- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Flow Cytometric Analyses of Autophagic Activity using LC3-GFP fluorescence
Published: Vol 4, Iss 7, Apr 5, 2014 DOI: 10.21769/BioProtoc.1091 Views: 14178
Reviewed by: Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Isolation and Ex Vivo Testing of CD8+ T-Cell Division and Activation Using Mouse Splenocytes
Melissa Dolan [...] John M.L. Ebos
Aug 20, 2025 4012 Views

Detection of Autophagy in Human Peripheral Blood Mononuclear Cells Using Guava® Autophagy and Flow Cytometry
Melanie Scherer [...] Jörg Bergemann
Sep 20, 2025 1448 Views
Protocol for the Isolation and Analysis of Extracellular Vesicles From Peripheral Blood: Red Cell, Endothelial, and Platelet-Derived Extracellular Vesicles
Bhawani Yasassri Alvitigala [...] Lallindra Viranjan Gooneratne
Nov 5, 2025 1517 Views
Abstract
Flow cytometry allows very sensitive and reliable high-throughput analysis of autophagic flux. This methodology permits to screen cells in flow and capture multi-component images. Using this technology autophagic flux may be analysed accurately in both suspension as well as adherent cells upon trypsinization independent of how heterogeneous the LC3 punctae content might be. The method is based on the fact that intra-cellularly expressed LC3-GFP serves as a potential autophagic substrate for degradation. Therefore changes in total intracellular LC3-GFP fluorescence intensity is used as an indicator of cellular autophagic activity in living cells. Increased autophagic flux is expected to result in a progressive delivery of LC3-GFP to autolysosome where this substrate undergoes degradation. Therefore, enhanced autophagic flux is detected as a decreased total cellular GFP signal. On the other hand an inhibition of autophagic flux independent of the stage (autophagosome formation, maturation or acidification) leads to accumulation of undegraded LC3-GFP and may be detected as an enhanced intracellular GFP signal. (Caution: This methodology is based on the assumption that LC3-GFP is expressed constitutively by the model system. Data from analysis of substances or conditions influencing cellular LC3-GFP expression should be interpreted with care.)
Materials and Reagents
- Cell lines of interest (HepG2, HUH7, CMK, K562 etc.) stably expressing LC3-GFP
We recommend the following commercially available plasmids: pBABEpuro GFP-LC3 (plasmid 22405) and pBABE-puro mCherry-EGFP-LC3B (plasmid 22418) generated by Jayanta Debnath from Addgene to be inserted into retroviral constructs and used for cell transduction
- Eagle's minimal essential medium (EMEM) (ATCC, catalog number: 30-2003 ) containing 10% fetal bovine serum (FBS) with 100 U/100 μg/ml penicillin/streptomycin (Life Technologies, Gibco®, catalog number: 15140-122 )
- RPMI 1640 with L-glutamine (Lonza, catalog number: BE12-702F ) containing 10% FBS with 100 U/100 μg/ml penicillin/streptomycin
- FBS (Biochrom, catalog number: S0615 )
- Dulbecco’s Phosphate Buffered Saline (DPBS) (Biochrom, catalog number: L1825 )
- 1x 0.05% Trypsin-EDTA (phenol red) (Life Technologies, catalog number: 25300 )
- Hanks Balanced Salt Solution (HBSS) (Life Technologies, Gibco®, catalog number: 14025 ) containing 6 mM glucose (starvation medium)
- Rapamycin from Streptomyces hygroscopicus (1-5 µmol/L) (Sigma-Aldrich, catalog number: R0395 )
- PP242 hydrate (1-5 µmol/L) (Sigma-Aldrich, catalog number: P0037 )
- 3-methyladenine (3-MA) (3-10 mmol/L) (Sigma-Aldrich, catalog number: M9281 )
- Wortmannin (30-100 nmol/L) (Sigma-Aldrich, catalog number: W3144 )
- LY294002 (7-20 µmol/L) (Sigma-Aldrich, catalog number: L9908 )
- Nocodazole (12-50 µmol/L) (Sigma-Aldrich, catalog number: M1404 )
- Vinblastine (12-50 µmol/L) (Sigma-Aldrich, catalog number: V1377 )
- Ammonium chloride (NH4Cl) (10-20 mmol/L) (Sigma-Aldrich, catalog number: A0171 )
- Hydrohychloroquine sulphate (HCQ) (5-10 µmol/L) (Sigma-Aldrich, catalog number: H0915 )
- Chloroquine (CQ) (5-10 µmol/L) (Sigma-Aldrich, catalog number: C6628 )
- Dimethyl sulfoxide DMSO (Sigma-Aldrich, catalog number: D8418 )
Equipment
- 37 °C, 5% CO2 humidified incubator
- Centrifuge
- FACSCalibur, LSR II (BD) or analogous equipment
Procedure
- Maintain the cells under standard tissue culture conditions at 37 ºC, 5% CO2 in a humidified incubator. Keep cell density below 1 x 106/ml and analyse at subconfluent stages.
Caution: Prior to analysis cell should be kept for several hours (min 12 h) in fresh medium to avoid potential activation of autophagy due to nutrients exhaustion. Generally culture medium contains autophagy affecting substances: amino acids, glucose, growth factors, hormones etc. Take care when comparing autophagic flux under different conditions to normalize for all the necessary factors. Normalize also for the solvent used when analysing the effect of different substances on autophagy – for example DMSO, ethanol etc. might affect autopagic flux.
- Incubate the cells for the desired time and under the conditions of interest.
Caution: When analysing prolonged periods of time under conditions potentially affecting cell numbers or viability, differences in nutrient consumption and therefore abundance might occur and influence your results as autophagic activity is highly related to the nutritional status.
- Positive controls [rapamycin (1-5 µmol/L), PP242 (1-5 µmol/L), Hanks’ Balanced Salt Solution containing 6 mmol/L glucose (starvation medium)] and negative controls [3-methyladenine (3-MA) (3–10 mmol/L), wortmannin (30–100 nmol/L), LY294002 (7–20 µmol/L), nocodazole (12–50 µmol/L), vinblastine (12–50 µmol/L), ammonium chloride (NH4Cl) (10–20 mmol/L), hydrohychloroquine (HCQ) or chloroquine (CQ) (5–10 µmol/L)] may be also included. Positive controls activate autophagy and should result in a decreased total cellular LC3-GFP signal. Negative controls inhibit different stages of autophagic process and generally should lead to an increase in total cellular LC3-GFP signal.
We also recommend analysing the effect of a new substance not only on basic, but also on rapamycin, PP242 or starvation activated autophagic flux.
- At the end of incubation period transfer the cells to FACS tubes, put them on ice and analyse by flow cytometry plotting the data of cell counts as GFP/FITC fluorescence intensity measured on FL1 channel. For adherent cells, wash once with DPBS, trypsinize, wash again with DPBS, resuspend in DPBS (2% FBS), put on ice and analyse.
Caution: Generally pharmacological inhibitors of autophagy lack specificity and therefore some cell type specific effects might occur.
Table 1 may be used to facilitate the interpretation of the results.
Table 1. Analysis of unknown substance XX plus
Medium
Activator
I
→
↓↓
II
↓↓
↓↓↓
III
↑↑
↓ or ↑
Notes:- →↑↓: Change in LC3-GFP mean fluorescence intensity (MFI).
- (Activator): autophagy induction [rapamycin (1-5 µmol/L), PP242 (1-5 µmol/L) or starvation medium]. Suggested incubation time 4 to 6 h.
- Analysis of unknown substance X should include co incubation with one or more activators. Table 1 illustrates three possible scenarios. a) Scenario (I) the unknown substance X does not impact cellular autophagic activity; b) Scenario (II) X is an activator; c) Scenario (III) X is an inhibitor of autophagy.
- Caution: Although considered the most accurate sensitive and reliable method for analysis of autophygic flux, flow cytometry determination of LC3-GFP turnover should be combined with alternative methods with non overlapping limitations such as electron microscopy (EM) fluorescence microscopy (FM), western blotting (WB) etc.
- Example data:
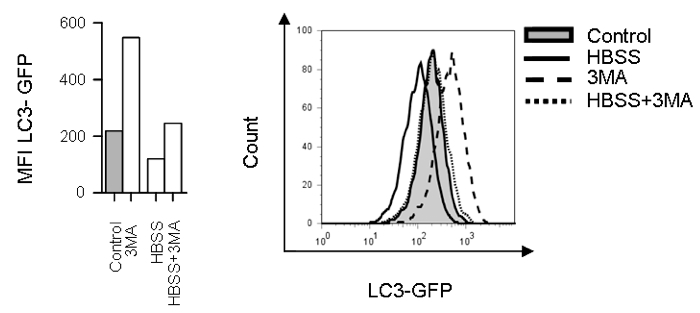
Figure 1. Flow cytometry analysis of autophagic flux in K562 cells expressing LC3-GFP fusion protein incubated with autophagy inhibitor 3MA (10 mM) in the presence or absence of autophagy activation through starvation (HBSS) (left) with representative histograms (right)
- →↑↓: Change in LC3-GFP mean fluorescence intensity (MFI).
Acknowledgments
Stankov, M and Behrens, G were supported by the German Research Foundation (KFO 250, BE 2089/2-1 and Excellence Cluster REBIRTH, EXC 62/1). Leverkus, M was supported by the German Research Foundation (LE-953/5-1 and LE-953/6-1).
References
- Sinicrope, F. A., Sirko, A., Siu, P. M. et al. (2012). Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8(4): 445-544.
- Mizushima, N., Yoshimori, T. and Levine, B. (2010). Methods in mammalian autophagy research. Cell 140(3): 313-326.
- Stankov, M. V., El Khatib, M., Kumar Thakur, B., Heitmann, K., Panayotova-Dimitrova, D., Schoening, J., Bourquin, J. P., Schweitzer, N., Leverkus, M., Welte, K., Reinhardt, D., Li, Z., Orkin, S. H., Behrens, G. M. and Klusmann, J. H. (2014). Histone deacetylase inhibitors induce apoptosis in myeloid leukemia by suppressing autophagy. Leukemia 28(3): 577-588.
- Stankov, M. V., Panayotova-Dimitrova, D., Leverkus, M., Schmidt, R. E. and Behrens, G. M. (2013). Thymidine analogues suppress autophagy and adipogenesis in cultured adipocytes. Antimicrob Agents Chemother 57(1): 543-551.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Stankov, M., Panayotova-Dimitrova, D., Leverkus, M. and Behrens, G. (2014). Flow Cytometric Analyses of Autophagic Activity using LC3-GFP fluorescence. Bio-protocol 4(7): e1091. DOI: 10.21769/BioProtoc.1091.
Category
Microbiology > Antimicrobial assay > Autophagy assay
Cell Biology > Cell-based analysis > Flow cytometry
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










