- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Grafting Arabidopsis
Published: Vol 4, Iss 13, Jul 5, 2014 DOI: 10.21769/BioProtoc.1164 Views: 17549
Reviewed by: Ru Zhang

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
A Novel Imaging Protocol for Investigating Arabidopsis thaliana Siliques and Seeds Using X-rays
Brylie A. Ritchie [...] Ansul Lokdarshi
Oct 5, 2023 2217 Views

Enzymatic Starch Quantification in Developing Flower Primordia of Sweet Cherry
Nestor Santolaria [...] Afif Hedhly
Apr 5, 2025 1932 Views

Direct Plant Regeneration From Immature Male Inflorescence of Banana (Musa spp.)
Pradeep Chand Deo
Oct 20, 2025 1478 Views
Abstract
In Arabidopsis thaliana, hypocotyl micrografting has been used to investigate transport of flowering signals, mobile silencing signals and other peptides, proteins and secondary compounds. The effects of transported signals on target tissues require that a good vascular connection is re-established across the graft junction between the cut hypocotyls (stumps) of the root (rootstock) and shoot (scion) tissues. We outline here a method that requires only that the cut stumps be placed in close proximity, so that they touch, followed by 3-5 days of undisturbed recovery time during which the grafts are allowed to dry out somewhat. This method is quick, easy to monitor and has up to 90% success rate.
Materials and Reagents
- Young Arabidopsis seedlings (grown for 3-6 days on ½ MS containing agar plates)
- Sterile distilled water
- 96 % ethanol
Equipment
- Sterile 9 cm petri dishes
- Sterile standard Whatman No. 1 filter paper circles Grade 1:11 µm with diameter about 0.5 cm smaller than the sterile petri dish (autoclaved wrapped in foil beforehand) (8.5 cm diameter) (Whatman)
- Sterile cellulose nitrate filters (Whatman, catalog number: NC 45 ST)
- Sterile forceps
- Sterile 1 ml pipette tips
- 1 ml pipettor
- Micropore tape (3 M, MicroporeTM)
- Sapphire knife holder (World Precision Instruments, catalog number: 500317 ) or diamond knife plus blade (Electron Microscopy Sciences, Diamond Dissecting Knife, Type M-M, catalog number: 72025 )
- Sapphire blade (World Precision Instruments, catalog number: 504072 ) or diamond blade (Electron Microscopy Sciences, Blade Resharpening, Type M-M, catalog number: 72025-R ; Electron Microscopy Sciences, Replacement Blade, Type M-M, catalog number: 72025-L )
- Flow cabinet mounted dissecting microscope with a minimum of 5x magnification
Note: Any brand we have tried worked.
- Parafilm
- Hot glass bead sterilizer (any brand) unless using 96 % ethanol for sterilization
Procedure
- Preparation for grafting
Require:- Sterile 9 cm petri dishes.
- Sterile regular filter paper with diameter about 0.5 cm smaller than the sterile petri dish (autoclaved wrapped in foil beforehand).
- Sterile cellulose nitrate filters (not to be autoclaved, are wrapped in sterile packing).
- Sterile forceps.
- Sterile distilled water.
- Sterile 1 ml pipette tips.
- 1 ml pipettor.
- Micropore tape.
- Ethanol for flaming, unless using a hot glass bead steriliser for forceps.
- Sapphire or diamond knife.
- Dissecting microscope with attached lamp for use in laminar flow cabinet.
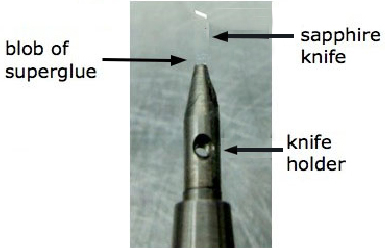
Figure 1. Gluing the sapphire knife
Note: The sapphire blade must be superglued into its holder before use. If the ethanol dip used for sterilisation loosens it, it falls out. This is not needed if using a diamond blade.
- Sterile 9 cm petri dishes.
- Grafting Arabidopsis seedlings
- Set up the materials in the laminar flow cabinet as below: Rinse hands in 70% ethanol, wipe laminar flow surface and dissecting microscope with 96% ethanol.

Figure 2. Setup of equipment in the laminar flow cabinet
- Add two sterile filter papers to a sterile petri dish. Wet the filter papers with 2 ml sterile water. Place two cellulose nitrate filters side-by-side on top of these and add an addtional 1 ml sterile water. The purpose of the filters is to provide a firm, yet water penetrable layer that allows both the grafting procedure and subsequent growth of the seedlings to occur. The filters must not be too wet, but must not be so dry that the plants dry out during and after grafting.
- Use young seedlings that have been grown vertically on ½ MS containing agar plates for 3-6 days, no more than 2-3 cm in total length, with the cotyledons sufficiently expanded to be almost horizontal-see Figure 4. The first two true leaves should not be visible or only barely visible.

Figure 3. Putting seedlings onto the nitrocellulose plates
- Dip a pair of forceps in ethanol and flame, or, place forceps in hot glass bead steriliser (250 °C) for approx. 60 sec. After either procedure, press tips of forceps into the agar to cool so seedlings are not damaged by heat. Under the dissecting microscope, select 6-8 very straight seedlings for use as the scions, and place them on the top half of the wet cellulose nitrate filters. Replace the lid on the agar plate containing seedlings, and store vertically to one side of the cabinet.
- Now take a second plate of seedlings and use the dissecting microscope to select 6-8 very straight seedlings for use as the rootstocks of the grafts, and place them on the bottom half of the wet cellulose nitrate filter. Replace the lid on the agar plate containing seedlings, and store vertically to one side of the cabinet.
- Take the sapphire/diamond knife, dip into 96% ethanol and hold upright in the cabinet until dry - this knife must not be flamed or heated as it destroys the blade.
- Orient the seedlings under the microscope so they are ready to cut. They should all be oriented so that their cotyledons are lying horizontally on the nitrocellulose membrane as in the left-most image in Figure 4. Use the sapphire/diamond knife to remove the cotyledons from all seedlings, and move these to one side of the filter paper, or remove with forceps.
- Now use the knife to cut the scion seedlings cleanly, and exactly horizontally, across the hypocotyl just below the cotyledon stumps-as in the image second from right in Figure 4.

Figure 4. Aligning seedlings for cutting
- Make sure the rootstock seedlings were also lying with their cotyledons flat and horizontal. Now cut the rootstock seedlings cleanly, and exactly horizontally, across the hypocotyl just below the cotyledon stumps.
- Very important: With the forceps, very gently grab a rootstock piece by the longest root hairs on the oldest part of the root just below the hypocotyl and place it so the cut stump touches the cut stump of a scion cutting. Repeat for all rootstocks. It is very important for the graft efficiency to minimize moving or touching the scion after it has been cut! The most successful grafts are those in which the two phloem strands match up from rootstock to scion, and this is ensured if the cotyledons are lying at the same angle before cutting. Successful grafts also have rootstock and scion well-matched for size, with the “stumps” cut very cleanly and squarely, so that the cut ends of rootstock and scion can be pressed very closely together, as in the righthand image in Figure 5. Separation between rootstock and scion, as in the central image of Figure 5, generally results in failure to connect.
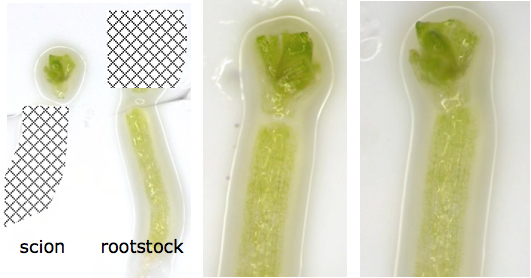
Figure 5. Scion/rootstock connection after cutting
- With care, the reciprocal cuttings can be placed, stump to stump, to form the reciprocal grafts.
- The cellulose nitrate filter paper must be kept moist, but not running wet, at all times.
After all grafts are complete, the paper must be just damp, not wet, or the grafts will be less successful. When transferred to the growth room, there must not be too much water in the base of the vertical petri dish. It should look as in Figure 6.

Figure 6. Amount of water that should be present in the petridish
- When grafting is complete, seal the petri dish with three layers of micropore tape and keep the grafts vertical at all times. Sealing with parafilm is not recommended-it is important that the grafts dry out somewhat in order to reconnect and they also need good aeration.
- Place petri dishes with grafts into growth room. They should remain undisturbed for at least 3 days, and can be left for up to 5 days. They may need addition of a little sterile water on day 3 or 4, but not too much!
- Set up the materials in the laminar flow cabinet as below: Rinse hands in 70% ethanol, wipe laminar flow surface and dissecting microscope with 96% ethanol.
Notes
- Good sharp double-edge razor blades will probably work as well as the sapphire or diamond knives, if used only a few times and discarded as soon as there is any resistance to cutting. Similarly, when the cutting is less smooth with either sapphire or diamond scalpels, exchange the blade. Diamond scalpels work very well, are robust, retain their sharp cutting edge for several months, and can be re-sharpened, but they are very expensive! Stainless steel surgical scalpels may also work well but these have not been tested by us. The advantage of the sapphire and diamond blades is that they are transparent, so it is easy to see exactly where you are cutting, which is important when dissecting very small tissues.
- The scion hypocotyls are cut immediately below where the cotyledon petioles attach to the hypocotyl. This helps to prevent adventitious roots growing from the scion and causing the graft to fail, especially if it takes a little longer for vascular tissue to rejoin. In general, adventitious roots emerging from the scion indicate a less successful graft union. If the adventitious roots are removed when still small, the graft will generally survive.
Acknowledgments
Note that a similar protocol has recently been described (Marsch-Martinez et al., 2013).
References
- Andersen, T. G., Nour-Eldin, H. H., Fuller, V. L., Olsen, C. E., Burow, M. and Halkier, B. A. (2013). Integration of biosynthesis and long-distance transport establish organ-specific glucosinolate profiles in vegetative Arabidopsis. Plant Cell 25(8): 3133-3145.
- Chen, A., Komives, E. A. and Schroeder, J. I. (2006). An improved grafting technique for mature Arabidopsis plants demonstrates long-distance shoot-to-root transport of phytochelatins in Arabidopsis. Plant Physiol 141(1): 108-120.
- Flaishman, M. A., Loginovsky, K., Golobowich, S. and Lev-Yadun, S. (2008). Arabidopsis thaliana as a model system for graft union development in homografts and heterografts. J Plant Growth Regul 27(3): 231-239.
- Turnbull, C. G., Booker, J. P. and Leyser, H. M. (2002). Micrografting techniques for testing long-distance signalling in Arabidopsis. Plant J 32(2): 255-262.
- Marsch-Martinez, N., Franken, J., Gonzalez-Aguilera, K. L., de Folter, S., Angenent, G. and Alvarez-Buylla, E. R. (2013). An efficient flat-surface collar-free grafting method for Arabidopsis thaliana seedlings. Plant Methods 9(1): 14.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Andersen, T. G., Liang, D., Halkier, B. A. and White, R. (2014). Grafting Arabidopsis. Bio-protocol 4(13): e1164. DOI: 10.21769/BioProtoc.1164.
Category
Plant Science > Plant physiology > Plant growth
Plant Science > Plant developmental biology > Morphogenesis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










