- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Pot Level Drought Stress Tolerance Assay in Tobacco through Plant Phenotyping and Antioxidant Assay
Published: Vol 5, Iss 19, Oct 5, 2015 DOI: 10.21769/BioProtoc.1605 Views: 13201
Reviewed by: Fanglian HeAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
A Simple Sonication Method to Isolate the Chloroplast Lumen in Arabidopsis thaliana
Jingfang Hao and Alizée Malnoë
Aug 5, 2023 2309 Views

A Plate Growth Assay to Quantify Embryonic Root Development of Zea mays
Jason T. Roberts [...] David M. Braun
Oct 20, 2023 2280 Views
Detection and Quantification of Programmed Cell Death in Chlamydomonas reinhardtii: The Example of S-Nitrosoglutathione
Lou Lambert and Antoine Danon
Aug 5, 2024 1607 Views
Abstract
Drought is an important abiotic factor which has a huge detrimental impact on crop productivity. Study of plant responses towards drought stress and investigating the mechanism of drought tolerance is crucial for achieving the target of developing drought-tolerant plants. Phenotyping is a cost effective approach which can be adopted to evaluate the severity of drought stress in a plant. Next to phenotyping parameters, biochemical parameters such as the study of antioxidant enzyme activity play significant roles in assessing the extent of drought stress caused injury in a plant. Among the antioxidant enzymes, ascorbate peroxidase is an enzyme which plays a crucial role in drought tolerance in plants. It has been well established that the activity of this enzyme increases under drought stress. Here, we present a simple and reproducible protocol to investigate the response of tobacco plants towards drought stress through measurement of phenotypic parameters and antioxidant enzyme activity. Though, these experiments have been conducted with tobacco plants, this protocol could be adopted for other crop species.
Keywords: DroughtMaterials and Reagents
- Pipette (BrandTech Scientific, Transferpette, catalog numbers: 704770 , 704778 , 704780 )
- Centrifuge tube (1.5 ml) (Corning, Axygen®, catalog number: MCT-150-C-S )
- Tobacco seed samples under study: Nicotiana tabacum L. cv. Petit Havana, wild type and transgenic lines
Note: The complete open reading frame of histone-gene binding protein of rice (OsHBP1b) was cloned at BglII and SpeI sites in the plant expression vector (pCAMBIA1304) and used for transformation of Agrobacterium strain (LBA 4404). Fifteen days old tobacco seedlings were used for transformation through tissue culture method. The putative transgenic T0 plants were transferred to the greenhouse for the purpose of seeds multiplication. The seeds of T2 transgenic lines were used for experimental purpose. - Distilled H2O (IndiaMART InterMESH Ltd, Mars Scientific Instruments Co., catalog number: BASIC/pH & XL )
- Agro peat (Agro peat super) (AswiniAgrotech)
- Vermiculite (Manidharma Biotech Private Limited)
- Sucrose (Sigma-Aldrich, catalog number: S0389 )
- Stress reagent (Polyethylene glycol 6000 or PEG 6000) (Sigma-Aldrich, catalog number: 81260 )
- Ascorbate (Sigma-Aldrich, catalog number: A4034 )
- Liquid Nitrogen (Advanced Instrumentation Research Facility, Jawaharlal Nehru University, New Delhi)
- Hydrogen peroxide (H2O2) (Sigma-Aldrich, catalog number: 216763 )
- Bovine serum albumin (Sigma-Aldrich, catalog number: A2153 )
- Murashige & Skoog (MS) with vitamins (Caisson Laboratories, catalog number: MSP09-1lt )
- K2HPO4 (Sigma-Aldrich, catalog number: 1551128 )
- KH2PO4 (Sigma-Aldrich, catalog number: 1551139 )
- Coomassie Brilliant Blue G-250-100 mg (Sigma-Aldrich, catalog number: 27815 )
- Methanol (100%) (Sigma-Aldrich, catalog number: 34860 )
- Phosphoric acid (85%) (Sigma-Aldrich, catalog number: W290017 )
- Phosphate buffer (see Recipes)
- Bradford reagent (see Recipes)
- MS media (see Recipes)
Equipment
- Plastic pots (12 cm diameter and 13 cm depth) (Garden Aids, India)
- Forceps (ACE Surgical Co, catalog number: 10)
- Growth chamber (Daihan LabTech India Pvt. Ltd., model: LGC-S201 )
- Oven (Hicon India)
- pH meter (Control Dynamic Instrumentation Pvt. Ltd., model: APX175E )
Note: Currently, it is “CD Hightech Pvt. Ltd., model: APX175E ”. - Plastic ice tray
- Liquid nitrogen container (Kailash Gases, Cryocan, model: BA-11 )
- Motor and pestle (local supplier, New Delhi)
- Cuvette (both plastic and quartz) (Sigma-Aldrich, catalog number: Z276677 )
- Spectrophotometer (Cary 300 UV-Vis)
- Image capturing device (SONY CORPORATION OF AMERICA, model: DSC-HX300 )
- Weighing machine (Sartorius, model: BSA224S-CW )
- Scale (30 cm plastic scale)
Software
- GraphPad InStat3 software
Procedure
- Seed germination
- Imbibe wild type (WT) and transgenic tobacco seeds in distilled water for overnight.
- Allow twenty to twenty five seeds of tobacco to germinate on soil (equal quantity of agro peat and vermiculite should be mixed to prepare the soil) filled pots inside a growth chamber at 28 ± 1 °C with PPFD of 100 μmol/m2/s, a relative humidity of 75-80%, and a photoperiod of 12/12 h light/dark. Supplement 50 ml of MS media every day in the soil-filed pots to provide nutrition to the plants.
- Transfer two-week-old seedlings individually to separate soil-filled pots.
- Allow the WT and transgenic plants to grow up to 25 days for drought tolerance assay at pot level (Lakra et al., 2015).
- Imbibe wild type (WT) and transgenic tobacco seeds in distilled water for overnight.
- Drought treatment and recovery
- For drought treatment, prepare 5% PEG solution in MS media and supplement 100 ml of this solution to the pots individually, on a regular basis (once a day), for 5- or 10- days (Kumar et al., 2012; Lakra et al., 2015).
Note: Polyethylene glycol or PEG is a flexible, water-soluble polymer, it can be used to create high osmotic pressures. These properties make PEG one of the most useful molecules for applying osmotic pressure in plants to induce water deficit stress. - After 5 or 10 days of drought treatment, allow the plants to recover by supplementing 50 ml of MS media for 7 days and subsequently, carry out drought tolerance assay.
- For drought treatment, prepare 5% PEG solution in MS media and supplement 100 ml of this solution to the pots individually, on a regular basis (once a day), for 5- or 10- days (Kumar et al., 2012; Lakra et al., 2015).
- Assessment of drought stress response after recovery
Evaluation of drought stress in WT and transgenic tobacco can be done by analysis of phenotypic and biochemical parameters. - Phenotypic assessment
- As phenotypic parameters, number of yellow or green leaf, root length, shoot length, shoot fresh weight, root fresh weight, shoot dry weight and root dry weight of WT and transgenic tobacco plants can be considered (Lakra et al., 2015). As representative data, phenotype of WT and transgenic tobacco plants after drought stress has been shown here (Figure 1A).
- Measure the shoot and root lengths with the help of a scale.
- For shoot and root dry weight, cut shoot and root samples from the plant and dry them in an oven for 72 h. Subsequently, measure the dry weight of the samples through a weighing machine.
Note: Use at least three individual lines from each type of plants for each experimental analysis. Analyse data for variance (ANOVA) by GraphPad InStat3 software.
- As phenotypic parameters, number of yellow or green leaf, root length, shoot length, shoot fresh weight, root fresh weight, shoot dry weight and root dry weight of WT and transgenic tobacco plants can be considered (Lakra et al., 2015). As representative data, phenotype of WT and transgenic tobacco plants after drought stress has been shown here (Figure 1A).
- Biochemical assessment
- As biochemical parameter, ascorbate peroxidase (APX) activity can be measured through spectrophotometric method. As representative data, comparison of APX activity in WT and transgenic tobacco before and after drought stress has been shown (Figure 1B-C).
Note: Under control (before drought stress) conditions no significant difference in APX activity was observed between WT and transgenic plants. - For this purpose, weigh 100 mg of WT and transgenic tobacco leaf tissue and grind these separately using chilled motors and pestles (Figure 2A).
Note: One may take young or old leaves, but, consistency should be maintained to take same aged leaves from each sample type. - Supplement liquid nitrogen for fine crushing of tissue.
- Add 1 ml of potassium phosphate buffer (prepare fresh and keep at room temperature) immediately and then add 0.2 mM of PMSF to avoid protein degradation.
- Then transfer the homogenate (Figure 2B) to microcentrifuge tubes (Figure 2C) and centrifuge at 10,000 x g for 15 min. Maintain the temperature of the centrifuge machine at 4 °C.
- After centrifugation, the pellet and the clear supernatant will be visible (Figure 2D). Subsequently, transfer the supernatant to fresh microcentrifuge tubes (Figure 2E) which will serve as crude protein/enzyme extract and will be used for APX activity assay.
- Measure the concentration of crude protein by using Bradford method (Bradford, 1976). Use BSA standard curve for protein quantification.
Note: Previously prepared standard curve can be used here. - For APX activity assay, use double beam spectrophotometer and quartz cuvette (Figure 3A and B).
- Prepare 1 ml of reaction mixture (sample) which will contain 50 mM K-PO4 buffer (pH 7.0), 10 µg crude enzyme extract and 0.35 mM ascorbate. Initiate the reaction by adding 5 µl of 10 mM H2O2.
Note: H2O2 and ascorbate should be prepared in 50 mM K-PO4 buffer to maintain the buffer molarity.
Reaction formula: The reaction initiates immediately upon addition of H2O2 where H2O2-dependent oxidation of ascorbate starts. The gradual decrease in absorbance can be visualized on the computer screen after initiation of the reaction. - Use the reaction mixture without H2O2 as reference.
- Follow H2O2-dependent oxidation of ascorbate spectrophotometrically by recording the decrease in absorbance at 290 nm (€ = 2.8 mM-1 cm-1).
Note: The reaction time is 120 sec and the reaction temperature is 20 °C. - Recollect that Beer's law is expressed as A = €CL where, ‘A’ is absorbance, ‘€’ is absorption coefficient (a constant that reflects the efficiency or the extent of absorption at selected wavelengths), ‘C’ is concentration of the substance and ‘L’ is the path length or the thickness of the cuvette. To find the concentration for a solution, you will first need to find the slope of the best-fit line. From the slope of the best-fit line together with the absorbance, you can now calculate the concentration for that solution (i.e., Concentration = Absorbance/Slope)
- Here, slope value of absorbance in 290 nm (Δ OD s-1) from first 0.6 min is considered for rate calculation (Figure 4).
Note: The slope of the best-fit line in this case is actually the product of the molar absorptivity constant and the path length (1.00 cm).Cary 300 UV-Vis instrument has the option to show the slope value of the graph. - The activity of APX can be represented by the unit ‘µmol/min/mg protein’ or ‘µmol/min/g protein’.

Figure 1. Plant Morphology and APX activity of WT and transgenic tobacco leaves following drought stress treatment and subsequent recovery. (A) Shows yellow and green leaves in WT and transgenic tobacco after drought stress. (B) APX activity in WT and transgenic tobacco leaves before drought stress treatment. (C) APX activity in WT and transgenic tobacco leaves after drought stress treatment.
Figure 2. Protein extraction from tobacco leaves for measuring APX activity. (A) Set up for tissue crushing. Red, yellow, black and blue arrows show liquid N2 container, motor, pestle and ice container respectively. (B) Tissue homogenate after crushing. (C) Tissue homogenate after transferring to microcentrifuge tube. (D) Supernatant (Red arrow) and Pellet (Blue arrow) after centrifugation. (E) Supernatant containing the crude protein/enzyme after transferring to a fresh microcentrifuge tube.
Figure 3. View of double beam spectrophotometer and cuvette. (A) Double beam UV-Vis spectrophotometer. Red arrow shows cuvette holder for reference and blue arrow shows cuvette holder for sample. (B) Quartz cuvettes used to read reference and sample absorbance.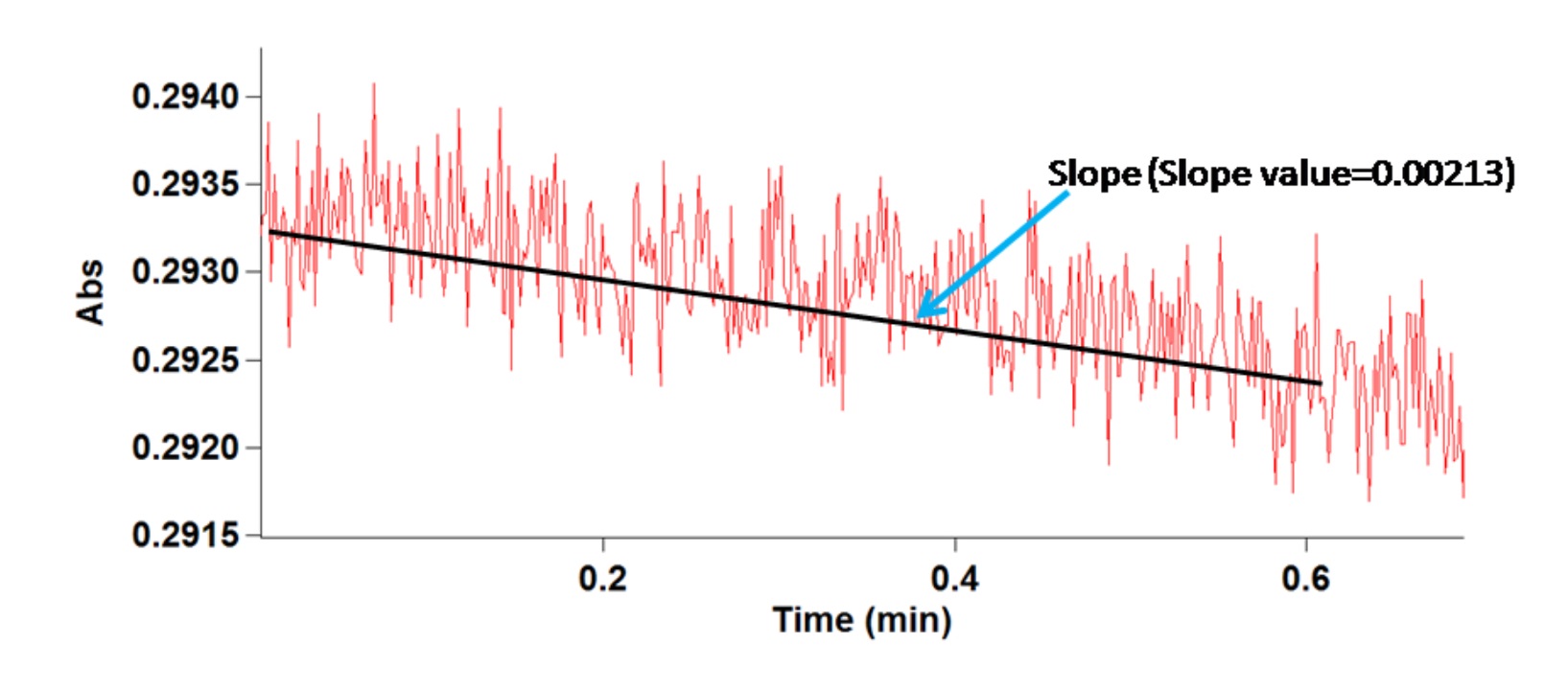
Figure 4. APX activity graph showing decline in enzyme activity due to depletion of H2O2 in unit time. Black line indicates the slope and blue arrow indicates the slope value.
- As biochemical parameter, ascorbate peroxidase (APX) activity can be measured through spectrophotometric method. As representative data, comparison of APX activity in WT and transgenic tobacco before and after drought stress has been shown (Figure 1B-C).
Notes
Precautions to be followed:
- Proper maintenance of nutrient medium is required for plant growth in pots.
- Light, temperature and humidity should be maintained properly.
- If algal growth is observed on the soil surface, it can be removed by removing the algal layer along with the thin upper layer of soil with the help of a spatula. If it appears that soil surface is reducing after removing the algal layer along with a layer of soil, additional soil can be added to the pot carefully without harming the plant.
- Cuvette should be cleaned properly before use.
Recipes
- Phosphate buffer
Make 10 ml of 50 mM K2HPO4
Make 10 ml of 50 mM KH2PO4
Place 50mM of K2HPO4 under pH meter
Add required amount of KH2PO4 to get 7.0 - Bradford reagent
Take Coomassie Brilliant Blue G-250 100 mg
Add 47 ml methanol (100%) to it
Add 100 ml phosphoric acid (85%) to it
Add required distilled water to get final volume of 1 L - MS media
Weigh 4.4 mg of readily available MS powder
Dissolve 4.4 mg of MS powder in 1 L of distilled water
Add 30 g of sucrose to 1 L of MS solution
Adjust pH 5.8
Acknowledgments
Authors are thankful to Department of Science and Technology (Indo-Sri Lanka Program) and University Grants Commission, Govt. of India for the financial support. Authors acknowledge previous work published by Lakra et al. (2015), from which this protocol is adopted.
References
- Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254.
- Kumar, G., Kushwaha, H. R., Panjabi-Sabharwal, V., Kumari, S., Joshi, R., Karan, R., Mittal, S., Pareek, S. L. and Pareek, A. (2012). Clustered metallothionein genes are co-regulated in rice and ectopic expression of OsMT1e-P confers multiple abiotic stress tolerance in tobacco via ROS scavenging. BMC Plant Biol 12: 107.
- Lakra, N., Nutan, K. K., Das, P., Anwar, K., Singla-Pareek, S. L. and Pareek, A. (2015). A nuclear-localized histone-gene binding protein from rice (OsHBP1b) functions in salinity and drought stress tolerance by maintaining chlorophyll content and improving the antioxidant machinery. J Plant Physiol 176: 36-46.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Das, P., Lakra, N., Nutan, K. K., Singla-Pareek, S. L. and Pareek, A. (2015). Pot Level Drought Stress Tolerance Assay in Tobacco through Plant Phenotyping and Antioxidant Assay. Bio-protocol 5(19): e1605. DOI: 10.21769/BioProtoc.1605.
Category
Plant Science > Plant physiology > Abiotic stress
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









