- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Extraction of Chloroplast Proteins from Transiently Transformed Nicotiana benthamiana Leaves
Published: Vol 4, Iss 18, Sep 20, 2014 DOI: 10.21769/BioProtoc.1238 Views: 20677
Reviewed by: Sam-Geun KongTie Liu

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Streamlining Protein Fractional Synthesis Rates Using SP3 Beads and Stable Isotope Mass Spectrometry: A Case Study on the Plant Ribosome
Dione Gentry-Torfer [...] Federico Martinez-Seidel
May 5, 2024 2900 Views

An Activity-Based Proteomics with Two-Dimensional Polyacrylamide Gel Electrophoresis (2D-PAGE) for Identifying Target Proteases in Arabidopsis Apoplastic Fluid
Sayaka Matsui and Yoshikatsu Matsubayashi
Mar 5, 2025 2032 Views
Advancing 2-DE Techniques: High-Efficiency Protein Extraction From Lupine Roots
Sebastian Burchardt [...] Emilia Wilmowicz
Oct 5, 2025 1801 Views
Abstract
This rapid protocol allows the extraction of chloroplast enriched proteins from Nicotiana benthamiana (N. benthamiana) leaves that were transiently transformed to express an epitope tagged protein of interest. Thus, it can serve as a tool to study the chloroplastidic localization of the protein of interest when it is combined with western-blot analysis.
Agrobacterium-mediated transformation (Agroinfiltration, Romeis et al., 2001) is used to transiently express a protein carrying an epitope tag in tobacco leaves. Here, co-infiltration with an Agrobacterium strain harboring 19 K from soil-borne wheat mosaic virus suppresses posttranscriptional gene silencing and therefore increases transformation efficiency (Te et al., 2005).
The chloroplast isolation of the transformed leaves is based with modifications on Romeis et al. (2001), and includes mechanical breakage of cell wall and membranes, the removal of unbroken tissue by filtration and the separation of intact chloroplasts by centrifugation through a Percoll layer.
Materials and Reagents
- Nicotiana benthamiana plants
- Agrobacterium tumefaciens (A. tumefaciens) strain GV3101 carrying a recombinant binary plasmid with the gene of interest fused to an epitope tag
- Agrobacterium tumefaciens strain GV3101 harboring K19 (Te et al., 2005)
- Tryptone (AppliChem GmbH, catalog number: 403682 .1210)
- Yeast extract (AppliChem GmbH, catalog number: A1552 , 0500)
- Sucrose (AppliChem GmbH, catalog number: A2211 , 0500)
- MgSO4 (Merck KGaA, catalog number: 105886 )
- Appropriate antibiotic
- Agar (Carl Roth, catalog number: 2266.2 )
- Acetosyringone (Sigma-Aldrich, catalog number: D134406 )
- MES buffer (Carl Roth, catalog number: 4256.3 )
- Liquid nitrogen
- Percoll (GE healthcare, catalog number: 17-0891-01 )
- EDTA (Carl Roth, catalog number: 8040.1 )
- DTT (AppliChem GmbH, catalog number: A2948 )
- MgCl2 (Merck KGaA, catalog number: 105833 )
- Glycerol (AppliChem GmbH, catalog number A3739 ,0500)
- Hepes (Carl Roth, catalog number: 9105 )
- Sorbitol (Merck KGaA, catalog number: 56755 )
- Serum bovine albumin (Carl Roth, catalog number 8076.2 )
- Protease inhibitor cocktail Complete Mini EGTA-free (Roche Diagnostics, catalog number: 11 836 153 001 )
- YEP medium (see Recipes)
- Agromix buffer (see Recipes)
- Isolation buffer (see Recipes)
- Protein extraction buffer (see Recipes)
Equipment
- 1 ml syringe (Terumo Medical Corporation, catalog number: BS-01H )
- Centrifuge cooled at 4 °C
- 47 μm nylon mesh (Carl Roth, catalog number: XA63.1 )
- 2 ml glass homogenizer with round bottom shape (A. Hartenstein, catalog number: HOG2 )
- Light microscope with phase contrast condenser and phase contrast compatible objective lens, such as Objective LD A-Plan 40x/0.5 Ph2 (ZEISS)
- Incubator shaker
- Rotary shaker
- Photospectrometer for OD600
- 1.5 ml reaction tubes (SARSTEDT AG, catalog number 72.706 )
- 50 ml conical centrifuge tubes (SARSTEDT AG, catalog number 62.548.004 )
Procedure
- Transient transformation of N. benthamiana
- Agrobacterium tumefaciens GV3101 strain harboring 19 K and Agrobacterium tumefaciens GV3101 strain carrying the recombinant binary plasmid for the transient expression of the gene of interest (with epitope tag) were streaked from glycerol stocks on YEP Agar plates (containing the appropriate antibiotics), respectively. Plates were incubated at 28 °C for 48 h to obtain single colonies.
- Single colonies from plates not older than 1 week were inoculated into 15 ml YEP medium (containing the appropriate antibiotics) in 50 ml tubes and incubated at 28 °C overnight at 180 rpm in an shaker.
- The next day the overnight cultures (OD600 between 1.0 and 1.5) were centrifuged at 2,500 x g for 10 min at room temperature. The supernatant was discarded.
- Each pellet was re-suspended in 5 ml of agromix buffer.
- The Agromix was incubated for 2 h in the dark at 120 rpm on a rotary shaker at room temperature.
- Both bacterial suspensions (19 K, gene of interest) were combined such that the final suspension with an OD600 between 0.5 and 1.0 consisted of 20% agrobacteria with 19 K and of 80% agrobacteria carrying the recombinant binary plasmid for the transient expression of the gene of interest in total.
Note: This ratio for 19K in the infiltration mixture has been proven to be effective to enhance the transient expression of several proteins due to the suppression of gene silencing by 19K. However, the optimum ratio might be different for other proteins. An OD600 higher than 1.0 in the infiltration mixture could lead to effects of toxicity in tobacco. - This mix was infiltrated by pressing the tip of a syringe without needle onto the lower leaf surface of 3-4 weeks old, well watered, Nicotiana benthamiana. The third, fourth and fifth youngest leaf were used for agroinfiltration (Figure 1A). Every leaf half was once injected with 100-150 µl of the mix (see Video 1). The infiltrated area on the leaves were marked using a marker pen (Figure 1B).
Note: It is important to water the plants in the morning of the day on which the agroinfiltration takes place. Otherwise, it is difficult to infiltrate the leaf tissue successfully without damaging too much the tissue.
Video 1. Agroinfiltration procedure - Agrobacterium tumefaciens GV3101 strain harboring 19 K and Agrobacterium tumefaciens GV3101 strain carrying the recombinant binary plasmid for the transient expression of the gene of interest (with epitope tag) were streaked from glycerol stocks on YEP Agar plates (containing the appropriate antibiotics), respectively. Plates were incubated at 28 °C for 48 h to obtain single colonies.
- Chloroplast isolation of transiently transformed N. benthamiana
Preliminary remark: For optimal yield of intact chloroplasts the plant material should be harvested from tobacco plants at the end of the dark cycle and kept in the dark to avoid excess levels of starch accumulation that can rupture the chloroplast envelope during centrifugation. In addition, all protocol steps are carried out at 4 °C.- 100 mg of plant material of transiently transformed N. benthamiana leaves, 2-3 days after agro-infiltration, were homogenized in 400 μl of ice-cold isolation buffer using a precooled glass homogenizer.
- The homogenate was filtered through a 47 μm nylon mesh and kept on ice and in the dark.
- Preparation of 40% Percoll layer: 132 µl of Percoll were mixed thoroughly with 198 µl of isolation buffer and the layer of 40% Percoll was deposited in the bottom of a 1.5 ml reaction tube.
- 400 µl of the filtered chloroplast containing homogenate was placed carefully on top of the 40% Percoll layer and centrifuged at 7,000 x g at 4 °C for 1 min. The intact chloroplasts will sediment as a green pellet, whereas the broken chloroplasts remain on the top of the Percoll layer (Figure 2).
Notes:- Alternatively, the centrifugation of intact chloroplasts can also be performed more gently at 1,700 x g for 6 min at 4 °C.
- The supernatant was discarded and the chloroplasts were re-suspended gently in a small volume (20-30 µl) of isolation buffer. The chloroplast enriched suspension was kept on ice.
- Alternatively, the centrifugation of intact chloroplasts can also be performed more gently at 1,700 x g for 6 min at 4 °C.
- The quality of the pelleted chloroplasts was confirmed by applying phase contrast microscopy. Intact chloroplasts appear opaque with a halo surrounding them (Figure 3A). Broken chloroplasts exhibit a dark green, granular, and non-refractive appearance (Figure 3B).
- Subsequently, the chloroplasts were centrifuged at 1,000 x g at 4 °C for 5 min to pellet the chloroplasts and the supernatant was removed and discarded.
- Chloroplasts were shock frozen in liquid nitrogen.
- The freeze-fractured chloroplasts were re-suspended in 50µl of ice-cold protein extraction buffer and centrifuged at 15,000 x g at 4 °C for 10 min.
- The supernatant (with soluble chloroplastidic proteins) was transferred to a new tube.
- The pellet, enriched with non-soluble chloroplastidic proteins, was re-suspended in 50 µl of ice-cold protein extraction buffer.
- Samples could either be stored at -70 °C or directly be used for western blot analyis to compare the protein expression in the chloroplast fraction versus total crude extract from transiently transformed tobacco leaves.
Note: The protein yield for isolation of chloroplastidic proteins from 100 mg leaf material is in the range of 10 and 20 µg in total. 5-10 µg can be used for the western blot analysis.
- 100 mg of plant material of transiently transformed N. benthamiana leaves, 2-3 days after agro-infiltration, were homogenized in 400 μl of ice-cold isolation buffer using a precooled glass homogenizer.
Representative data

Figure 1. Agroinfiltration procedure. A. The leaves indicated with number 3, 4 and 5 are best used for agroinfiltration. B. The infiltrated areas are marked with a marker pen.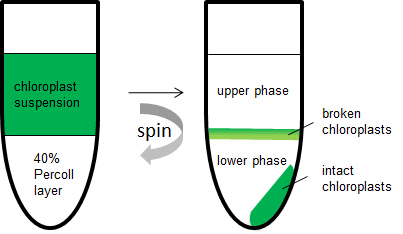
Figure 2. Separation of intact from broken chloroplasts after centrifugation through a 40% Percoll layerl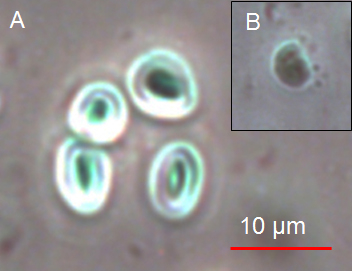
Figure 3. A Intact chloroplasts and B broken chloroplast visualized by phase contrast microscopy
Recipes
- YEP medium
0.5 % tryptone (m/v)
0.5% yeast extract (m/v)
0.5% sucrose (m/v)
50 mM MgSO4
For plates: 1.5% agar (m/v) was added
The YEP medium was autoclaved - Agromix buffer (has to be prepared fresh)
10 mM MES (adjusted to pH=5.8 with 1 M KOH)
10 mM MgCl2
150 μM acetosyringone
In purified (deionized and autoclaved) water - Isolation buffer (has to be prepared fresh)
0.33 M Sorbitol
50 mM Hepes (adjusted with 1 M KOH to pH=7.0)
0.1% (m/v) serum bovine albumin
2 mM EDTA
1 mM MgCl2
In purified (deionized and autoclaved) water - Protein extraction buffer (has to be prepared fresh)
10% (m/v) glycerol (from autoclaved stock solution)
5 mM EDTA (from autoclaved stock solution)
10 mM DTT
100 mM HEPES (adjusted with 1 M KOH to pH=7.2, sterilfiltrated)
Protease inhibitor cocktail
In purified (deionized and autoclaved) water
Acknowledgments
This protocol was adapted from previous work (Romeis et al., 2001; Te et al., 2005; Pineda et al., 2010). I would like to acknowledge R. Deeken (Department of Molecular Plant Physiology and Biophysics, University of Wuerzburg, Germany) and the DFG Research Training Group 1342 (University of Wuerzburg, Germany) for funding. In addition, I would like to than C.W. Lee for help and advice.
References
- Pineda, M., Sajnani, C. and Baron, M. (2010). Changes induced by the Pepper mild mottle tobamovirus on the chloroplast proteome of Nicotiana benthamiana. Photosynth Res 103(1): 31-45.
- Romeis, T., Ludwig, A. A., Martin, R. and Jones, J. D. (2001). Calcium-dependent protein kinases play an essential role in a plant defence response. EMBO J 20(20): 5556-5567.
- Te, J., Melcher, U., Howard, A. and Verchot-Lubicz, J. (2005). Soilborne wheat mosaic virus (SBWMV) 19K protein belongs to a class of cysteine rich proteins that suppress RNA silencing. Virol J 2: 18.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Klinkenberg, J. (2014). Extraction of Chloroplast Proteins from Transiently Transformed Nicotiana benthamiana Leaves. Bio-protocol 4(18): e1238. DOI: 10.21769/BioProtoc.1238.
- Klinkenberg, J., Faist, H., Saupe, S., Lambertz, S., Krischke, M., Stingl, N., Fekete, A., Mueller, M. J., Feussner, I., Hedrich, R. and Deeken, R. (2014). Two fatty acid desaturases, STEAROYL-ACYL CARRIER PROTEIN Delta9-DESATURASE6 and FATTY ACID DESATURASE3, are involved in drought and hypoxia stress signaling in Arabidopsis crown galls. Plant Physiol 164(2): 570-583.
Category
Plant Science > Plant cell biology > Organelle isolation
Plant Science > Plant biochemistry > Protein > Isolation and purification
Cell Biology > Organelle isolation > Chloroplast
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










