- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Bioluminescence Resonance Energy Transfer (BRET) Assay for Determination of Molecular Interactions in Living Cells
Published: Vol 7, Iss 22, Nov 20, 2017 DOI: 10.21769/BioProtoc.2904 Views: 22573
Reviewed by: Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Determination of Dissociation Constants for the Interaction of Myosin-5a with its Cargo Protein Using Microscale Thermophoresis (MST)
Rui Zhou [...] Xiang-Dong Li
Feb 5, 2025 1738 Views
Cell-Sonar, an Easy and Low-cost Method to Track a Target Protein by Expression Changes of Specific Protein Markers
Sabrina Brockmöller [...] Simone Rothmiller
Feb 5, 2025 1657 Views
SiMPull-POP: Quantification of Membrane Protein Assembly via Single Molecule Photobleaching
Ryan J. Schuck [...] Rajan Lamichhane
Jan 5, 2026 287 Views
Abstract
The bioluminescence resonance energy transfer (BRET) assay can be used as an indicator of molecular approximation and/or interaction. A significant resonance energy transfer signal is generated when the acceptor, having the appropriate spectral overlap with the donor emission, is approximated with the donor. In the example provided, proteins tagged with bioluminescent Renilla luciferase (Rlu) as donor and yellow fluorescent protein (YFP) as acceptor were co-expressed in cells. This pair of donor and acceptor have an approximate Förster distance of 4.4 nm, providing the optimal working distance (Dacres et al., 2010). This technique can be used to explore the time-course of specific molecular interactions that occur in living cells.
Keywords: BRET assayBackground
Bioluminescence resonance energy transfer (BRET) studies, using a bioluminescence donor and a fluorescence acceptor, can monitor molecular interactions (such as between labeled proteins, peptides, or small molecules) occurring in real time in living cells. This approach is dependent on spatial approximation between the donor and acceptor, as well as appropriate spectral overlap to yield a meaningful signal (Figure 1). The example currently provided utilizes a Rlu-tagged protein as the donor and a YFP-tagged protein as acceptor (Harikumar et al., 2007). This has been very successfully applied to establish the presence of physiologically-relevant protein-protein interactions in the plasma membrane of living cells. It is important, however, to include controls for levels of expression that could cause non-specific protein-protein approximation and energy transfer (bystander effect), such as the use of similar levels of expression of a known non-associated protein. Also, competition with an unlabeled protein can help to establish the saturability of the interaction and the specificity of the signal. 
Figure 1. Illustration of relevant events in a BRET experiment in a living cell. The energy transfer reaction is initiated by adding the luciferase substrate, coelenterazine-h, to cells expressing both molecules tagged with Rlu (donor) and with yellow fluorescent protein (acceptor). The Rlu emits light with a wavelength of approximately 475 nm that then excites the YFP to emit light at approximately 525 nm that can be quantified to represent the BRET signal. The approximate Fӧrster distance for this pair of donor-acceptor is approximately 4.4 nm.
Materials and Reagents
- Pipette tips (USA Scientific, catalog number: 1111-1700 )
- 100-mm tissue culture grade plastic plates (SARSTEDT, catalog number: 83.3902 )
- 5 ml tissue culture tubes (Corning, Falcon®, catalog number: 352052 )
- Cell culture flasks (Corning, catalog number: 3056 )
- 96-well OptiPlates (PerkinElmer, catalog number: 6005290 )
- 15 ml conical tube (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 339650 )
- Pasteur pipette (Fisher Scientific, catalog number: 13-678-20B )
- Rapid flow bottle-top filter unit with polyethersulfone (PES) membrane (Thermo Fisher Scientific, Thermo ScientificTM, catalog number: 595-3320 )
- PS1/PS2-deleted HTL cells
Note: This cell line was derived by Xu et al., 2016, utilizing CRISPR/Cas9 to delete PS1/PS2 from HTL cells, representing a cell line derived from HEK293 cells by Barnea and Axel who stably integrated a luciferase reporter under the control of the bacterial operator element tetO. These cells were used in this case to correlate with specific functional assays requiring a specialized cell type. In a general protocol, any transfectable cell type is fine.
- Dulbecco’s modified Eagle’s medium powder (DMEM) (Thermo Fisher Scientific, GibcoTM, catalog number: 12100-038 ) (see Recipe 6 for media preparation)
- Fetal Clone II supplement, a bovine serum product (GE Healthcare, HycloneTM, catalog number: SH30066.03 )
- Trypsin 0.25%-EDTA (Thermo Fisher Scientific, GibcoTM, catalog number: 25200056 ) diluted to 0.05% with 1x Dulbecco’s phosphate buffered saline, pH 6.80
- DEAE-dextran hydrochloride (Sigma-Aldrich, catalog number: D9885 )
- Dimethyl sulfoxide (DMSO) (Fisher Biotech, catalog number: BP231-1 )
- Chloroquine diphosphate salt (Sigma-Aldrich, catalog number: C6628 )
- Enzyme-free cell dissociation solution (EMD Millipore, catalog number: S-014-C )
- Sodium phosphate dibasic (Na2HPO4) (Sigma-Aldrich, catalog number: S0876 )
- Potassium phosphate monobasic (KH2PO4) (Fisher Scientific, catalog number: P285 )
- Sodium chloride (NaCl) (Fisher Scientific, catalog number: S671-500 )
- Potassium chloride (KCl) (Sigma-Aldrich, catalog number: P9541 )
- HEPES (Research Products International, catalog number: H75030-1000 )
- Magnesium sulfate heptahydrate (MgSO4·7H2O) (Sigma-Aldrich, catalog number: M1880 )
- Calcium chloride dihydrate (CaCl2·2H2O) (Fisher Scientific, catalog number: C79-500 )
- Sodium hydroxide (NaOH) (VWR, catalog number: BDH7247-1 )
- Coelenterazine-h (AAT Bioquest, catalog number: 21165 ) (see Recipe 5 for preparation of stock solution)
- Methanol (Honeywell International, catalog number: 24229 )
- Penicillin-streptomycin (Thermo Fisher Scientific, GibcoTM, catalog number: 15140122 )
- Sodium bicarbonate (Fisher Scientific, catalog number: BP328-500 )
- Hydrochloric acid (HCl) (Fisher Scientific, catalog number: A144-212 )
- 10x phosphate-buffered saline (PBS buffer) (see Recipes)
- 10x Kreb’s-Ringers-HEPES stock solution (10x KRH) (see Recipes)
- 50x CaCl2 stock solution (see Recipes)
- 1x KRH working solution (see Recipes)
- Coelenterazine-h solution (see Recipes)
- 1x DMEM tissue culture medium (see Recipes)
Equipment
- 37 °C, 5% CO2 incubator (Thermo Fisher Scientific, Thermo ScientificTM, model: FormaTM Series II 3110 Water-Jacketed)
- Cell culture microscope (Fisher Scientific)
- Micropipettes (Gilson)
- 2103 EnVision Plate Reader (PerkinElmer, model: 2103 EnVisionTM )
- Centrifuge (Thermo Fisher Scientific, Thermo ScientificTM, model: SorvallTM LegendTM XT/XF centrifuge , catalog number : 75216362)
- 500 ml screw-cap autoclaved glass bottles (WHEATON, catalog number: 219759 )
Software
- GraphPad Prism 6 (GraphPad Software, Inc. USA)
Procedure
- Cell culture
- The PS1/PS2-deleted HTL cells are routinely grown in DMEM medium supplemented with 10% (v/v) Fetal Clone II and including 1x penicillin-streptomycin (see Recipes) at 37 °C under a humidified 5% CO2 atmosphere.
- Treat the cells with 0.05% trypsin-EDTA for 5 min at 37 °C in an incubator to lift the cells. Wash the cells with medium, and count them to allow a plating density of 1.5 x 106 cells per 100 mm dish one day prior to transfection.
- In preparation for transfection, visualize the confluence of the cells and their health using a cell culture microscope with phase optics.
- The PS1/PS2-deleted HTL cells are routinely grown in DMEM medium supplemented with 10% (v/v) Fetal Clone II and including 1x penicillin-streptomycin (see Recipes) at 37 °C under a humidified 5% CO2 atmosphere.
- Transfection
- To a sterile 5 ml tissue culture tube, add 1.5 ml DMEM medium without serum.
- Add 75 µl DEAE-dextran solution and mix well.
- Add 1.0 µg of total DNA (0.5 µg of donor + 0.5 µg of acceptor prepared using standard DNA isolation techniques) and mix well.
- Use a micropipette to transfer 1.575 µl of DNA-DEAE-dextran complex to each plate.
- Incubate at 37 °C in a tissue culture incubator for 2 h without shaking.
- Aspirate the medium from each plate, add 4 ml of DMEM containing 10% DMSO and incubate at room temperature for 2 min without shaking.
- Aspirate the medium from each plate, add 4 ml of DMEM containing 0.1 µM chloroquine and incubate for 2 h at 37 °C without shaking.
- Aspirate the medium from each plate, and add 8 ml of serum-containing complete DMEM medium (see Recipes).
- Incubate the cells in the incubator at 37 °C in a humidified atmosphere containing 5% CO2 for 48 h.
- To a sterile 5 ml tissue culture tube, add 1.5 ml DMEM medium without serum.
- Luminescence and fluorescence measurement
- 48 h post-transfection, remove the medium from the cultured cells using a Pasteur pipette attached to a vacuum source and gently add 5 ml of PBS (see Recipes) to each plate to rinse the cells.
- Dispense 1 ml of enzyme-free cell dissociation solution into each plate and incubate without shaking for 5 min at 37 °C in tissue culture incubator to release the cells.
- Transfer the cells into a 15 ml conical tube and centrifuge the cells at 175 x g for 5 min at room temperature.
- Resuspend the cell pellet in approximately 1 ml of Kreb’s-Ringers-HEPES medium, pH 7.4 (see Recipes) to achieve a density of 0.20 x 106 cells per ml. Keep this tube at room temperature in preparation for the assay.
- Dispense 100 µl of cell suspension (approximately 20,000 cells) into a white 96-well OptiPlate, add 5 µl coelenterazine h (see Recipes), mix the cells by swirling the plate, and measure both the luminescence signal and the YFP fluorescence signal using an Envision Plate Reader with a BRET module.

- Luminescence and fluorescence signals should be collected immediately after the addition of coelenterazine h solution. The spectrofluorometer instrument should be set up for the specific pair of donor and acceptor chosen. Here, for the proposed BRET studies, luminescence emission should be collected using a dedicated filter set (460 nm, with bandwidth 25 nm) and the fluorescence emission should be collected simultaneously using a dedicated filter set (535 nm, bandwidth 25 nm). The measurement time is 0.2 sec and measurement height is 6.5 mm. (Harikumar et al., 2007). In the example, YFP fluorescence was measured by exciting the sample at 475 nm (bandwidth 25 nm) and the emission was collected at 535 nm (bandwidth 25 nm), with detector gain 150 and number of flashes 20.
- A representative example of data collection and analysis follows:
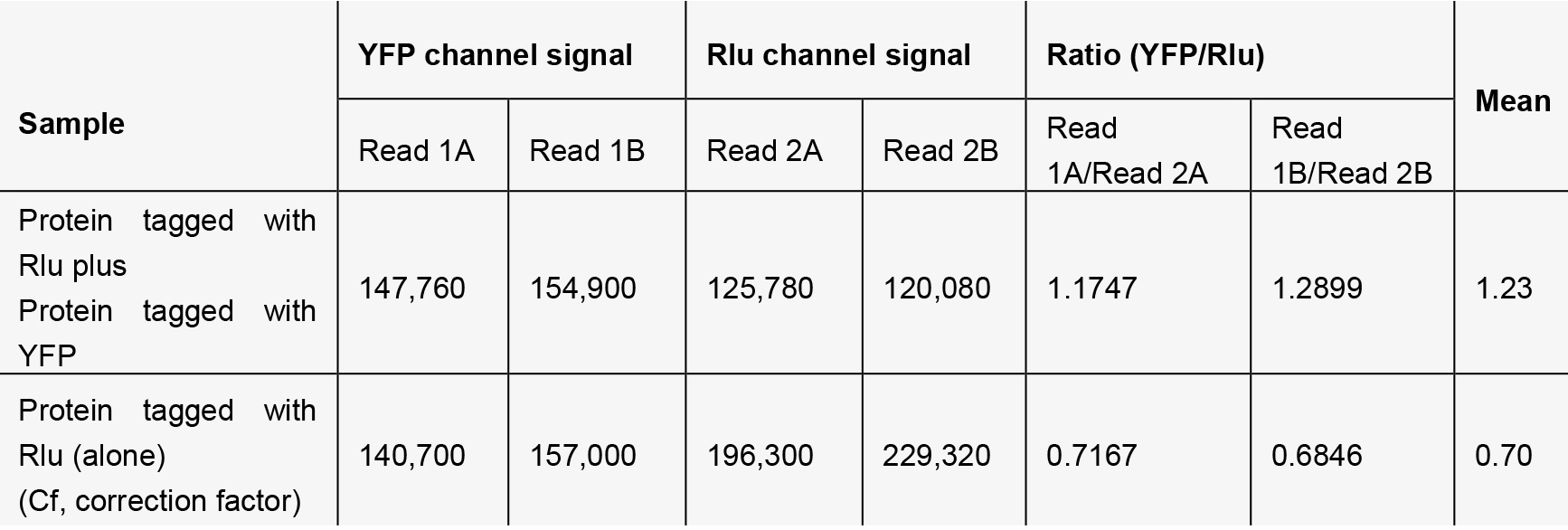
Calculated BRET ratio: 1.23 - 0.70 = 0.53
- 48 h post-transfection, remove the medium from the cultured cells using a Pasteur pipette attached to a vacuum source and gently add 5 ml of PBS (see Recipes) to each plate to rinse the cells.
Data analysis
Data should be collected in at least duplicate conditions in a minimum of three independent experiments. The statistical analysis of data can be accomplished with GraphPad Prism 6, using a t-test with the Mann-Whitney post-test.
Notes
Controls for levels of expression of donor and acceptor are important to eliminate bystander energy transfer. A significant BRET signal should be observed at levels of expression that exist physiologically. Additionally, saturability and specificity of the BRET signal are important, and can be established using competing non-labeled protein of interest. For positive control, a construct encoding a fusion protein containing both luciferase and yellow fluorescent protein (Rlu-YFP) was used to transfect the cells, and the BRET signal was measured.
Recipes
- 10x phosphate-buffered saline (10x PBS buffer) (1 L)
11.5 g Na2HPO4
2 g KH2PO4
80 g NaCl
2 g KCl
Dissolve in 1 L of sterile, deionized water
Adjust the pH of 1x PBS to 7.4
Store at 4 °C
- 10x Kreb’s-Ringers-HEPES stock solution (10x KRH) (1 L)
59.58 g HEPES
60.74 g NaCl
3.72 g KCl
1.36 g KH2PO4
2.96 g MgSO4
Dissolve the components in deionized water and bring the volume to 1 L, do not adjust the pH
Store at 4 °C
- 50x CaCl2 stock solution (100 mM) (100 ml)
1.47 g CaCl2
Bring the volume to 100 ml with deionized water; do not adjust pH
Store at 4 °C
- 1x KRH working solution (100 ml)
10 ml 10x KRH stock solution
2 ml 50x CaCl2 stock solution
Bring the volume to 100 ml with deionized water; adjust the pH to 7.4 with NaOH
- Coelenterazine-h solution
0.5 mg coelenterazine-h powder
Dissolve in 12.5 ml of 5% methanol/95% water
Store at -20 °C
- 1x DMEM tissue culture medium (1 L)
- One 1-L packet of Dulbecco’s modified Eagle medium (13.4 g) and 3.7 g of sodium bicarbonate are added to 1 L of deionized water
- Adjust the pH to 6.90 with HCl, filter into 500 ml screw-cap autoclaved glass bottles using a 0.2 µm rapid flow bottle-top filter unit containing a polyethersulfone (PES) membrane
- Store at 4 °C
- One 1-L packet of Dulbecco’s modified Eagle medium (13.4 g) and 3.7 g of sodium bicarbonate are added to 1 L of deionized water
Acknowledgments
This work was supported by Mayo Clinic (L.J.M.), the Van Andel Research Institute, the National Natural Science Foundation of China (31300607, 31300245 and 91217311), Ministry of Science and Technology grants 2012ZX09301001, 2012CB910403, and 2013CB910600, XDB08020303, 2013ZX09507001, Shanghai Science and Technology Committee (13ZR1447600), Shanghai Rising-Star Program (14QA1404300), and the National Institute of Health grants DK071662 (H.E.X.), GM102545 and GM104212 (K. M.). Authors have no conflicts of interest or competing interests.
References
- Dacres, H., Wang, J., Dumancic, M. M. and Trowell (2010). Experimental determination of the Förster distance for two commonly used Bioluminescence resonance energy transfer pairs. Anal Chem 82: 432-435.
- Harikumar, K. G., Pinon, D. I. and Miller, L. J. (2007). Transmembrane segment IV contributes a functionally important interface for oligomerization of the Class II G protein-coupled secretin receptor. J Biol Chem 282: 30363-30372.
- Xu, T. H., Yan, Y., Kang, Y., Jiang, Y., Melcher, K. and Xu, H. E. (2016). Alzheimer's disease-associated mutations increase amyloid precursor protein resistance to γ-secretase cleavage and the Aβ42/Aβ40 ratio. Cell Discov 2: 16026.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Harikumar, K. G., Yan, Y., Xu, T., Melcher, K., Xu, H. E. and Miller, L. J. (2017). Bioluminescence Resonance Energy Transfer (BRET) Assay for Determination of Molecular Interactions in Living Cells. Bio-protocol 7(22): e2904. DOI: 10.21769/BioProtoc.2904.
- Yan, Y., Xu, T. H., Harikumar, K. G., Miller, L. J., Melcher, K. and Xu, H. E. (2017a). Dimerization of the transmembrane domain of amyloid precursor protein is determined by residues around the gamma-secretase cleavage sites. J Biol Chem, 292: 15826-15837.
Category
Biochemistry > Protein > Interaction > Protein-protein interaction
Molecular Biology > Protein > Protein-protein interaction
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











