- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
An ex vivo Model of HIV-1 Infection in Human Lymphoid Tissue and Cervico-vaginal Tissue
Published: Vol 4, Iss 4, Feb 20, 2014 DOI: 10.21769/BioProtoc.1047 Views: 15787
Reviewed by: Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Isolation of Exosomes from Semen for in vitro Uptake and HIV-1 Infection Assays
Marisa N. Madison [...] Chioma M. Okeoma
Apr 5, 2017 12866 Views
Establishment of a Human Cell Line Persistently Infected with Sendai Virus
Christopher Coakley [...] Saurabh Chattopadhyay
Aug 20, 2017 9139 Views
Ex vivo Human Skin Infection with Herpes Simplex Virus 1
Nydia C. De La Cruz [...] Dagmar Knebel-Mörsdorf
May 5, 2022 3088 Views
Abstract
Human tissue explants are a valuable tool to study the interactions between host and infectious agents. They reliably mimic many important aspects of tissue cytoarchitecture and functions and allow us the investigation of the mechanisms of microbial pathogenesis under controlled laboratory conditions. One of the advantages of this system is that, unlike isolated cells, infection of tissue blocks with HIV-1 does not require exogenous stimulation with mitogens or activating factors. Here we describe a protocol to infect with HIV-1 human lymphoid tissue from tonsils and cervico-vaginal tissue and maintain them in culture in a non-polarized setting. These ex vivo infected tissues can be used as fruitful models to study HIV-1 pathogenesis and HIV-1 vaginal transmission, respectively, as well as an efficient platform for testing anti-HIV therapeutic and preventative strategies.
Materials and Reagents
- Tonsillar tissue obtained from routine tonsillectomy (Age range of patients: 2-10)
- Cervico-vaginal tissue obtained from routine hysterectomy (Age range of patients: 35-55)
- Gelfoam 12-7 mm adsorbable gelatin sponge (Pfizer, NDC: 0009-0315-08 )
- RPMI 1640 (Life Technologies, Gibco®, catalog number: 31870-025 )
- 100x Modified Eagle's medium (MEM)-nonessential amino acids (10 mM) (Life Technologies, Gibco®, catalog number: 11140-035 )
- 100x MEM sodium-pyruvate (100 mM) (Life Technologies, Gibco®, catalog number: 11360-070 )
- 1,000x Gentamicin (50 mg/ml) (Corning, cellgro®, catalog number: 30-005-CR )
- 100x Fungizone (250 μg/ml, amphotericin B) (Life Technologies, Gibco®, catalog number: 15290-018 )
- Fetal bovine serum (FBS) (Gemini Bio-products, catalog number: 100-106 )
Note: We advise testing several lots of serum for culture optimization and use the same lot of FBS for an entire series of experiments. We always test several serum lots on tissues from several donors and select the lot that gives the highest HIV-1 replication. Also, FBS can affect the ability of tissue to secrete cytokines in culture medium. - Phosphate Buffered Saline (PBS) (pH 7.4) (Life Technologies, Gibco®, catalog number: 10010-023 )
- Sterile water, cell culture grade (Quality Biological, catalog number: 118-162-101 )
- HIV-1 viral preparation(s)
Note: For most of our experiments we use the following viral preparations: HIV-1BaL and HIV-1LAI.04 obtained from the Virology Quality Assurance Laboratory at Rush University (Chicago, IL). Viral stocks were obtained from the clarified culture medium of peripheral blood mononuclear cell cultures inoculated with either HIV-1BaL or HIV-1LAI.04, originally received from the NIH AIDS Reagent Program. HIV-1 p24gag concentrations were 49 ± 3 ng/ml and 53 ± 3 ng/ml for HIV-1BaL and HIV-1LAI.04 stock, respectively. For more viral preparations used in our experimental setting see Introini et al., 2013 and Vanpouille et al., 2012. - Timentin (GlaxoSmithKline, NDC: 0029-6571-26 ) (see Recipes)
Note: Timentin is the commercial name of a mix of the antibiotics ticarcillin and clavulanate that are commercially available as individual reagents. These antibiotics efficiently prevent growth of bacteria that can occasionally contaminate tissue samples after surgery. Penicillin and streptomycin can be used instead of Timentin, although they have different properties. For example, Timentin displays low stability at room temperature or 37 °C (about 24 h) therefore, once added to culture medium, it remains active only for the first day of culture. - Culture medium (CM) (see Recipes)
Equipment
- Petri dish (100 mm x 20 mm) (BD, Falcon®, catalog number: 353003 )
- Petri dish (150 mm x 25 mm) (BD, Falcon®, catalog number: 353025 )
- 6-well plates (Corning, Costar®, catalog number: 3506 )
- 12-well plates (Corning, Costar®, catalog number: 3513 )
- 5, 10-ml pipettes (BD, Falcon®)
- Sealed screw-cap 1.5/2-ml tubes (SARSTEDT AG)
- Thermomixer with block for 1.5 ml-tubes (Eppendorf)
- Forceps or tweezers
- Scalpels and blades nos. 10 and 23
- Scissors
- Flat weighing metallic spatula
- 37 °C, 5% CO2 incubator set at 90% humidity
- Water bath
- 10, 50-ml syringe plunger
Procedure
- Allow CM enough time to reach room temperature or put it in a water bath pre-warmed at 37 °C.
- Calculate the number of Gelfoam pieces needed per experiment on the basis of the type of tissue used and the number of wells required to perform the experiment (Table 1).
Table 1. Culture setupTissue type No. of Gelfoam pieces (from 1 sponge) Culture plate Volume of culture medium (per well) (ml) No. of blocks (per Gelfoam piece) No. of wells (replicates) per experimental condition Tonsillar 4 6-well 3 9 3 or more Cervico-vaginal 5-6 12-well 1 8 2 or more - Depending on the number of Gelfoam pieces needed, either fill a 100 mm x 20 mm Petri dish with about 100 ml of CM supplemented with Timentin or a 150 mm x 25 mm Petri dish with about 200 ml (if more than 3 Gelfoam sponges are required).
- Put the Gelfoam sponge(s) into the Petri dish using ethanol-sterilized forceps and press the sponge(s) against the bottom of the Petri dish for about 2 min using a bent flat spatula sterilized with ethanol.
Note: The Gelfoam is extremely brittle when dehydrated. The hydration process should be done carefully especially when pushing down on the foam to chase the air out. The Gelfoam should be as free of air as possible: the presence of air will block the capillaries through which nutrients reach the tissue. - Use ethanol-sterilized scissors to cut the rehydrated Gelfoam sponge(s) into pieces of the appropriate size (Table 1).
- Place one piece of Gelfoam into each well using the forceps and add the appropriate amount of CM containing Timentin with a pipette (Table 1).
- Place the plates into the incubator.
Note: The dissection and infection protocols vary depending on the tissue in use. If using tonsils follow option A; if using cervico-vaginal tissue, follow option B.
- Tonsillar tissue
- Put one tonsil into a 100-mm Petri dish containing CM supplemented with Timentin and cut the tonsil into several large pieces (4–5 pieces).
- Transfer one piece into the lid of a Petri dish containing few ml of CM. Cover the Petri dish containing the remaining tonsil pieces to avoid desiccation of tissue.
- With a 10- or 23-gauge blade scalpel and forceps, remove cauterized parts and fibroid tissue. Remove bloody/inflamed tissue and any parts containing tonsillolithes and/or with green-brownish color (Figure 1, see Grivel and Margolis, 2009 for additional pictures).
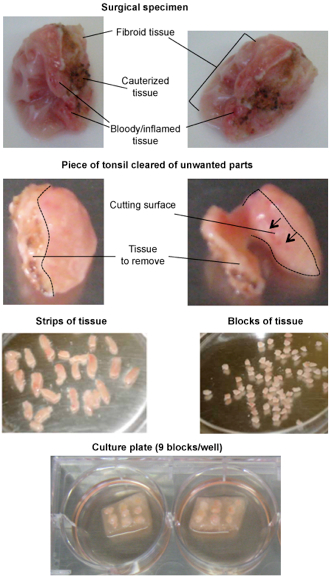
Figure 1. Preparation of tonsillar tissue for culture
(Up) In the received surgical specimens the parts that should be removed are residual fibroid tissue from the capsule surrounding palatine tonsils (white), cauterized tissue (brown-black), bloody/inflamed tissue (red) and any other parts with brown-greenish color.
(Middle) Remove unwanted large parts of tissue with a neat cut holding the specimen with forceps. The dotted line delimitates the cutting surface to remove a layer of cauterized tissue. For small parts it may be easier to directly pinch them with forceps to cut them out.
(Down) Strips of tissue are dissected into blocks of approximately 8 mm3. 9 blocks are transferred on each Gelfoam piece in a 6-well plate. - Cut the tissue piece into slices of about 2 mm in thickness. Remove any undesirable part as mentioned above, and cut the slices into strips 2 mm thick. Then cut the strips into blocks 2 mm thick. This should result in blocks of roughly 8 mm3.
- Cut as many blocks as needed, at least 27 blocks are required per experimental condition (Table 1). With the forceps, transfer the blocks into a clean Petri dish containing CM supplemented with Timentin to avoid desiccation of tissue blocks while proceeding with dissection.
Note: Since dissection may take long time, it is important to keep adding medium to the Petri dish lid to maintain tissue wet while cutting. - Swirl the plate to randomize the distribution of the blocks.
- Take out the six-well plate(s) containing Gelfoam from the incubator and place 9 blocks of tissue on top of each Gelfoam piece using the forceps.
- Return the plate(s) to the incubator for overnight culture or proceed with infection (go to step A11).
Note: We prefer performing infection after overnight incubation to make sure that no bacterial or fungal contamination develops. In addition, a large population of cells egresses tonsillar tissue blocks after dissection. This process is largely completed within the first 24 h of culture. - If tissue was dissected the previous night, aspirate the CM in the six-well plate(s) with a pipette and discard it. Tilt the plate and gently push the Gelfoam back to the upper part of the well to allow the medium to gather at the bottom, aspirate and discard it.
- With a pipette, add to each well 3 ml of Timentin-containing CM pre-warmed at 37 °C.
- Thaw HIV-1 viral preparation(s) in the water bath pre-warmed at 37 °C. If necessary, dilute viral stock to an appropriate concentration. It is preferable to have a dilution such that the desired inoculum is contained in 5 to 8 μl of medium.
Note: For most of our experiments we use 6.0 to 7.5 μl of undiluted HIV-1BaL or HIV-1LAI.04 viral stock (Material and Reagents 12) to infect each tonsillar tissue block. It is critical for an efficient infection to allow the minimum time required between thawing HIV-1 viral preparation and depositing the inoculum on tissue blocks. - Pipette the inoculum directly on top of each of the 9 blocks on a Gelfoam piece.
Note: For an accurate delivery of the inoculum volume we suggest using a reverse pipetting technique and changing tip for every well. - Return the plate to the incubator as soon as infection is completed.
- Cervico-vaginal tissue
- Put the tissue piece(s) in a 100-mm Petri dish containing CM supplemented with Timentin.
- Transfer one piece of tissue into the lid of a Petri dish containing few ml of CM. Cover the Petri dish containing the remaining tissue to avoid desiccation.
- With a 23-gauge blade scalpel and forceps separate the mucosa of ectocervix and endocervix from the stromal tissue beneath to obtain strips of mucosa (Figure 2).
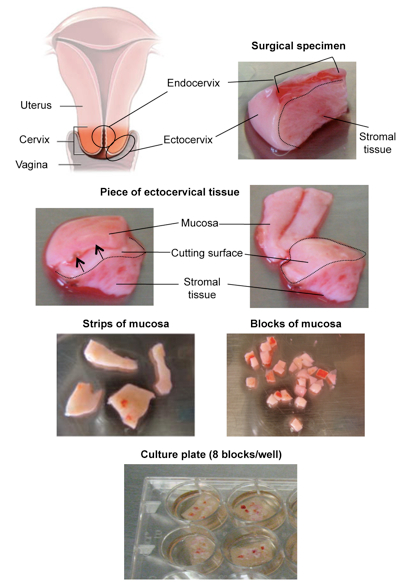
Figure 2. Preparation of cervico-vaginal tissue for culture
(Up) The endocervix is the part of the uterus lining the lumen of the canal that provides communication between the uterine cavity and that of the vagina. The portion of the cervix that projects into the vagina through the upper anterior vaginal wall is called ectocervix. In the received cervico-vaginal tissue specimens the endocervical mucosa is readily recognized as the part with darker-red color and coated by thick mucus, whereas the ectocervical mucosa has a clearer color.
(Middle) The surgical specimen can be cut into smaller pieces to facilitate the removal of the mucosa. Holding the stromal tissue with forceps, separate it from the mucosa cutting the tissue specimens in the longitudinal direction (indicated by the arrows) with respect to the mucosal layer. The dotted line delimitates the cutting surface and should be located 2-3 mm below the epithelial edge of the tissue specimen.
(Down) Strips of mucosa are dissected into blocks of approximately 8 mm3. After infection 8 blocks are transferred on each Gelfoam piece in a 12-well plate. - Cut the mucosa strips into strips of 2 mm in thickness, remove and discard as much connective-muscular tissue as possible, leaving only a layer 2 mm thick comprising the mucosal epithelium, sub-mucosa and part of the underlying stroma. Cut the strips into blocks 2 mm thick. This should result in blocks of roughly 8 mm3.
- Cut as many blocks as needed, at least 16 blocks are required per experimental condition (Table 1). With the forceps, transfer the blocks into a clean Petri dish containing CM supplemented with Timentin to avoid desiccation of tissue blocks while proceeding with dissection.
Note: For most of our experiments we pool blocks from the endocervix and ectocervix. One may consider keeping them separated because of their different structural and immunologic features (Pudney et al., 2005). Also, the endocervix usually continues to produce mucus in culture and this might interfere with subsequent assays. Note that in the received cervico-vaginal tissue specimens the endocervix is much smaller than the ectocervix. - Swirl the plate to randomize the distribution of the blocks.
- With the forceps, transfer the blocks into sterile 1.5-ml conical tube(s) (maximum 16 blocks per tube).
- Thaw HIV-1 viral preparation(s) in the water bath pre-warmed at 37 °C. If necessary, dilute the viral stock to an appropriate concentration and transfer it into the tube(s) containing the blocks.
Notes:
a. Cervico-vaginal tissue is best infected at the time of dissection. We recommend including an experimental condition in which both the virus and a powerful antiviral drug are combined. This is a useful control to ascertain whether the virus actually replicates rather than being adsorbed and released by the tissue.
b. For most of our experiments we use 500 μl of undiluted HIV-1BaL viral stock (Material and Reagents 12) to infect 12-16 cervico-vaginal tissue blocks. We add lamivudine 10 μM to HIV-1 preparation and to CM during the entire culture length as control for HIV-1 release. - Transfer the tubes into a Thermomixer pre-warmed at 37 °C and set mixing at 300 revolutions per minute. Incubate for 2 h, gently inverting the tubes few times every half an hour.
Note: It is critical for an efficient infection to allow the minimum time required between thawing HIV-1 viral preparation and transferring the tubes to 37 °C. - Fill the wells of a 6-well plate (or more plates if more than six tubes were used for infection) with 3-4 ml of PBS.
- Discard the viral preparation from the tube(s). Using the forceps, gently transfer the blocks to the 6-well plate lid to remove the excess of viral preparation remained on the blocks, and then transfer the blocks into a well containing PBS.
- Swirl gently with a pipette and wait for 1 min. With a pipette, aspirate the PBS taking care not to aspirate the blocks.
- Add 3-4 ml of PBS to the well, swirl gently with a pipette tip and wait for 1 min.
- Repeat steps B12-13 three times.
Note: These washing steps are necessary because HIV-1 replication in cervico-vaginal tissue is not as high as in tonsillar tissue and can be masked by the subsequent release of viruses absorbed on the tissue. - Take a 12-well plate out of the incubator and transfer 8 blocks on top of each Gelfoam piece using the forceps.
- Return the plate to the incubator.
Common to A and B
- Three days after infection with HIV-1, harvest an aliquot of medium from each of the replicate wells and transfer it into a screw cap tube.
Note: We usually pool aliquots of replicate wells. - Using ethanol-sterilized forceps put back any block of tissue that may have dropped from the Gelfoam.
- With a pipette, discard the remaining medium from the plate. Tilt the plate, gently bring the Gelfoam to the upper part of the well and let the medium flow down to the bottom, aspirate and discard it.
- With a pipette, add the adequate volume of fresh CM pre-warmed at 37 °C (Table 1).
Note: It is not required to supplement CM with Timentin at this stage. - Return the plate(s) to the incubator.
- Store medium aliquots at -80 °C.
- Repeat steps 1 to 5 every 3 days until day 12-15.
- At day 12-15 post-infection collect tissue blocks using forceps and process them as required by the experiment.
- For RNA extraction we usually keep tissue blocks in RNA later overnight at 4 °C before storing them at -80 °C. For flow cytometry we immediately process tissue blocks by digesting them with an enzymatic cocktail to obtain a single-cell suspension.
Note: During culture, tissue outgrows into the Gelfoam pieces. Also individual cells may migrate out of the tissue into the gel. One can analyze the part of tissue that is above the gel separately by pinching it out with the forceps. The cells within the gel can be squeezed out using the forceps or a syringe plunger. However, one should keep in mind that what extracted from the gel is not just a fraction of migrated cells but rather a mixture of such cells and tissue structures. - Dispose plates with any residual CM, tissue blocks and Gelfoam as per the institution’s biosafety practices required for handling human infectious waste.
- A number of techniques can be used to assess HIV-1 production and replication in tissue blocks. Our standard read-out is to measure HIV-1 p24gag in culture supernatants harvested throughout culture time using a fluorescent bead-based immunoassay (Biancotto et al., 2009).
Note: Tissues from different donors infected with the same inoculum of the same HIV-1 stock may give different results as measured by p24gag accumulation in culture supernatant (Table 2). This may be due to several factors, such as the number and the activation status of HIV-1 target cells and the presence of co-infecting pathogens, which affect HIV-1 replication and production in tissue blocks. Therefore it is pivotal in the set up of different conditions of an experiment to use tissue from only one donor (donor-matched conditions).
Table 2. Reference values of HIV-1 p24gag inoculum and relative production in tissue explants. HIV-1 p24gag production is indicated as cumulative value of the p24gag values measured in culture supernatant sampled every 3 days over 12 days of culture (day6+day9+day12). The value of p24gag measured at day 3 was excluded because it does also contain p24gag released from the HIV-1 inoculum absorbed on tissue blocks together with newly produced p24gag as a result of viral replication. The value of HIV-1 production in cervico-vaginal tissue infected with HIV-1BaL is normalized by the p24gag value measured in culture supernatant of donor-matched blocks infected and cultured in the presence of lamivudine 10 μM. Infection of cervico-vaginal tissue with HIV-1LAI.04 does not lead to detectable HIV-1 replication in our experimental setting. HIV-1 p24gag was measured using a fluorescent bead-based immunoassay (Biancotto et al., 2009).Tissue type Virus Inoculum per block (pg of HIV-1 p24gag) No. of blocks HIV-1 p24gagproduction (ng/ml)Tonsillar HIV-1BaL 367.5 27 1-10 HIV-1LAI.04 397.5 27 10-100 Cervico-vaginal HIV-1BaL 1531.2 16 1-5 HIV-1LAI.04 1656.2 16 Undetectable
Notes
- For our experiments we use tissues collected from indicated surgeries that are not harvested for the purpose of the study and therefore can be considered as 'pathological waste'. In any case, human tissues must be harvested according to a protocol approved by an ethical review board. Collecting personal and medical data, such as use of drugs, menstrual cycle, sexual behavior, use of contraceptives, etc. from tissue donors may provide important information to interpret experimental results. For an example see Saba et al., 2013. Collection of these data poses ethical issues concerning informed consent and privacy that must be addressed in the protocol for tissue harvest.
- The tissue delivered to the laboratory should be in good condition, having a “healthy” pinkish color and lacking large blood clots and greenish necrotic centers. The state of the tissue is also dependent on how quickly it is delivered for processing. It is important for long-term culture to start the experiment with a healthy tissue. Tonsillar tissue is soft and prone to deterioration. According to our experience, this tissue should be delivered within 3-5 h post-surgery. We have noticed that cooling on ice during delivery makes it less suitable for long-term culture. Therefore we maintain tonsillar tissues in PBS at room temperature during delivery. Cervico-vaginal tissue is much sturdier and we successfully infected with HIV-1 and cultured tissues delivered 24 h post-surgery. We use ice-cold RPMI 1640 as delivery medium.
- The operator should be informed on the risks of working with blood-borne pathogens and receive proper training to work in a Biosafety Level 2 laboratory in order to handle tissue specimens, even if the experiments do not involve the use of HIV-1.
- Take note, and pictures for the more meticulous experimenters, of any feature of tissue specimens that you suspect may affect experimental results, such as bloody, cauterized and necrotic parts. We suggest keeping the medium in which the dissection is performed as well as any unused piece(s) of tissue because they might be useful to assess cell viability, for archiving DNA/RNA or histological analysis.
- The values of HIV-1 inoculum reported in Table 2 were optimized to perform infection with our viral stocks of tissue specimens delivered to the laboratory and processed following standard procedures to minimize any variability not due to the intrinsic nature of the tissue. Nevertheless, the production of HIV-1 varies remarkably between tissues from different donors. Therefore we invite the readers to take the values reported in Table 2 as a reference, bearing in mind that multiple inter-donor factors affect the outcome of an infection. For this reason we recommend to use exclusively donor-matched experimental conditions and perform an appropriate statistical analysis on data averaged from multiple donors.
- The experimental system described above is non-polarized meaning that penetration of HIV-1 may occur from all sides of a tissue block. The aim of this system is not to simulate HIV-1 transmission through the epithelium but rather to investigate the mechanisms that regulate HIV-1 spreading in tissues. For cervico-vaginal tissue, this culture system may still model HIV-1 transmission as there is evidence in vivo that HIV-1 can reach the subepithelial layer of the mucosa through pre-existing microabrasions of the epithelium or caused by sexual intercourse. Experimental settings of polarized infection with HIV-1 and culture of tissue explants have been described by others in Dezzutti and Hladik, 2013. Advantages and disadvantages of both systems as a model of HIV-1 transmission are described in Merbah et al., 2011.
Recipes
- Timentin
Prepare a working 100x stock solution by adding 100 ml of sterile water to a 3.1 g vial of Timentin. Aliquot in sterile screw-cap tubes, freeze and keep aliquots at -30 °C. Unused thawed aliquots should be discarded. - Culture medium (CM)
Remove 90 ml (set aside for other use) from a 500 ml bottle of RPMI1640
add
75 ml of FBS
5 ml of 100x MEM-nonessential amino acids
5 ml of 100x fungizone
5 ml of 100x MEM-sodium pyruvate
500 μl of 1,000x gentamicin
Stored at 4 °C
Supplement CM with Timentin only before use. Dilute 1 ml of freshly thawed 100x Timentin in 99 ml of CM. Prepare the volume needed for the experiment on the basis of the indications in Table 1 considering that extra CM is required to prepare Gelfoam and cut tissues.
Note: We usually prepare large volume of CM (4 bottles) that we store at 4 °C and use to culture tissues within 2 weeks from preparation. Older CM can be used to keep tissue blocks during dissection, after being supplemented with Timentin.
Acknowledgments
This protocol was adapted from the following publications: Introini et al. (2013) and Grivel and Margolis (2009). This work was supported by the Intramural program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
References
- Biancotto, A., Brichacek, B., Chen, S. S., Fitzgerald, W., Lisco, A., Vanpouille, C., Margolis, L. and Grivel, J. C. (2009). A highly sensitive and dynamic immunofluorescent cytometric bead assay for the detection of HIV-1 p24. J Virol Methods 157(1): 98-101.
- Dezzutti, C. S. and Hladik, F. (2013). Use of human mucosal tissue to study HIV-1 pathogenesis and evaluate HIV-1 prevention modalities. Curr HIV/AIDS Rep 10(1): 12-20.
- Grivel, J. C. and Margolis, L. (2009). Use of human tissue explants to study human infectious agents. Nat Protoc 4(2): 256-269.
- Introini, A., Vanpouille, C., Lisco, A., Grivel, J. C. and Margolis, L. (2013). Interleukin-7 facilitates HIV-1 transmission to cervico-vaginal tissue ex vivo. PLoS Pathog 9(2): e1003148.
- Merbah, M., Introini, A., Fitzgerald, W., Grivel, J. C., Lisco, A., Vanpouille, C. and Margolis, L. (2011). Cervico-vaginal tissue ex vivo as a model to study early events in HIV-1 infection. Am J Reprod Immunol 65(3): 268-278.
- Pudney, J., Quayle, A. J. and Anderson, D. J. (2005). Immunological microenvironments in the human vagina and cervix: mediators of cellular immunity are concentrated in the cervical transformation zone. Biol Reprod 73(6): 1253-1263.
- Saba, E., Grivel, J.C., Vanpouille, C., Brichacek, B., Fitzgerald, W., Margolis, L. and Lisco, A. (2010). HIV-1 sexual transmission: early events of HIV-1 infection of human cervico-vaginal tissue in an optimized ex vivo model. Mucosal Immunol 3(3): 280-290.
- Saba, E., Origoni, M., Taccagni, G., Ferrari, D., Doglioni, C., Nava, A., Lisco, A., Grivel, J. C., Margolis, L. and Poli, G. (2013). Productive HIV-1 infection of human cervical tissue ex vivo is associated with the secretory phase of the menstrual cycle. Mucosal Immunol 6(6): 1081-1090.
- Vanpouille, C., Lisco, A., Introini, A., Grivel, J.C., Munawwar, A., Merbah, M., Schinazi, R. F., Derudas, M., McGuigan, C., Balzarini, J. and Margolis, L. (2012). Exploiting the anti-HIV-1 activity of acyclovir: suppression of primary and drug-resistant HIV isolates and potentiation of the activity by ribavirin. Antimicrob Agents Chemother 56(5): 2604-2611.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Introini, A., Vanpouille, C., Grivel, J. C. and Margolis, L. (2014). An ex vivo Model of HIV-1 Infection in Human Lymphoid Tissue and Cervico-vaginal Tissue. Bio-protocol 4(4): e1047. DOI: 10.21769/BioProtoc.1047.
- Introini, A., Vanpouille, C., Lisco, A., Grivel, J. C. and Margolis, L. (2013). Interleukin-7 facilitates HIV-1 transmission to cervico-vaginal tissue ex vivo. PLoS Pathog 9(2): e1003148.
Category
Microbiology > Microbe-host interactions > Ex vivo model
Immunology > Host defense > Human
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











