- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Peptide Synthesis
Published: Vol 2, Iss 14, Jul 20, 2012 DOI: 10.21769/BioProtoc.233 Views: 29164

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
In vitro Assay for Bacterial Membrane Protein Integration into Proteoliposomes
Hanako Nishikawa [...] Ken-ichi Nishiyama
May 20, 2020 5668 Views
A Method for User-defined Mutagenesis by Integrating Oligo Pool Synthesis Technology with Nicking Mutagenesis
Paul J. Steiner [...] Timothy A. Whitehead
Aug 5, 2020 7014 Views

Artificial Metalloenzymes in Artificial Sanctuaries Through Liquid–Liquid Phase Separation
Kaixin Wang [...] Tong Wu
Oct 5, 2025 1566 Views
Abstract
This protocol describes the synthesis of peptides for affinity testing and bioconjugate with solid phase peptide synthesizer at a small scale.
Keywords: Solid phaseMaterials and Reagents
- Dichloromethane (DCM)
- Dimetylformamide (DMF) (Fisher Science, catalog number: D119-4 )
- Trifluoroacetic acid (TFA) (ACRO, catalog number: 13972 )
- Thioanisole (TIS) (Sigma-Aldrich, 233781 )
- Ethanedithiol (EDT) (TCI, catalog number: E0032 )
- 2-(1H-Benzotriazol-1-yl)-1,1,3,3-tertramethyluronumhexafluorophosphate (HBTU) (Anaspec, catalog number: AS-21001 )
- N,N-diisopropylethylamine (DIEA) (Santa Cruz Biotech, catalog number: sc-215490 )
- Piperidine (biotech. grade, ≥99.5%) (Sigma-Aldrich, catalog number: 571261 )
- Dithiothriotol (DTT)
- Pyridine
- Potassium cyanide
- Ninhydrin
- Fmoc protected amino acids:
Fmoc-Ile-OH, Fmoc-Glu (OtBu)-OH, Fmoc-Met-OH, Fmoc-Leu-OH, Fmoc-Lys (Boc)-OH, Fmoc-Arg (Pbf)-OH, Fmoc-Asn (Trt)-OH, Fmoc-Ser (tBu)-OH, Fmoc-Ala-OH, Fmoc-His (Trt)-OH, Fmoc-Gln (Trt)-OH, Fmoc-Val-OH, Fmoc-Phe-OH, Fmoc-Thr(tBu)-OH, Fmoc-Pro-OH and Fmoc-Asp (OtBu)-OH, Fmoc-Gly-OH, Fmoc-Cys(Trt)-OH, Fmoc-Tyr(tBu)-OH, Fmoc-Trp(Boc)-OH. - Rink Amide Resin (0.66 mmol/g) (AAPPTec, catalog number: RRZ001 or equivalent)
- C-18 column (Vydac, catalog number: 218TP510 or equivalent)
All reagents are ACS grade or synthesis grade - Anhydrous diethyl ether
Equipment
- Peptide synthesizer CS-Bio 336x or equivalent
- Analytical balance
- Swing bucket eppendorf centrifuge or equivalent
- High performance liquid chromatograph (HPLC)
- Reaction vessel (SKU: PS-G002 )
- Liquid chromatograph-mass spectrometer
Procedure
- Resin preparation
- Weigh 303 mg Rink-Amide resin (0.20 mmol equivalent) into a reaction vessel with filters and connect the up and down outlets to the solvent line and the waste line respectively.
- Add 8.0 ml DMF to the resin (washing step), stir gently for 5 min and then remove the solvent via nitrogen pressure (4- 8 psi). Repeat 3 times.
- Weigh 303 mg Rink-Amide resin (0.20 mmol equivalent) into a reaction vessel with filters and connect the up and down outlets to the solvent line and the waste line respectively.
- Deprotection/coupling cycle
- Add 8.0 ml of 20% piperidine/DMF (vol/vol), stir gently for 1 min (first Fmoc removal step), and then remove the solvent by nitrogen pressure.
- Add 8.0 ml of 20% piperidine/DMF (vol/vol), stir gently for 10 min (second Fmoc removal step), and then remove the solvent by nitrogen pressure.
- Add 8.0 ml DMF (washing step 1), shake for 30 sec, and then remove the solvent by nitrogen pressure.
- Add 8.0 ml DMF (washing step 2), shake for 5 min, and remove the solvent by nitrogen pressure.
- Repeat Step 4 four times.
- Dissolve 1.0 mmol Fmoc protected amino acid and HBTU (341.37 mg, 0.9 mmol) in 8.0 ml DMF. Mix well by bubbling nitrogen for 30 sec.
- Add 2.0 mmol DIEA (1 M, 2 ml) and bubbling nitrogen for 30 sec and wait for 5 min. Add the mixture to resin. Shake for 1 h. and then remove the solvent by nitrogen pressure filtration.
- (CRITICAL STEP: Intermittent stirring is preferred over continuous stirring when doing manual synthesis because of the fragility of some resin, in order to minimize the fragmentation of the resin in small-sized particles which can lead to difficulties during filtration. As an alternative, continuous gentle shaking or nitrogen bubbling through the reaction vessel can be applied).
- Repeat steps 6 and 7 for double coupling (optional).
- Add 8.0 ml DMF (washing step), stir for 30 sec and remove the solvent by nitrogen pressure filtration.
- Repeat Step 10 three times.
- Repeat the cycle starting from step 3 going to step 12 for each of the subsequent amino acids according to the peptide sequence using the above Fmoc-amino acid derivatives.
CRITICAL STEP: The progress of the synthesis can be followed at every cycle by using microcleavage test (see below). To speed up the assembly, single couplings can be performed instead of double couplings).
The requirement for the repetition of the coupling step can be derived from the result of the Kaiser test (see below).
PAUSE POINT: the synthesis can, in principle, be interrupted at the end of every coupling cycle, when the N-terminal amino group is protected. To avoid undesired removal of the Fmoc group during storage in DMF, wash the peptide-resin five times with DCM and let it dry at room temperature (18-22 °C). Close the syringe with its plunger and cap, and store at 4 °C. Before resuming the synthesis, let the sample reach room temperature, and swell the dry resin as described in step 2.
- Add 8.0 ml of 20% piperidine/DMF (vol/vol), stir gently for 1 min (first Fmoc removal step), and then remove the solvent by nitrogen pressure.
- Removal of the N-terminal Fmoc-group and drying of the peptide resin
- Add 8.0 ml of 20% piperidine/DMF (vol/vol), stir gently for 1 min (first Fmoc removal step), and then remove the solvent by nitrogen pressure filtration.
- Add 8.0 ml of 20% piperidine/DMF (vol/vol), stir gently for 10 min (second Fmoc removal step), and then remove the solvent by nitrogen pressure filtration.
- Add 8.0 ml DMF (washing Step 1), stir for 30 sec, remove the solvent by vacuum filtration.
- Add 8.0 ml DMF (washing step 2), stir for 5 min, remove the solvent by nitrogen pressure filtration.
- Repeat step 5 four times with DMF.
- Wash the resin four times with DCM in a glass filter.
- Air-dry the peptide resin.
- Add 8.0 ml of 20% piperidine/DMF (vol/vol), stir gently for 1 min (first Fmoc removal step), and then remove the solvent by nitrogen pressure filtration.
- Peptide cleavage and purification (manually)
- Peptide cleavage and deprotection were carried out by a 3 h incubation in a mixture of TFA/TIS/EDT/H2O (94:2:2:2).
- The mixture was filtered, and the peptide in solution was precipitated with ice cold anhydrous diethyl ether and centrifuged in eppendorf swing bucket at speed of 1,000 rcf. The resulting peptide was washed four times with ice-cold anhydrous diethyl ether, dried, and dissolved in suitable buffer or solvent (add 1 mM DTT if there is a cysteine).
- The peptide was purified by RP-HPLC on a C-18 column. Fractions were collected and lyophilized. The target product was characterized by ESI-MS and was ready for use in the next step in the reaction.
- Peptide cleavage and deprotection were carried out by a 3 h incubation in a mixture of TFA/TIS/EDT/H2O (94:2:2:2).
Representative data
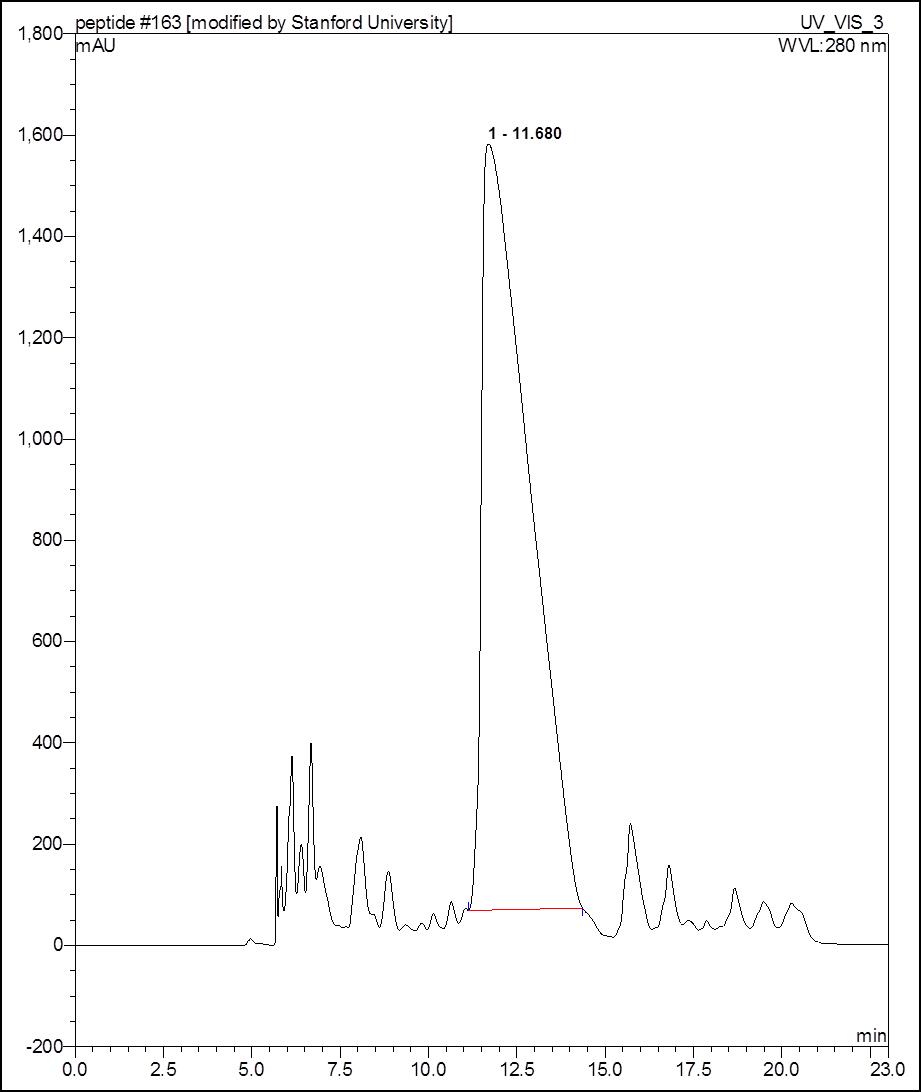
Figure 1. Representative HPLC chromatography of a peptide synthesized with this protocol. Peak at retention time of 11.68 min is the product peak.
Supplement
- Kaiser test
The Kaiser test is a qualitative test for the presence or absence of free primary amino groups, and it can be a useful indication about the completeness of a coupling step. The test is based on the reaction of ninhydrin with primary amines, which gives a characteristic dark blue color. The test requires minimal amounts of analyte and is completed within a few minutes.- Reagents
- 0.5 g ninhydrin in 10 ml ethanol (EtOH)
- 0.4 ml of 0.001 M potassium cyanide (KCN ) in 20 ml pyridine
- 0.5 g ninhydrin in 10 ml ethanol (EtOH)
- Procedure
- Transfer a few resin beads to a small glass tube or an Eppendorf tube and wash several times with ethanol.
- Add 100 ml of each of the solutions mentioned above (see reagents in this box).
- Mix well and place the tube in a preheated oven (115 °C) for 5 min. Light green or no color is negative. Dark blue or purple is positive.
Critical step to reduce the incidence of false negative results, it is recommended to carry out a parallel positive control. The test is not applicable to N-terminal Pro residues (secondary amine) and N-alkyl amino acids. The test may give false negative results when applied to aggregate sequences.
- Transfer a few resin beads to a small glass tube or an Eppendorf tube and wash several times with ethanol.
- Reagents
- Microcleavage
When Fmoc removal data show anomalies, before performing a step that requires the use of particularly expensive materials, or after a critical step, and in general when assembling longer sequences, it may be useful to cleave and analyze a small amount of intermediate product.- Procedure
- Transfer a sample containing B1-2 mg dry peptide-resin to a small syringe (2 ml).
- Add 300 ml of the cleavage cocktail (Trifluoroacetic acid/H2O/phenol/triisopropylsilane 8.5/0.5/0.5/0.5) to the dried peptide resin, stir gently for 30 s and wait for 3 h (stir gently in-between).
- Collect the solution in a small HPLC vial, dilute with 400 ml acetonitrile/water 1/1 and mix.
- At this point, the solution can be analyzed in an analytical HPLC system (inject 20 ml) and/or further diluted (1/10) to be injected (2 ml) in liquid chromatograph-mass spectrometer.
- Transfer a sample containing B1-2 mg dry peptide-resin to a small syringe (2 ml).
- Procedure
Recipes
- Amino acid reagent vial
1.0 mmol Fmoc amino acid per 0.2 mmol equivalent resin
0.9 mmol HBTU per 0.2 mmol equivalent resin - Peptide cleavage cocktail (5 ml for 200 mg ~300 mg dry peptide-resin)
4.7 ml TFA
0.1 ml EDT
0.1 ml TIS
0.1 ml water
Acknowledgments
This protocol was adapted or modified from previous work (Coin et al. (2007); Miao et al. (2011); Ren et al. (2010)). This work was supported, in part, by the California Breast Cancer Research Program 14IB-0091 and an SNM Pilot Research Grant (ZC).
References
- Coin, I., Beyermann, M. and Bienert, M. (2007). Solid-phase peptide synthesis: from standard procedures to the synthesis of difficult sequences. Nat Protoc 2(12): 3247-3256.
- Miao, Z., Ren, G., Jiang, L., Liu, H., Webster, J. M., Zhang, R., Namavari, M., Gambhir, S. S., Syud, F. and Cheng, Z. (2011). A novel 18F-labeled two-helix scaffold protein for PET imaging of HER2-positive tumor. Eur J Nucl Med Mol Imaging 38(11): 1977-1984.
- Ren, G., Liu, S., Liu, H., Miao, Z. and Cheng, Z. (2010). Radiofluorinated rhenium cyclized alpha-MSH analogues for PET imaging of melanocortin receptor 1. Bioconjug Chem 21(12): 2355-2360.
Article Information
Copyright
© 2012 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Miao, Z. and Cheng, Z. (2012). Peptide Synthesis. Bio-protocol 2(14): e233. DOI: 10.21769/BioProtoc.233.
Category
Biochemistry > Protein > Synthesis
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









