in a sterile hood. Vortex the tube containing 10,000 mASCs/ml to resuspend the cells and carefully add 100 μl of cell suspension (prepared in step C1e)/well in duplicates being careful not to touch the bottom of the wells with the tips. The final volume/well should now be 200 μl.
- Leave the mASCs in the xCELLigence RTCA station in the cell incubator for 7 days, reading the impedance every hour (Note 8).
- Plot the cell index versus time in culture (see Figure 2 below).
- In a sterile hood, add 100 μl/well of complete Mesencult in a 16-well E-plate, put on the lid and place it on the xCELLigence RTCA station. Check the connections and the cell index readings to make sure that the 16-well E-plate is correctly installed. Leave the 16-well E-plate on the xCELLigence RTCA station for 30-45 min and then measure the background impedance in the absence of cells.
- CellTiter-Blue
This method is based on the conversion of resazurin to the fluorescent product resorufin by metabolically active cells. The fluorescence signal obtained is proportional to the number of viable cells in the culture.- Add 150 μl of cell suspension/well in a flat-bottomed 96-well tissue culture plate (each well will contain approximately 1,000 cells) in triplicates. Make 1 plate/time point. Add culture medium alone in triplicates to each plate to serve as background control. Place the plates in a cell incubator set at 37 °C, 5% CO2, 21% O2, 90% RH.
- Read the plates on day 1, 3 and 5-6. Add 30 μl of CellTiter-Blue reagent/well, tap the sides of the plate gently and incubate the plate for another 4 h.
- Transfer 100 μl from each well to a black 96-well plate and read fluorescence in a fluorometro (a Glomax multidetection system or equivalent) at 560Ex/590Em.
- Subtract the average background fluorescence from sample fluorescence and plot fluorescence (proliferation) versus time (see Figure 2 below).
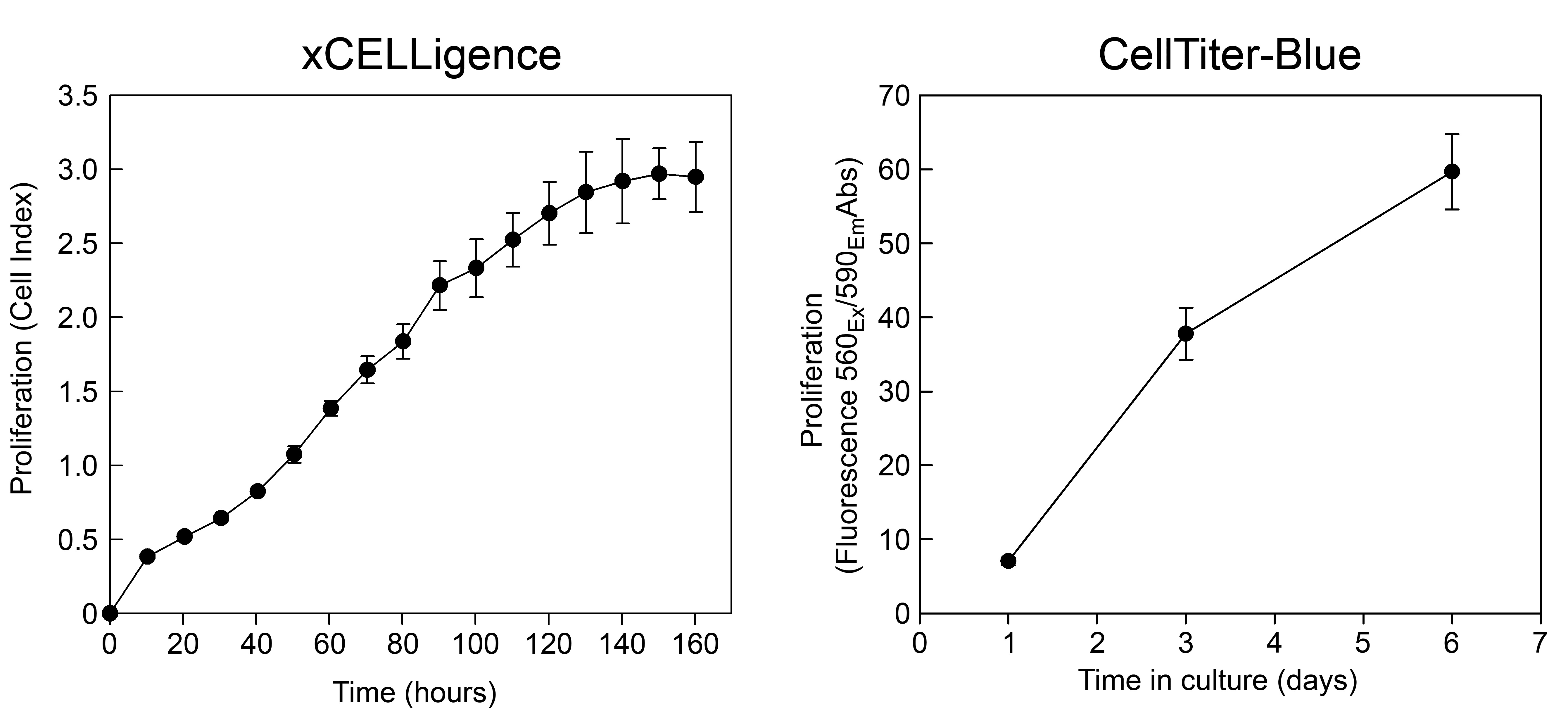
Figure 2. Representative data showing the proliferation of mASCs using the xCELLigence system (left panel) and the CellTiter-Blue reagent (right panel)
- Add 150 μl of cell suspension/well in a flat-bottomed 96-well tissue culture plate (each well will contain approximately 1,000 cells) in triplicates. Make 1 plate/time point. Add culture medium alone in triplicates to each plate to serve as background control. Place the plates in a cell incubator set at 37 °C, 5% CO2, 21% O2, 90% RH.
- Preparing the mASCs (passage 3-7) for the proliferation assays.
Notes
- Thoroughly spray the abdomen of the sacrificed mouse with 70% ethanol and secure the animal, ventral side up, on a polystyrene support using medical syringe needles. Use one set of sterile scissors/forceps to carefully make a midline incision, being careful not to damage the peritoneum. Separate the skin from the peritoneum and secure the skin flaps with syringe needles. When acquiring the inguinal fat pads care should be taken to remove the inguinal lymph nodes that are embedded into the fat at the Y-shaped junction of blood vessels. Using a new set of sterile scissors/forceps, collect both inguinal fat pads and transfer them to a 50 ml tube containing 10 ml HBSS and keep on ice. Proceed to cut open the peritoneum, taking care not to damage the intestines. Collect the gonadal fat pads attached to the testes and ovaries in male and female mice, respectively. We pool the fat from both locations when isolating mASCs. However, depot-specific differences in MSC phenotype have been reported and depending on your scientific aims you may have to isolate mASCs from one location or the other.
- The solution can be viscous and it might be necessary to use several cell strainers.
- The pellet in the following steps is loose and care should be taken when removing the supernatants.
- We seed the mASCs at a concentration of 10,000 cells/cm2 which allows us to passage the cells every 3-4 days. Under these culturing conditions we have been able to measure the proliferation of mASC from passage 3-7 using the xCELLigence system or CellTiter-Blue reagent. However, it should be noted that the size of mASCs increases with increasing passage number whereas their proliferative capacity decreases. It is therefore best to use mASC cell lines with the same passage number when comparing their proliferative capacity with the above methods.
- We provide two protocols for measuring mASC proliferation. The CellTiter-Blue reagent is easy to use and feasible in most laboratories. However, some studies have shown that resazurin can affect cell viability upon prolonged exposure and inclusion of drugs that increase mitochondrial number/activity can give false results. The xCELLigence system requires the purchase of the RTCA station/software and the cost of specialized E-plates. However, the xCELLigence system enables the label-free and non-invasive measurement of proliferation in real-time. We generally use both methods in parallel when studying mASC proliferation in vitro. To obtain more information about the CellTiter-Blue reagent and xCELLigence system, please read the manufacturer's instructions.
- We prefer to count in average 20-50 cells/corner square in order to obtain reliable cell concentrations. Calculate the cell concentration using the following formula: (total number of cells in the four corner squares/4) x 2 (dilution factor) x 10,000 = no. cells/ml.
- We found that the addition of 1,000 mASCs/well (3,125 cells/cm2) allows the monitoring of mASC proliferation for 5-7 days before the culture reaches confluence. This cell number gives reproducible growth curve which includes an initial 24-48 h lag phase followed by a proliferative phase according to Figure 1.
- We usually do not add fresh media to the E-plates during the experiment. However, it is possible to pause the experiment, remove the E-plates, add another 100 µl medium/well and restart the impedance measurements.
Recipes
- Collagenase Type I
The collagenase Type I (100 mg) is dissolved in 10 ml of HBSS to a concentration of 10 mg/ml
Aliquots of 0.5 ml are stored at -20 °C - Complete MesenCult medium
Thaw MesenCultTM stem cell stimulatory supplements (mouse), stable glutamine, penicillin/streptomycin at 4 °C and mix:
400 ml of MesenCult MSC basal medium (mouse)
100 ml MesenCult stem cell stimulatory supplements (mouse)
5 ml stable glutamine (final concentration: 2 mM)
5 ml penicillin/streptomycin (final concentration: 100 U/ml)
0.5 ml Mesenpure (optional)
Store the complete MesenCult at 4 °C and use it within one month
Acknowledgments
This work has been financed by Fondo de Investigaciones Sanitarias ISCIII (Spain) and Fondo Europeo de Desarrollo Regional (FEDER) from the European Union, through the research grants No. PI12/01390 (P.A), No. PI12/01097 and ISCIII Red de Terapia Celular (TerCel: RD12/0019/0006) (F.M), by the Consejería de Innovación Ciencia y Empresa (grants No. P09-CTS-04532 and PAIDI-Bio-326) and Consejería de Salud (grant No. PI0001/2009) from the Junta de Andalucía and FEDER/Fondo de Cohesion Europeo (FSE) de Andalucía 2007-2013 to F.M. P.A has a Miguel Servet Contract (CP09/00228), co-financed by the Fondo Europeo de Desarollo Regional (FEDER) from the European Union. F.M. is funded by the Fundación Progreso y Salud (Consejería de Salud - Junta de Andalucía).
References
- Anderson, P., Carrillo-Gálvez, A. B., Garcia-Perez, A., Cobo, M. and Martín, F. (2013). CD105 (endoglin)-negative murine mesenchymal stromal cells define a new multipotent subpopulation with distinct differentiation and immunomodulatory capacities. PLoS One 8(10): e76979.
- Carrillo-Gálvez, A. B., Cobo, M., Cuevas-Ocana, S., Gutierrez-Guerrero, A., Sanchez-Gilabert, A., Bongarzone, P., Garcia-Perez, A., Munoz, P., Benabdellah, K., Toscano, M. G., Martín, F. and Anderson, P. (2015). Mesenchymal stromal cells express GARP/LRRC32 on their surface: effects on their biology and immunomodulatory capacity. Stem Cells 33(1): 183-195.
- Peister, A., Mellad, J. A., Larson, B. L., Hall, B. M., Gibson, L. F. and Prockop, D. J. (2004). Adult stem cells from bone marrow (MSCs) isolated from different strains of inbred mice vary in surface epitopes, rates of proliferation, and differentiation potential. Blood 103(5): 1662-1668.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Anderson, P., Carrillo-Gálvez, A. B. and Martín, F. (2015). Isolation of Murine Adipose Tissue-derived Mesenchymal Stromal Cells (mASCs) and the Analysis of Their Proliferation in vitro. Bio-protocol 5(21): e1642. DOI: 10.21769/BioProtoc.1642.
Category
Stem Cell > Adult stem cell > Hematopoietic stem cell
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










