- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Grafting of Potato Plants
Published: Vol 2, Iss 23, Dec 5, 2012 DOI: 10.21769/BioProtoc.303 Views: 12203

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Live Imaging of the Shoot Apical Meristem of Intact, Soil-Grown, Flowering Arabidopsis Plants
Gabriele Bradamante
Jun 20, 2024 2271 Views

Micrografting Technique of Hevea brasiliensis In Vitro Plantlets
Florence Dessailly [...] Julie Leclercq
Feb 20, 2025 1479 Views

Enzymatic Starch Quantification in Developing Flower Primordia of Sweet Cherry
Nestor Santolaria [...] Afif Hedhly
Apr 5, 2025 1885 Views
Abstract
Grafting is a very useful technique for testing the transmission of long-distance signals in plants and is used in agriculture and gardening for different purposes. This protocol, based on a previously published one (Jackson et al., 1998), describes a grafting method for potato plants, which has successfully been used to test the transmission of tuber-inducing signals (Martin et al., 2009; González-Schain et al., 2012). We describe first the procedures for plant growth and then the grafting procedure. Although this method has been used for grafting plants that were initially grown in vitro, it should work as well with plants grown from tubers in soil. This protocol probably works well for other plant species, with small modifications.
Keywords: GraftingMaterials and Reagents
- Murashige & Skoog medium including vitamins (Duchefa Biochemie, catalog number: M0222 )
- Sucrose
- MES (Sigma-Aldrich, catalog number: M8250 )
- ddH2O
- KOH
- GelriteTM (Duchefa Biochemie, catalog number: G1101.5000 )
- Soil composed of blocking compost (Plantaflor® Presstopfsubstrat) and sand in a 3:1 proportion
- KNO3
- NH4NO3
- K2HPO4
- KH2PO4
- Ca(NO3)2.4H2O
- MgSO42.7H2O
- FeSO42.7H2O
- Kelamix
- Fertilization solution: modified Hoagland’s solution (Johnson et al., 1957) diluted 1/60 (see Recipes)
- 2MS medium (see Recipes)
Equipment
- Plant growth chamber
- Laminar flow hood
- Sterile forceps
- Glass jars, glass tubes or plastic containers suitable for in vitro growth
Note: Equipments 1-4 is not required if plants are grown from tubers in soil.
- Sterile scalpels
- Pots
- Stakes
- Plant twist ties (e.g. Garden Odyssey, catalog number: T001 )
- Beaker
- Paper surgical tape (e.g. 3 M Micropore medical tape, catalog number: 1530-0 )
- Transparent plastic bags
- Adhesive tape
Procedure
- Micropropagate in vitro grown plants under sterile conditions in a flow hood, according to steps 2-4.
- Carefully hold the plant stem with sterile forceps and use a sterile scalpel to cut a single-node stem piece.
Note: The forceps and scalpel can be surface sterilized by dipping them in ethanol and passing them through a flame.
- Put the cutting in fresh 2MS medium.
- Repeat steps 2 and 3 as many times as required.
Note: It is advisable to propagate 10% more cuttings than the number of plants required for grafting. This number will depend on the purpose of the grafting. For our tuberization experiments, in order to have at least 10 successful grafted plants per type, we made between 12 and 20 grafts. For example, to test the transmission of signals between 2 genotypes A and B, the following grafts will be required: A/B, B/A, A/A (as control) and B/B (as control), as well as intact A and B plants. To make 15 grafts of each type and leave 15 intact controls, you will need 45 A and 45 B plants. Then, propagate 50 cuttings per genotype.
- Grow plants for approximately two weeks at 23 °C under long day conditions (LD: 16 hours light and 8 h darkness), until the roots are well developed.
- Pull the plants out of the medium, taking care not to damage the stem or the roots.
- Wash the roots by dipping into water and carefully remove the remaining medium.
- Transfer the plants to soil, previously watered with fertilization solution.
- Cover the plants with a plant propagator cover, an inverted transparent plastic bag or cling film to maintain the humidity high.
- Grow the plants under appropriate conditions, watering them as needed with fertilization solution during the whole procedure.
Note: We usually grow the plants at 23 °C under LD conditions in the greenhouse. Plants grown under LDs are more vigorous than plants grown under short days, at least for Solanum tuberosum ssp. andigena.
- After 3 days, allow the humidity to slowly decrease for 3-4 more days to gradually acclimate the plants from high humidity (in vitro) to low humidity (greenhouse). If you are using a propagator, open first one vent, one day later open another vent and 2-3 days later remove the cover. If you are using a plastic bag or cling film, make small holes (approximately 0.5 cm diameter, 40 holes/m2), gradually increase the size of the holes during the next 2-3 days and finally remove the cover.
- Tie the plants to stakes as needed. Upright plants are grafted more easily than sprawling or bending plants.
- Grow the plants for at least 3 weeks.
Note: We have successfully grafted 3-5 week old plants, but plants of other ages could also be grafted. Grafting younger plants is more difficult as the stem is weaker and is more easily damaged during the grafting procedure.
- Start with the plant that will be used as scion. Use a scalpel to cut the stem in a V shape below the third or fourth leaf from the apex.
- Put the scion in water while the plant that will be used as stock is cut.
- Use a scalpel to cut the stem of the stock plant in a V shape below the third or fourth leaf from the apex.
Note: Grafting works best when the diameters of the scion and stock are similar.
- Add a drop of water to the top of the cut stock.
Note: Water prevents the stock and scion from drying out.
- Slightly trim the end of the scion stem, maintaining the V-shape, immediately before inserting the scion into the stock.

- Remove the water from the cut stock with a tissue paper, but do not dry it completely. Leave just enough water to keep the slit of the stock moist.
- Immediately insert the scion into the top of the stock. If the scion and the stock do not fit well, slightly trim one of them to improve the fitting.
- Wrap the graft junction with paper surgical tape to keep the scion in place.
- Cover the scion with an inverted transparent plastic bag slightly bigger than the scion and use adhesive tape to close the bag around the stem, without tightening, below the graft junction. This maintains a high humidity, which helps the graft to succeed. Alternatively, use a big plastic bag to cover all the plants or groups of plants.
Note: Try to keep the bag loose enough to prevent it from clinging to the plant leaves. Holding a side of the bag to the stake using adhesive tape can help to keep the bag in place. Avoid moving the grafted plants to prevent the scion from detaching from the stock.
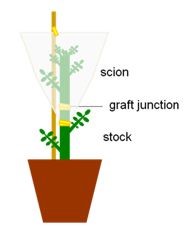
- Grow the plants for 2 days in the LD greenhouse or under the same conditions used from step 10. Avoid direct sunlight. During this time - and until the graft heals - check the plants daily. If axillary branches grow in the stock, remove them.
- Cut off a corner of the plastic bag to allow the humidity to slowly decrease.
- One day later, cut off a second corner of the bag.
- Two days later, remove the plastic bag.
- Wait a few more days until the graft heals.
Note: It can take up to 7-10 days for the graft to heal. When the grafting is successful, the scion looks healthy and vigorous and resumes growth. Otherwise, scions wilt quickly.
- Your grafted plants are ready.
Note: Grafting is easy and works very well in potato plants, with efficiencies as high as 90-100%. However, it is a good idea to practice before starting to graft your valuable plants. Experienced grafters usually obtain higher efficiencies than inexperienced ones.
Recipes
- 2MS medium (1 L)
4.40 g Murashige & Skoog medium including vitamins
20 g Sucrose
0.5 g MES
Add ddH2O to approximately 800 ml
Adjust pH to 5.8 with 1 N KOH
Add ddH2O to final volume
Add 2 g GelriteTM
Autoclave
Pour into appropriate sterile containers before the medium gelifies.
- Fertilization solution (1 L)
0.849 g KNO3
0.096 g NH4NO3
0.209 g K2HPO4
0.490 g KH2PO4
0.590 g Ca(NO3)22.4H2O
0.173 g MgSO42.7H2O
0.167 g SO4Fe2.7H2O
0.035 g Kelamix
0.400 g microelements Extra AZ (Intraplex Agrichem)
Dissolve in 800 ml dH2O
Adjust volume to 1 L
Acknowledgments
This protocol was adapted from Jackson et al. (1998) and Martin et al. (2009) and was optimized in our laboratory by Nahuel D. González-Schain. This work was funded by the Spanish Ministry of Education and Science (grants BIO2002-00933 and BIO2005-00717, co-financed by the European Regional Development Fund) and the Xarxa de Referència en Biotecnologia of the Generalitat de Catalunya. P.S.-L. was supported by the Ramón y Cajal and I3 programs of the Spanish Ministry of Education and Science.
References
- Gonzalez-Schain, N. D., Diaz-Mendoza, M., Zurczak, M. and Suarez-Lopez, P. (2012). Potato CONSTANS is involved in photoperiodic tuberization in a graft-transmissible manner. Plant J 70(4): 678-690.
- Jackson, S. D., James, P., Prat, S. and Thomas, B. (1998). Phytochrome B affects the levels of a graft-transmissible signal involved in tuberization. Plant Physiol 117(1): 29-32.
- Johnson, C. M., Stout, P. R., Broyer, T. C., Carlton, A. B. (1957). Comparative chlorine requirements of different plant species. Plant Soil 8(4): 337-353.
- Martin, A., Adam, H., Diaz-Mendoza, M., Zurczak, M., Gonzalez-Schain, N. D. and Suarez-Lopez, P. (2009). Graft-transmissible induction of potato tuberization by the microRNA miR172. Development 136(17): 2873-2881.
Article Information
Copyright
© 2012 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Suárez-López, P. (2012). Grafting of Potato Plants. Bio-protocol 2(23): e303. DOI: 10.21769/BioProtoc.303.
Category
Plant Science > Plant physiology > Plant growth
Plant Science > Plant developmental biology > General
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









