- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Protocol for Measurement of Intracellular pH
Published: Vol 4, Iss 2, Jan 20, 2014 DOI: 10.21769/BioProtoc.1027 Views: 23501
Reviewed by: Lin FangFanglian He

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Quantitative Determination of Ca2+-binding to Ca2+-sensor Proteins by Isothermal Titration Calorimetry
Seher Abbas and Karl-Wilhelm Koch
Apr 5, 2020 6338 Views

Quantification of Salivary Charged Metabolites Using Capillary Electrophoresis Time-of-flight-mass Spectrometry
Masahiro Sugimoto [...] Tomoyoshi Soga
Oct 20, 2020 4655 Views
Detection and Quantification of Calcium Ions in the Endoplasmic Reticulum and Cytoplasm of Cultured Cells Using Fluorescent Reporter Proteins and ImageJ Software
Shunsuke Saito and Kazutoshi Mori
Aug 20, 2023 2889 Views
Abstract
Intracellular pH (pHi) is an important physiological determinant of enzyme activity and cellular function (Kurkdjian and Guern, 1989). All proteins depend on a tightly regulated pH to maintain their structure and function. Protonation–deprotonation events can dictate the charge of biological surfaces and are integral steps in many metabolic reactions (Casey et al., 2010). Moreover, the proton gradient across the mitochondrial membrane is used to generate cellular energy and support other mitochondrial processes. As a result, cells have developed multiple mechanisms to maintain a narrow range of pHi in response to extra- and intracellular fluctuations in pH (Orij et al., 2012). Here, we describe a protocol for pHi measurement in live cells that uses fluorescent microscopy and the pH sensitive dye 2’,7’-Bis-(2-Carboxyethyl)-5-(and-6-)-Carboxyfluorescein Acetoxymethyl Ester (BCECF-AM). This method was recently used to determine the effects of intracellular pH changes on global histone acetylation levels (McBrian et al., 2013).
Materials and Reagents
- Hela cells
- Nigericin (Sigma-Aldrich, catalog number: N7143 )
Prepare 10 mM nigericin (1:10,000 ) stock in ethanol (store aliquots at -20 °C and keep on ice during experiment) - DMSO (Sigma-Aldrich, catalog number: D2650 )
- BCECF-AM (Life Technologies, catalog number: B1170 )
- Glucose
- 1% Antibiotic-Antimycotic (Gibco®, catalog number: 15240 )
- DMEM (Mediatech, Cellgro®, catalog number: 10-013-CV )
- 10% FBS
- Paraffin wax
- Calibration solution (see Recipes)
- 10x Earle’s balanced salt solution (EBSS) stock without glucose and without sodium bicarbonate (see Recipes)
- Loading solution (see Recipes)
- Cell culture medium (see Recipes)
Equipment
- Perfusion inserts for 35 mm Dishes (Warner Instruments, catalog number: RC-33DM )
- 35 mm poly-lysine coated glass bottom culture dishes (MatTek, catalog number: P35GC-1.0-14-C )
- 50 ml conical tube
- Needles
- Aluminum foil
- Axiovert 200 M Zeiss florescent microscope equipped with a high-resolution video camera (ZEISS, Axio CAM MRm)
- BCECF filter set (Chroma Technology Corporation, catalog number: 71001a )
- Neutralize density filter (ND 1.0 A - 10.0% transmission-25mm; this is custom-designed for a given microscope) (Chroma Technology Corporation)
- Tubing and connections for perfusion
- Flow pump (Rainin Peristaltic pump Dynamax RP-1. 4-channel; catalog number is different based on supplier)
- 5% CO2 tank
- 37 °C Water bath
- 5% CO2 incubator (make sure it is calibrated regularly; variations in CO2 concentration can have dramatic effects on pH, generating noise in the experiment)
Software
- AxioVision 4.8
- Slidebook 4.2
- SigmaPlot
- Excel or any spreadsheet analysis software
Procedure
- Grow cells (~105) in 35 mm glass bottom dishes to 40% confluency.
Note that in comparison to plastic dishes it takes longer for cells to attach to glass bottom dishes. - Treat cells with drug of interest or siRNA for the desired length of time up to when cells reach ~40% confluency.
- A perfusion system should be installed to reduce background signal due to BCECF leaking out of the cell and to keep media temperature and pH constant throughout pHi measurement.
- Outflow and inflow tubes should have the same length to prevent overflow or drying of the plate.
- 2 ml/min is the recommended rate of perfusion; faster rates may have a shearing effect on the cells which affect the pHi.
- To connect the outflow and inflow tubes to the dish, we used needles that we bent into the shape of hooks and fixed them to the dish with paraffin wax. Premade sets are available commercially: https://www.warneronline.com/product_info.cfm?id=958.
- For efficient flow and to prevent turbulence, we used perfusion inserts for 35 mm dishes and fixed them in the dish with paraffin wax. We recommend testing the smoothness of the flow by a visible dye such as trypan blue to confirm efficient flow before the start of experiment.
- Wash perfusion system with distilled water for 4 h prior to the experiment to remove any residue - especially nigericin from a previous experiment – inside tubes.
- Outflow and inflow tubes should have the same length to prevent overflow or drying of the plate.
- Set up a rack inside a water bath at 37 °C to hold a 50 ml conical tube containing the perfusion solution which is the same as the loading solution. Bubble 5% CO2 at slow to moderate rates (~1 bubble/sec) for at least 20 min into the loading solution prior the experiment to equilibrate pH, similar to the conditions inside an incubator. Continue bubbling 5% CO2 into the loading solution during the experiment to maintain pH.
- Have a calibrated pH meter handy. The pH of the loading solution can be continuously monitored to ensure consistency.
- Set up the perfusion system and the settings on the microscope. Install the Chroma filter-set for BCECF and define the channels for the software. Check the filter wheel and make sure the excitation and emission channels are defined correctly in the Axiovision software.
- Make sure bulb intensity is at 50% and filter density at 10% to prevent phototoxicity to the cells.
- Open Axiovision and set exposure times to 100 msec for 440 nm and 200 msec for 495 nm. Emission is set at 535 nm. Also specify the program to take 6 pictures at 30 sec interval.
- Dilute a vial of BCECF in 80 µl DMSO. Use pre-aliquotted BCECF powder to prevent repeated freezing and thawing cycles (the BCECF dye does not work well after 2 freeze-thaw cycles).
- Remove media from cells and wash cells with 3 ml of loading solution to remove residual media (certain amino acids interfere with dye loading).
- Pipet 2 ml 1x EBSS onto the plate of cells followed by addition of 10 µl of BCECF directly onto the plate. Mix gently by pipetting the EBSS-BCECF solution up and down.
- Place cells in the incubator for 25 min. Start flowing loading solution through the perfusion system 5 min before the incubation is done.
- To prevent photo-bleaching, wrap the cell plate with a piece of aluminum foil as soon as it is removed from the incubator and is being transferred to the microscope room. Turn off the lights in the room (the light from the computer screen should provide ample light).
- (Work fast in this step) Immediately mount the loaded dish on the microscope stage and fix it in place with paraffin wax to prevent any movement during image acquisition. Install perfusion insert and inflow and outflow tubes. Run the perfusion system and meanwhile focus the microscope to find a suitable area – about 40% confluent – for taking pictures. We recommend using the halogen light at the weakest possible intensity for focusing and then using fluorescent light for fine focusing to reduce the risk of cell toxicity. 10x–40x magnification could be used. With 10x magnification larger number of cells can be evaluated.
- Let loading solution run through the system for 2-3 min and then take 6 pictures at 30 sec intervals (automated at step 6) (Figure 1).

Figure 1. The final set up for intracellular pH measurement is shown. The components of the system are labeled. - Import the pictures to Slidebook (make sure you define the channels correctly in Slidebook).
- After importing the pictures, define the background by selecting an area close to the middle of the picture with some cells nearby. Then select the cells using the Mask tool. In the Ratio tool, define 495 as the numerator and 450 as the denominator. Extract the ratio data of 495/450 channels and import the data to Excel. Remove outliers. Convert the ratio data to pH values using the calibration equation (see below).
In situ calibration
- Intracellular pH measurements with BCECF are made by determining the ratio of emission intensity at 535 nm when the dye is excited at ~490 nm (pH-dependent) versus the emission intensity when the dye is excited at its isosbestic point of ~440 nm (non pH-dependent). In situ calibration of BCECF's fluorescence is done the same way but in presence of 10–50 µM nigericin and 100–150 mM K+ to equilibrate internal and external pH. In situ calibration should be done for every experiment.
- After pictures are taken, aspirate the loading solution carefully from the loaded and fixed plate on the microscope and add 2 ml of calibration solution slowly using the side of the plate. Aspirate the calibration solution and then slowly add 2 ml of calibration solution with nigericin using a pipette. Wait 5 min before taking 2 pictures. Repeat this process at pH values 5.5, 6, 6.5, 7, 7.5, 8 and 8.5. No flow is needed at this point.
- Import pictures to Slidebook and analyze as described above to obtain the 495/450 ratio for each calibration pH value.
- To convert the ratios to pH, use SigmaPlot to obtain an equation that best describes the data. The calibration data should fit a sigmoidal plot. Use the equation that describes the sigmoidal plot to convert the experimental ratios obtained above to pH values (Figure 2).
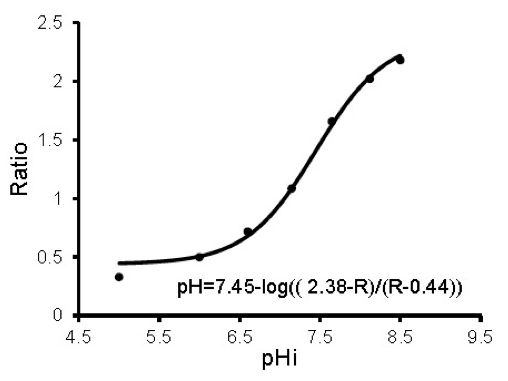
Figure 2. A typical sigmoidal calibration curve and the formula describing it are shown
Recipes
- Calibration solution
135 mM KCl
2 mM K2HPO2
20 mM HEPES
1.2 mM CaCl2
0.8 mM MgSO4
adjust pH to 5.5, 6, 6.5, 7, 7.5, 8, 8.5 by adding HCl or KOH - 10x EBSS stock (without glucose and without sodium bicarbonate)
CaCl2.2H2O 2.65 g/L MgSO4 (anhydrous) 0.9767 g/L KCl 4 g/L NaCl 68 g/L NaH2PO4 0.122 g/L - Loading solutionNotes:
EBSS 1x (from 10x stock) Glucose 1 g/L (from 100 g/L stock) NaHCO3 24 mM (for pH 7.4) - The amount of NaHCO3 needed is determined by the desired pH of the loading solution.
- Adjust the osmolarity of the solution to 290-310 mOsm by adding gluconate or sorbitol.
- Prepare enough solution to have about 25 ml per plate. Filter the solution and aliquot into 50 ml conical tubes. Place the tubes with their caps slightly open in a 5% CO2 incubator overnight for pH equilibration.
- The amount of NaHCO3 needed is determined by the desired pH of the loading solution.
- Cell culture medium
Note: Hela cells were cultured in 10 cm dishes using the following cell culture medium.
Cellgro DMEM
10% FBS
1% Antibiotic-Antimycotic
5% CO2, 37 °C incubator
Acknowledgments
This protocol has been modified from several previous publications as described in the references section, and recently used in McBrian et al. (2013).
References
- Casey, J. R., Grinstein, S. and Orlowski, J. (2010). Sensors and regulators of intracellular pH. Nat Rev Mol Cell Biol 11(1): 50-61.
- Kurkdjian, A. and Guern, J. (1989). Intracellular pH: measurement and importance in cell activity. Annu Rev Plant Biol 40(1): 271-303.
- McBrian, M. A., Behbahan, I. S., Ferrari, R., Su, T., Huang, T. W., Li, K., Hong, C. S., Christofk, H. R., Vogelauer, M., Seligson D. B. and Kurdistani, S. K. (2013). Histone acetylation regulates intracellular pH. Mol Cell 49(2): 310-321.
- Orij, R., Urbanus, M. L., Vizeacoumar, F. J., Giaever, G., Boone, C., Nislow, C., Brul, S. and Smits, G. J. (2012). Genome-wide analysis of intracellular pH reveals quantitative control of cell division rate by pH(c) in Saccharomyces cerevisiae. Genome Biol 13(9): R80.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Behbahan, I. S., McBrian, M. A. and Kurdistani, S. K. (2014). A Protocol for Measurement of Intracellular pH. Bio-protocol 4(2): e1027. DOI: 10.21769/BioProtoc.1027.
Category
Biochemistry > Other compound > Ion
Cell Biology > Cell-based analysis > Ion analysis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link












