- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Time-lapse Observation of Chromosomes, Cytoskeletons and Cell Organelles during Male Meiotic Divisions in Drosophila
Published: Vol 7, Iss 8, Apr 20, 2017 DOI: 10.21769/BioProtoc.2225 Views: 10972
Reviewed by: Jihyun KimFilipa VazAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Characterization of Biological Motion Using Motion Sensing Superpixels
Felix Y. Zhou [...] Xin Lu
Sep 20, 2019 5935 Views

Quantitation of Secretory Granule Size in Drosophila Larval Salivary Glands
Cheng-I J. Ma and Julie A. Brill
Jun 5, 2021 3877 Views
Live Imaging and Analysis of Meiotic Cytokinesis in Drosophila Testes
Govind Kunduri and Jairaj K. Acharya
Jan 20, 2024 3063 Views
Abstract
In this protocol, we provide an experimental procedure that perform time-lapse observation of intra-cellular structures such as chromosomes, cytoskeletons and cell organelles during meiotic cell divisions in Drosophila males. As primary spermatocyte is the largest dividing diploid cell in Drosophila, which is equivalent in size to mammalian cultured cells, one can observe dynamics of cellular components during division of the model cells more precisely. Using this protocol, we have showed that a microtubule-associated protein plays an essential role in microtubule dynamics and initiation of cleavage furrowing through interaction between microtubules and actomyosin filaments. We have also reported that nuclear membrane components are required for a formation and/or maintenance of the spindle envelope essential for cytokinesis in the Drosophila cells.
Keywords: DrosophilaBackground
In Drosophila, good cultured cell lines that proliferate well in a standard culture condition are also available. However, their cell size, particularly cytoplasmic volume, is much smaller than that of mammalian cells. This compromises the examination of cellular components during cell division. Spermatocytes, on the other hand, achieve distinct cell growth before initiation of first meiotic division. The primary spermatocytes are the largest diploid cells among proliferative cells to appear in Drosophila development. Thus, one can easily perform detailed observation of cellular structures in dividing cells using optical microscopes. In Drosophila melanogaster, well-advanced and sophisticated genetic techniques are available (Ashburner et al., 2004). Meiotic defects in chromosome segregation and in cytokinesis appear in cellular organization of spermatids just after completion of 2nd meiotic division. By observation of such early spermatids, one can easily find out even subtle meiotic abnormalities (Giansanti et al., 2012; Inoue et al., 2012). Furthermore, if a loss of microtubule integrity or dynamics would have occurred in normal cultured cells, their cell cycle progression should be arrested before metaphase. Therefore, it is hard to examine how microtubules would influence later processes of cell divisions in the somatic cells. Spermatocytes, on the other hand, are less sensitive to microtubule abnormalities at microtubule assembly checkpoint before metaphase. One can, therefore, examine a role of microtubule-related genes in cytokinesis without arresting cell cycle. We and other groups have established systems to facilitate dynamics of chromosomes or microtubules by expression of proteins fused with GFP fluorescence tag (Clarkson and Saint, 1999; Inoue et al., 2004; Rebollo and Gonzalez, 2004; Kitazawa et al., 2012).
Previous protocols can trap the male meiotic cells in a narrow space sandwiched between a coverslip and a slide glass, ensured by a small cushion materials and observe chromosome segregation under an upright microscope (Savoian et al., 2000; Inoue et al., 2004; Savoian, 2015). These protocols allowed us to collect clear images of microtubules. However, a preparation that makes the cells flattened often prevents initiation and/or progression of cytokinesis. In addition, it was difficult to add drugs or inhibitors to the living cells while time-lapse observation.
Therefore, we have established a new method that allows us to observe a whole meiosis I from prophase I to end of cytokinesis in an open chamber under an inverted microscope. We can add drugs in the cell culture in any timing of the imaging. We also improved the protocol so that we can perform a simultaneous observation of chromosomes and other cellular components such as microtubules, actin filaments, endoplasmic reticulum, Golgi apparatuses or mitochondria during male meiosis I. It can be achieved by a simultaneous expression of proteins fused with different fluorescent tags showing spectrally separable colors. As most aspects of division process seen in Drosophila meiotic cells are shared among higher eukaryotes, this protocol should be useful for studying cell division processes of other organisms as well as Drosophila somatic cell mitosis.
Materials and Reagents
- Cover slips (22 x 22 mm, No. 1, thickness 0.12-0.17 mm) (Matsunami Glass, catalog number: C022221 )
Note: It was argued that thorough cleaning steps of cover slips are required for maintenance of cell viability and for a success prolonged observation of living cells (Savoian, 2015). However, if these cover slips are used, any pretreatment is not basically necessary except a wipe with 70% ethanol just before using. - Plastic cover slip folder (76 x 26 mm)
Note: The folders were customized. It was 76 x 32 mm in length and width and 1.7 mm in thickness. There was a concavity (25 mm square) where the cover slip fell in the surface and a circular hole of 15 mm diameter in the center of the concavity) (see Figure 1).
Figure 1. A plastic cover slip holder. The holder should be set on the microscope stage. There was a concavity where the cover slip (22 x 22 mm) fell in the surface and a circular hole with 15 mm diameter in the center of the concavity. - 10 x 10 mm open frame that had adhesives on the bottom side (Gene Frame 25 μm) (Thermo Fisher Scientific Co., Waltham, USA)
- Plastic Petri dish (90 mm diameter) (AsOne, catalog number: 1-7484-01 )
- Kim-wipe (KCWW, Kimberly-Clark)
- bam-Gal4::vp16 (abbreviated as bam-Gal4) can be used as a Gal4 driver for testis-specific ectopic expression of fluorescence proteins (Kitazawa et al., 2012)
- bam-Gal4::vp16 UAS-dir2 was used as a Gal4 driver for testis-specific depletion (Kitazawa et al., 2014)
- P{His2AvT:Avic\GFP-S65T} (abbreviated as Histone2Av-GFP) can be used for expression of Histone 2Av fused with a GFP tag to visualize chromatin in living meiotic cells (Bloomington Drosophila Stock Center, catalog number: BL5941 )
- P{Ubi-mRFP-βTub85D} (abbreviated as RFP-βTubulin) can be used for ubiquitous expression of β-tubulin fused with a mRFP tag to visualize microtubules in living meiotic cells (Kitazawa et al., 2014)
- P{sqh-EYFP-Golgi} can be used for ubiquitous expression of Golgi components fused with a YFP tag to visualize Golgi apparatus in living meiotic cells (Bloomington Drosophila Stock Center, catalog number: BL7193 )
- P{sqh-EYFP-ER} can be used for ubiquitous expression of ER components fused with a YFP tag to visualize endoplasmic reticulum in living meiotic cells (Bloomington Drosophila Stock Center, catalog number: BL7195 )
- P{sqh-EYFP-Mito} can be used for ubiquitous expression of mitochondrial target sequences fused with a YFP tag to visualize mitochondria in living meiotic cells (Bloomington Drosophila Stock Center, catalog number: BL7194 )
- P{UASp-GFP-Orbit}, P{UASp-mRFP-Orbit}, and P{UASp-Venus-Orbit} can be used for visualization of Orbit protein, an essential microtubule-associated protein. UAS-stocks that can induce Orbit proteins fused with three different fluorescent tags are available (Miyauchi et al., 2013)
- P{UASp-mRFP-Actin5C} (Bloomington Drosophila Stock Center, catalog number: BL24777 ), P{UAS-GFP-Actin5C} (Bloomington Drosophila Stock Center, catalog number: BL9257 ) and P{UAS-CFP-Actin5C} (Miyauchi et al., 2013) can be used for ectopic expression of F-actin components fused with different tags to visualize F-actin in living meiotic cells
- P{UAS-GFP-anillin} (Bloomington Drosophila Stock Center, catalog number: BL51348 ) and P{Ub-mRFP-anillin} (Bloomington Drosophila Stock Center, catalog number: BL52220 ) can be used for visualization of a contractile ring in a living meiotic cell (gifts from J.A. Brill, now available from Bloomington Drosophila Stock Center)
- P{w[+mC]=sqh-GFP.RLC}3 can be used for ubiquitous expression of myosin light chain components fused with GFP tag to visualize MLC in living meiotic cells (a gift from R. Karess)
- P{PTT-GA}Pdi[G00198], a protein trap stock expressing GFP-Protein disulfide isomerase for visualization of intracellular membranous structures (a gift from L. Cooley, now available from Bloomington Drosophila Stock Center as catalog number: BL6839 ). A protein stock expressing GFP-LamC for visualization of nuclear lamina (a gift from L. Wallrath)
- P{UAS-PLCg-PH-GFP} for visualization of plasma membrane in male meiotic cells (a gift from J. A. Brill)
- P{UAS-mRFP-Nup107.K} 7.1 for visualization of nuclear pore complex in nuclear envelope of male meiotic cells (a gift from V. Doyle, now available from Bloomington Drosophila Stock Center as catalog number: BL35516 )
- P{UAS-GFP-Pav} and P{UAS-GFP-Polo} were used for visualization of a microtubule motor and an important cell division regulator in a living meiotic cell, respectively (gifts from D. Glover)
- For RNAi experiments in male meiotic cells, UAS-RNAi stocks for ectopic expression of dsRNA for each protein were obtained from VDRC stock center and Bloomington stock center. P{UAS-GFP RNAi} (Bloomington Drosophila Stock Center, catalog number: BL9330 ) can be used as a negative control of RNAi experiments
- Colchicine (50 μg/ml in BRB80 buffer) (≥ 95% colchicine) (Sigma-Aldrich, catalog number: C9754 )
Note: To examine requirement of microtubules for cellular dynamics, colchicine that is an inhibitor of microtubule polymerization was used. The BRB80 buffer containing colchicine was prepared before the dissection every time. The testes were dissected in the buffer containing colchicine and then, meiotic cells were spread under mineral oil. - Cytochalasin D (10 μg/ml in BRB80 buffer) (≥ 98% cytochalasin D) (Sigma-Aldrich, catalog number: C8273 )
Note: To examine requirement of F-actin for cellular dynamics, cytochalasin D that is an inhibitor of actin polymerization was used. The BRB80 buffer containing cytochalasin D was prepared before the dissection every time. The male flies were dissected to collect the testes in the buffer containing cytochalasin D and then, meiotic cells were spread under mineral oil. - Fetal calf serum (Thermo Fisher Scientific, GibcoTM, catalog number: 451456 or 10437 )
Note: The fetal calf serum can be kept at 4 °C for a month. - Mineral oil (Trinity Biotech, catalog number: 400-5-1000 )
Note: The mineral oil was replaced to a fresh one every time-lapse recording. - Brefeldin A (Cell Signaling, catalog number: 9972 ) or Exo1 (Sigma-Aldrich, catalog number: E8280 )
Note: To examine whether membrane trafficking mediated by COPI is required for cellular dynamics, each compound was used to inhibit αCOPI. They were directly added to the culture medium. The BRB80 buffer containing Brefeldin A or Exo1 was prepared before the dissection every time. The testis can be incubated in the culture medium for up to 14 h before isolation of spermatocytes. - PIPES (Dojindo Mecular Technologies, catalog number: 340-08255 )
- Magnesium chloride hexahydrate (MgCl2·6H2O) (Nacalai Tesque, catalog number: 20908-65 )
- Ethylene Glycol Bis (EGTA) (Nacalai Tesque, catalog number: 08907-42 )
- Potassium chloride (KCl) (Wako Pure Chemical Industries, catalog number: 162-17942 )
- Sodium chloride (NaCl) (Wako Pure Chemical Industries, catalog number: 191-01665 )
- Sodium phosphate dibasic (Na2HPO4)
- Potassium dihydrogen phosphate (KH2PO4)
- M3 medium (Sigma-Aldrich, catalog number: S8398 )
- BRB80 buffer (pH 6.8) (see Recipes)
- Insect M3 medium (see Recipes)
- Phosphate-buffered saline (PBS) (see Recipes)
Equipment
- Super fine forceps (Fine Science Tools, model: Dumont #5 )
- Dissection needles sharpened tungsten wire with 0.5 mm diameter
- Inverted fluorescence microscope (Olympus, model: IX81 ) outfitted with excitation, emission filter wheels (Olympus, Tokyo, Japan)
- Objectives; UPLFLN40XPH (NA=0.75), UPLSAPO60XO (NA=1.4), UPLSAPO100XO (NA=1.4) (Olympus, Tokyo, Japan)
- Hg lump (Olympus, catalog number: USH-1030L )
- Cooled CCD camera (Hamamatsu Photonics, model: C10600-10B )
- Autoclave
Software
- Metamorph software version 7.6 (Molecular Devices, Sunnyvale, USA)
Procedure
- To label cellular components of interest with fluorescence, one can induce fluorescence-tagged proteins, which consists of each target component by Drosophila ectopic gene expression system, Gal4/UAS (Ashburner et al., 2004; White-Cooper, 2012). Alternatively, one may use strains expressing such fluorescence proteins continuously. In former cases, F1 progenies obtained by crossing bam-Gal4::vp16 with an UAS stock in which a cDNA encoding the protein with a fluorescence tag is placed under a control of the UAS sequences can be used for the time-lapse observation (see the schematic diagram for genetic cross in Figure 2). In testes cells in the F1 progeny males, expression of the fluorescence protein is specifically induced. The F1 progenies are reared at 28 °C (not beyond this temperature) in order to induce effective expression. For depletion experiments, another UAS stock carrying a transgene so as to induce dsRNA for each target protein is used instead of the UAS stock described above. After selection of male flies for the dissection, any preparation should be performed at an ambient room temperature below 25 °C.
Note: One should adjust the room temperature around the microscope stage to 25 °C at least 1 h before observation.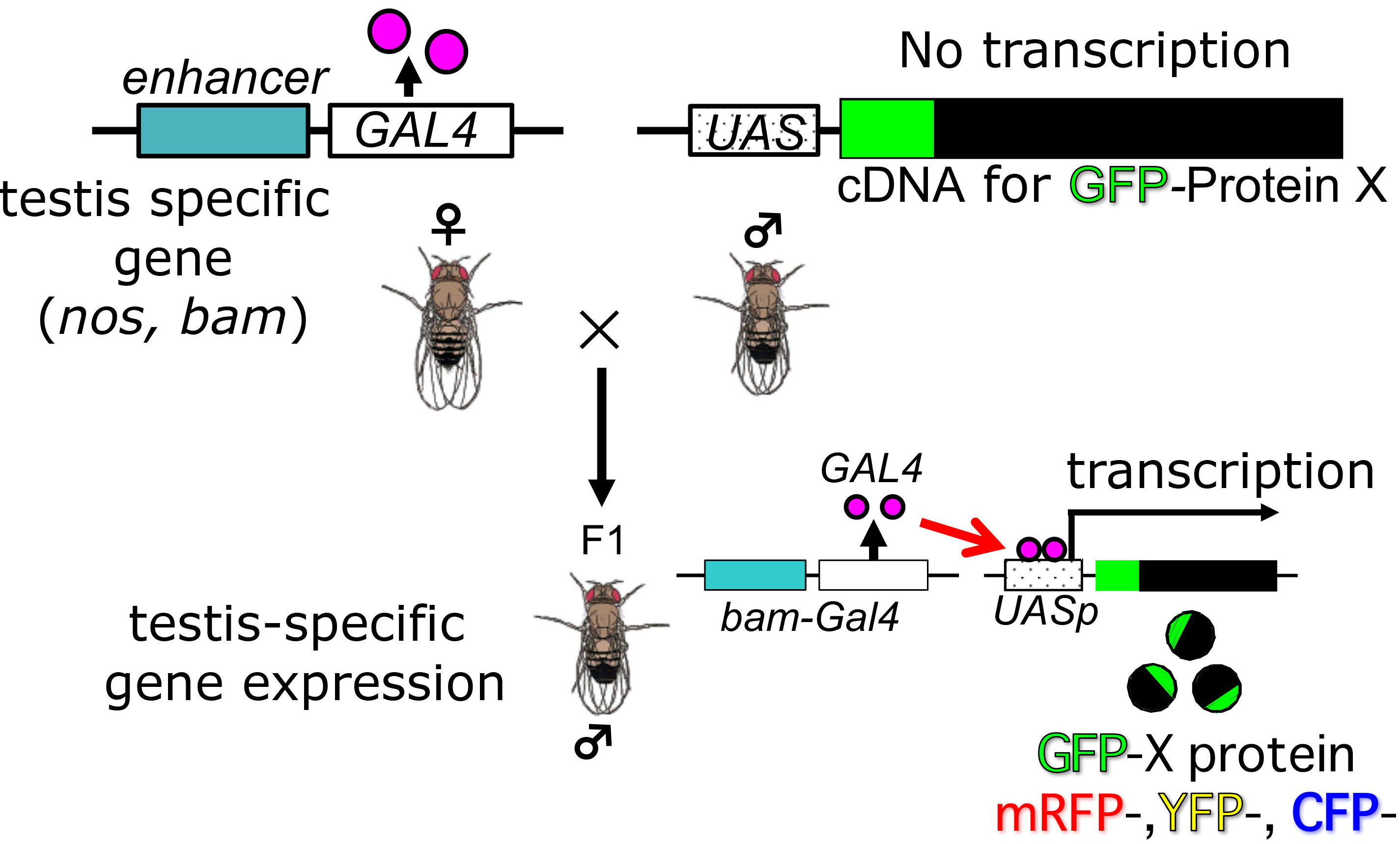
Figure 2. Genetic cross to generate F1 progenies producing fluorescence-tagged protein to label intracellular structures or F1 progenies expressing dsRNA to deplete a target protein. For labeling intra-cellular structures, induced expression of cDNA for a fusion protein X, which was consisting of the structures, with GFP fluorescence tag is carried out by Gal4/UAS system. For a depletion experiment, dsRNA representing a portion of target mRNA was induced instead of a fluorescence protein X. - A pair of testis was carefully collected from pharate adults or newly eclosed adult flies (0-1 day old) described above at a room temperature (Figure 3). To collect adult testes, a movie uploaded to the internet web site would be useful as a guide (https://www.youtube.com/watch?v=-ej8nF1YsRg). The fly was placed ventral side up in BRB80 buffer on a plastic Petri dish under a dissecting microscope. Using a pair of the forceps, their abdomens were clamped by forceps and gently pulled the external genitalia in opposite direction by another one. After carefully removed associated accessory tissues away from a pair of testes, a pair of coiled testes were isolated as shown in Figure 3C. In a case of colchicine or cytochalasin D treatment, this dissection step was carried out in a BRB80 buffer containing 50 μg/ml colchicine and 10 μg/ml cytochalasin D, respectively. For a longer treatment, the spermatocytes isolated from testes can be incubated in M3 culture medium containing 10% fetal calf serum and 50% male cell extracts with a drug until 14 h (Kitazawa et al., 2012).
Note: It is important to select young flies as much as possible because testes in aged flies contained less numbers of living spermatocytes instead of a large amount of sperm bundles. To collect young flies, all adult flies were discarded from fly culture tubes and newly eclosed flies were collected 12 h to 24 h after the discard. Remove a pair of testis gently and quickly using forceps. It is recommended that one complete the testis dissection (steps 1 to 4) within three minutes.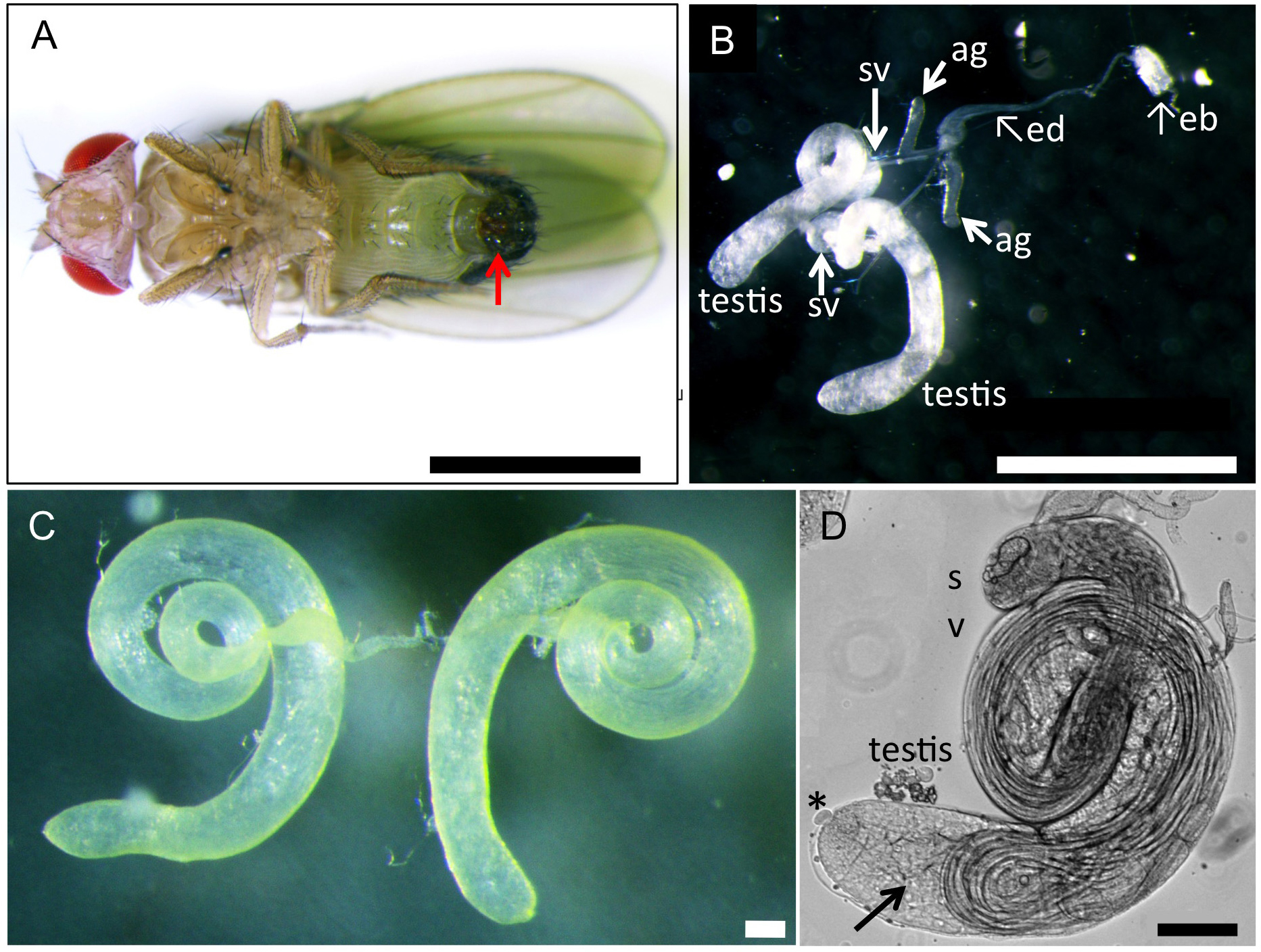
Figure 3. A reproductive tract including a pair of testis collected from a normal male adult. A. A ventral view of a wild-type adult male. An arrow indicates external genitalium. Scale bar = 1 mm. B. A reproductive tract collected from a normal pharate adult male. The male reproductive tract is consisting of a pair of testis, seminal vesicle (sv) and accessory gland (ag). Ejaculatory duct (ed) and ejaculatory bulb (eb) are also attached. Scale bar = 1 mm. C. A pair of testis isolated from an adult male after removal of associated tissues such as seminal vesicles and accessory glands away. Note that flies carrying a w mutation have colorless testes, while wild type testes display pale yellow as shown in this photo. Scale bar = 100 μm. D. A phase construct micrograph of a half pair of testis. There would be at least one or more meiotic cysts consisted of 16 primary spermatocytes undergoing meiosis I at one-third the away from the apical tip of the testis (arrow). The dissection should be carried out to tear the sheath open at this point. Apical tip (asterisk). Scale bar = 100 μm. - One or two testes were transferred and laid out under mineral oil (Trinity Biotech, Bray, Ireland) filled in open chambers on a clean glass cover slip (Figure 4). To prevent a drop of the oil to spread over the cover slip, it was surrounded by a 10 x 10 mm open frame that had adhesives on the bottom side (Gene Frame 25 μm, Thermo Fisher Scientific Co., Waltham, USA). Extra liquid associated around the testis should be sucked up using a string of Kim-wipe carefully in order to avoid an unexpected slip down of the testis cells due to the liquid remaining beneath the cells during a time-lapse recording (Figure 3D).
Note: It is recommended to collect intact and a health-looking testis complex in which ejaculatory pump is still actively contracting, if possible (Figure 3B). The testes punctured in the process of dissection should not be selected.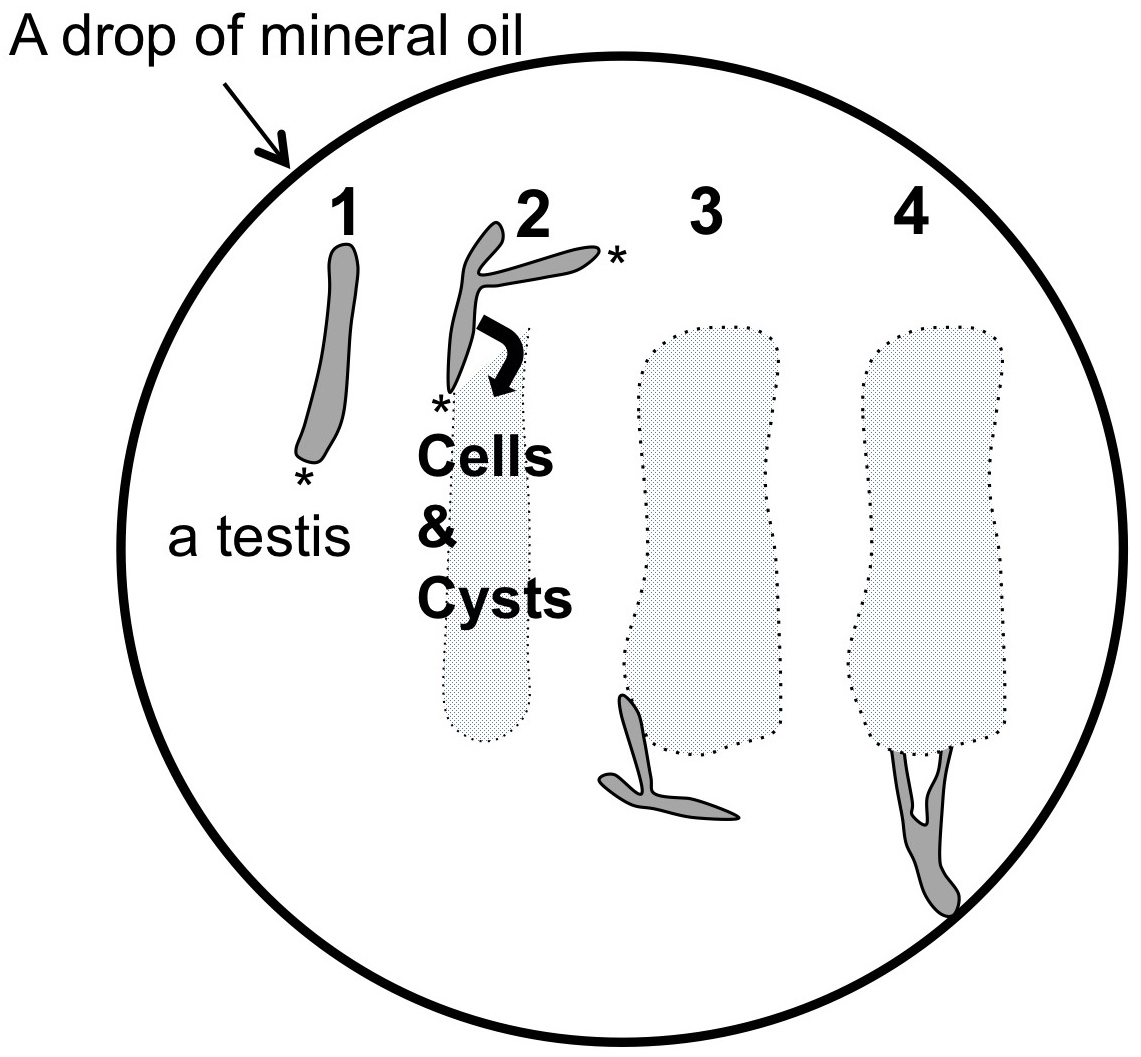
Figure 4. Schematic diagram of procedure to make cysts and cells within testis to spill into the mineral oil. 1. A half of testis pair illustrated here in a darker gray is located straight in a drop of mineral oil. One or two pairs of testis can be dissected in the drop of the oil. 2. Using a pair of tungsten needles, a sheath covering a testis is torn open from the apical tip (asterisks) until one-third of the testis. 3. By gently moving the testis sheath outward using needles, cysts consisting of 16 primary spermatocytes can be spread into the oil. The cells and cysts spread out are indicated in a light gray. On the other hand, each cyst should be kept intact as much as possible. The testis sheath is removed from the oil drop. 4. Using a pair of tungsten needles attached at the tip of holders in each hand, the sheath covering a testis is torn at a position at one third from the apical tip (arrow in Figure 3D) so as to allow a whole cyst consisting of 16 primary spermatocytes to release into the oil outside of the testis, while keeping intact. The oil should cover every testis cell at any time to prevent desiccation. Under these conditions, the cells within intact cysts remained viable for several hours (Kitazawa et al., 2012; Savoian, 2015).
Note: A few germline stem cells are located at the apical tip of testis (Ueishi et al., 2009). And 16 cell cysts undergoing 1st meiotic division are localized around one third from the apical tip (Figures 3C, 3D and Figure 4).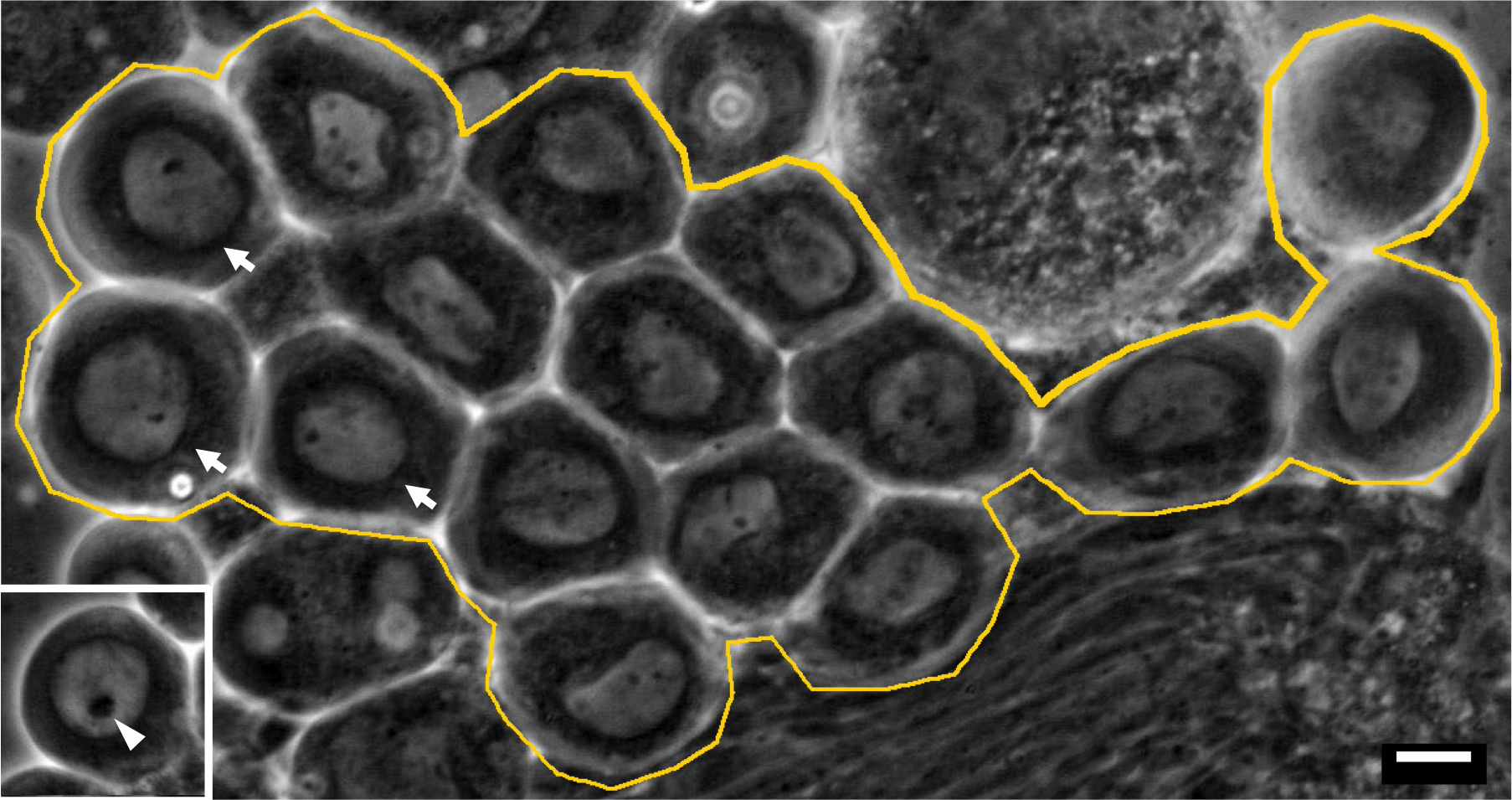
Figure 5. A phase contrast micrograph of a living 16-cell cyst of spermatocytes at early stage of meiosis I under mineral oil. Primary spermatocytes that have initiated meiotic division I are indicated by arrows. These cells should be selected for the time-lapse recording. (Inset) A premeiotic cell from a primary spermatocyte cyst at earlier stage. Note that it has a larger nucleolus (arrowhead). An intact 16-cell cyst is encircled by a yellow line. Bar = 10 μm. - The cover slips set on the plastic cover slip holder are placed on an inverted fluorescence microscope (Olympus, Tokyo, Japan), outfitted with excitation, emission filter wheels (Olympus, Tokyo, Japan). The fluorescence signals are collected using a 40x dry objective lens or a 100x oil immersion lens.
- As Drosophila spermatocytes are extremely sensitive to light (Rebollo and Gonzalez, 2004), one of most crucial points for the time-lapse imaging is to restrict the dose of irradiation to the living cell specimens as much as possible. Thus, after setting the holder on the microscope stage, one should look for prophase I cysts in which mature spermatocytes would initiate meiotic division I soon after using phase contrast optics under a transmission light through ND25 filter. In the prophase I cells, round-shaped multi-layers of nuclear envelope are formed, while oval-shaped elongated envelopes are observed at stages after prometaphase I. Primary spermatocytes within an intact cyst rather than cells stayed alone should be selected for the imaging as much as possible.
- A recording of time-lapse images started from timing when a fluorescence of GFP-Tubulin initiated to accumulate at spindle poles located opposite to each other (t = 0 min). Alternatively, one should start image collection from timing when nucleoli became almost disappeared under a phase contrast (t = 0 min) (Figure 6).
Note: It is advisable to initiate time-lapse imaging within ten minutes from the initial step of the dissection.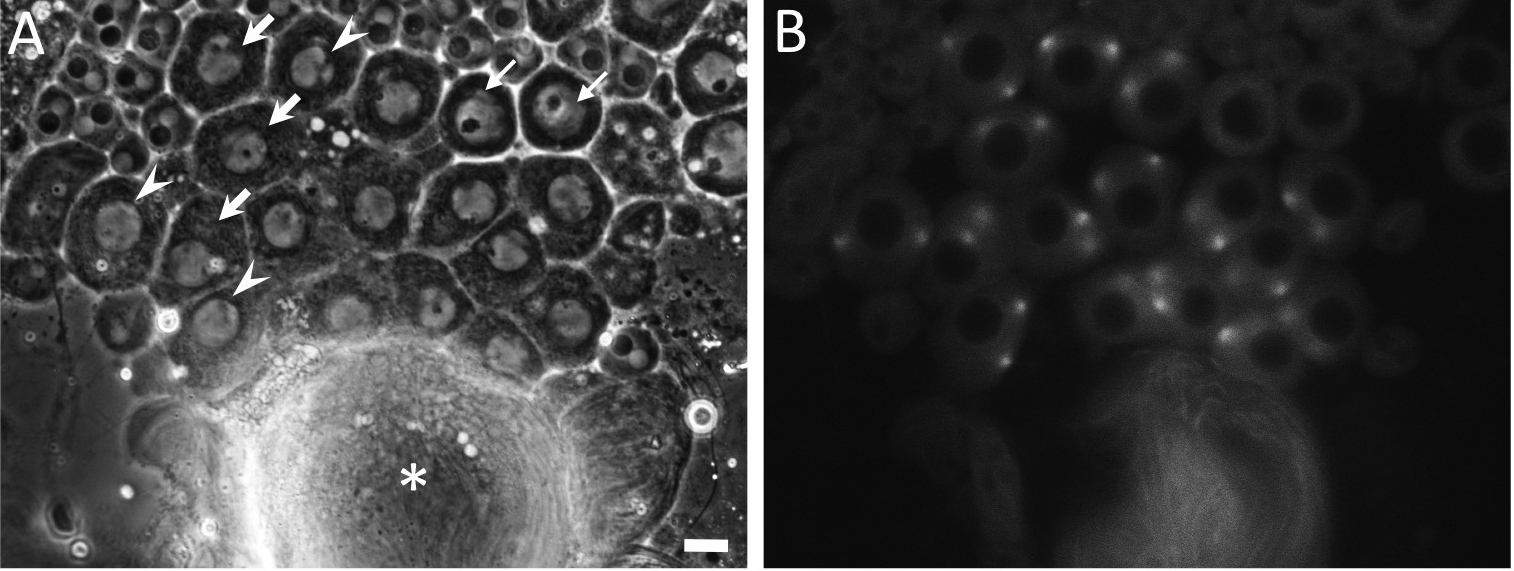
Figure 6. Phase-contrast observation and fluorescence observation of testis cells including primary spermatocytes expressing GFP-βTubulin just before or at onset of meiosis I. Primary spermatocytes at prophase I or cells that would initiate meiotic division I soon after should be found by a phase contrast observation under a transmission light in order to avoid a prolonged irradiation of excitation light from a Hg lamp. The spermatocytes before and at prophase I show round-shaped cell morphology, while cells elongate to shape oval morphology as meiosis progress after prometaphase I. Premeiotic spermatocytes at S5 stage (small arrows) contain single larger nucleoli. As meiotic cell cycle progress, the nucleoli become smaller and disintegrated at S6 stage (arrows). The spermatocytes at prophase I, which contained tiny almost invisible nucleoli (arrowheads, A), also show that intensity of a fluorescence of GFP-Tubulin at the spindle poles separated apart toward opposite direction becomes vigorous (B). Asterisk (*) indicates bundled tails of elongated spermatids. Image collection can start from timing when a nucleolus has disappeared under a phase contrast (t = 0 min). In the case that a fluorescence of GFP-Tubulin is simultaneously collected, the recording should be initiated at timing when the GFP fluorescence becomes stronger at the poles (t = 0 min). Scale bar = 10 μm. - At each 30-sec time interval, fluorescence-tagged proteins in the interested cells are excited by irradiation light from mercury lamp. Specimens are illuminated with UV filtered and shuttered light using the appropriate filter wheel combinations through a GFP/RFP filter cube. Near-simultaneous GFP and/or RFP fluorescence images are captured with a CCD camera (Hamamatsu Photonics, Shizuoka, Japan). For instance, a sequential collection of fluorescence images (10 msec exposure) and phase contrast images (300 msec exposure using ND12 filter) can be carried out for time-lapse observation to examine dynamics of cellular components in the primary spermatocytes. Image acquisition is controlled through the Metamorph software version 7.6 (Molecular Devices, Sunnyvale, CA, USA).
Note: One can adjust the focus and the microscope stage so as to make target cellular components clearly visible while looking at the computer screen. - For a drug treatment, we carried out short term in vitro culture of primary spermatocytes (Rebollo and Gonzalez, 2004; Kitazawa et al., 2012).
- A testis complex attached with accessory gland, ejaculatory duct and pomp was collected from adult males. A living testis complex in which ejaculatory pomp was actively contracting should be selected and transferred into the M3 culture medium.
- For a longer incubation with special drugs such as Brefeldin A or Exo1, these inhibitors for intracellular vesicle transport can be directly added to the modified culture medium consisting of the M3 medium without bicarbonates containing 10% fetal calf serum and 50% male cell extracts as prepared in according with (Kitazawa et al., 2012).
- The testis was incubated in the culture medium for 14 h before isolation of spermatocytes at room temperature.
Note: Using this protocol, one can carry out continuous observation of primary spermatocytes undergoing proper chromosome segregation and cytokinesis for at least an hour without any distinct abnormalities (Kitazawa et al., 2012 and 2014; Hayashi et al., 2016). A time-lapse imaging can continue to the end of meiosis II through meiosis I without medium changes, although a prolonged incubation of the cells without media replacement may result in cellular toxicity due to extra accumulation of metabolic wastes.
Data analysis
- This protocol usually allows us to perform a time-lapse observation of chromosomes, microtubules, F-actin, Golgi stacks, ER-based structures and mitochondria in primary spermatocytes undergoing meiosis I with good reproducibility. Spermatocytes carrying a transgene(s) (reagents 7, 8, 15, 16) to induce expression of fluorescence-tagged proteins consisting the intracellular structures can be used for the time-lapse experiments. Alternatively, spermatocytes are prepared from males generated by a cross between bam-Gal4 stock (reagent 5) and the UAS stocks (reagents 12-14, 17-19) to induce fluorescence proteins by Gal4/UAS system.
- One can also perform a simultaneous recording of multiple fluorescence having different wavelengths emitting from, for example GFP-Histone 2Av and RFP-βTubulin proteins. At prophase I, chromatins that have been homogeneously distributed before become assembled to form chromosomes at timing when accumulation of RFP-βtubulin becomes distinct at spindle poles (t = 0 min). Four foci of GFP-Histone 2Av corresponding to bivalents between two major autosomes, X-Y chromosomes and tiny 4th chromosomes should be formed at prometaphase I (t = 15 min). Bivalents of smaller 4th chromosomes are not usually observed because of overlapping with major chromosomes.
- At prometaphase I, the condensed bivalent chromosomes appeare within a nucleus in which nuclear membrane seems to be intact (t = 5 min). Astral microtubules emanating from spindle poles are constructed at prometaphase I. Developing asters have moved around nuclear membrane as to reach at opposite poles. Then, all chromosome complements appears to congress into a single chromosome mass at the center of the bipolar spindle structure until metaphase I (t = 55 min). Each chromosome performs poleward movement with an average velocity of 11.2 ± 1.2 µm/min until bipolar kinetochore attachment (Savoian et al., 2000). State of microtubule assembly is surveyed at the spindle checkpoint, although the checkpoint at male meiosis is less strict than that in somatic cells (Rebollo and Gonzalez, 2000). Anaphase I takes around 8 min and that the chromosomes moves poleward at 1.9 ± 0.1 µm/min after dyad disjoining (Savoian et al., 2000). At this stage, the nuclear membrane around spindle poles has already disintegrated and the spindle microtubules are free to elongate into the inside of the nuclear space. At onset of anaphase I, multi-layers of nuclear membrane surrounding the nuclear space separate spindle microtubules including thick kinetochore microtubules form astral microtubules. Two populations of central spindle microtubules appear after disjunction of bivalents (t = 60 to 70 min). A peripheral set of the microtubules become more dynamic as if they look for the cytoplasm towards the cell equator (Inoue et al., 2004). Another set of the microtubule bundles corresponding spindle microtubules is localized interiorly at the middle of the cell. The peripheral microtubules from opposite poles meet at equator and formed bubble-like structures protruding outwards (t = 60 min). The interior and most of the peripheral central spindles are then released from each pole and they form independent bundles at the equator. Furrow ingression is then observed soon after the peripheral microtubules from both poles contact the cell cortex. Chromosomes are de-condensing as the furrowing progresses (t = ~80 min).
- According to the procedure and stocks described above (reagents 9-11, 16-18), dynamics of Golgi stacks, endoplasmic reticulum-based structures, nuclear envelopes, plasma membranes and mitochondria in male meiosis can be also reproducibly examined (Inoue et al., 2012; Kitazawa et al., 2012; Hayashi et al., 2016). Distribution of F-actin and other cytoskeletons including contractile rings at cleavage furrow sites can also be visualized using several markers generated by stocks described above (reagents 13-15). A cellular localization of regulatory proteins for microtubules dynamics in cell division such as Orbit, Pavarotti and Polo can be examined in living spermatocytes using stocks (reagents 12 and 19). Excessive overexpression of a microtubule-associated protein, Orbit that stabilizes microtubules result in generation of abnormal spindle structures at a certain frequency (more than 10%). The details of the dynamics have been described elsewhere (Inoue et al., 2012; Kitazawa et al., 2014)
- In RNAi experiments by ectopic expression of dsRNA for target genes specifically in spermatocytes, one can examine whether dynamics of chromosomes, cytoskeletons and other cell organelles would be affected. Before the time-lapse recording, one should test whether UAS-RNAi stocks could perturb chromosome segregation, cytokinesis or mitochondrial partition in the presence of bam-Gal4 driver. It can be investigated by observation of post-meiotic spermatocytes under phase contrast microscope (see supplement table in Kitazawa et al., 2014). On the basis of overlapped phenotypes observed in multiple RNAi experiments using different UAS-RNAi stocks for the same genes, one should argue a role of the gene in male meiotic division.
- In a drug treatment, primary spermatocytes are pre-treated in a dissection buffer containing the drug. Alternatively, the time-lapse recording is carried out in the presence of the drug. One should consider the abnormalities observed exclusively in cells treated with the drug as cell phenotypes by the drug treatment. The cell phenotypes should be observed in a manner dependent on drug concentration. One should confirm that they appear at a higher frequency and that the phenotypes are enhanced as its concentration increases.
Notes
As a result of this protocol, dynamics of both chromosomes and microtubules as described above can be observed at good reproducibility. One should stop the time-lapse recording and discard the cells, if the cells quit cell division or abnormal microtubule structures are detected, such as multi-polar spindles that appear due to improper physiological condition. Homozygotes for P{His2Av-GFP} as well as those for P{His2Av-mRFP} generated abnormal spermatocytes which meiotic progression was arrested in the middle at a low frequency (less than 10%).
Recipes
Note: The ingredients of buffers or media and catalog number of each reagent described above are as follows. For preparation of all buffers, media and reagents, ultrapure water prepared by a water purification system such as Sartorius arium® should be used. Buffers described below can be kept at room temperature unless otherwise noted. Any materials used in this protocol were subject to MTAs.
- BRB80 buffer (pH 6.8)
80 mM PIPES
1 mM MgCl2
1 mM EGTA - Insect M3 medium
Insect M3 medium containing 10% fetal calf serum and 50% male cell extracts (Kitazawa et al., 2012) was prepared just before each time-lapse recording experiment under sterile working conditions - Phosphate-buffered saline (PBS) (pH 7.4)
137 mM NaCl
2.68 mM KCl
10.14 mM Na2HPO4
1.76 mM KH2PO4
To make 1 L of PBS, 8 g of NaCl, 0.2 g of KCl, 1.44 g of Na2HPO4 and 0.24 g of KH2PO4 were combined and dissolved in H2O to a total volume of 1 L. The pH was adjusted to 7.4. The buffer was sterilized by autoclaving. The buffer can be stored at room temperature
Acknowledgments
We are grateful to M. S. Savoian (Massey University, New Zealand) for sharing information about experimental procedures. We acknowledge V. Doyle (Institut Jacques Monod, Paris, France), L. Wallrani (University of Iowa, Iowa City, USA), D. Glover (University of Cambridge, Cambridge, UK), L. Cooley (Yale University, USA) and J. A. Brill (Toronto University, Toronto, Canada). We also thank Vienna Drosophila RNAi Center, Bloomington Stock Center and Drosophila Genetic Resource Center for providing fly stocks.
No competing or financial interests that may impact the design and implementation of their protocol. This work was partially supported by Japan Society for the Promotion of Science [grant number 26440188 to Y.H.I.]. This protocol was adapted or modified from our previous studies (Inoue et al., 2004; Kitazawa et al., 2012; Kitazawa et al., 2014; Hayashi et al., 2016).
References
- Ashburner, M., Golic, K. G. and Hawley, R. S. (2004). Drosophila: a laboratory handbook. 2nd. Cold Spring Harbor Laboratory Press.
- Clarkson, M. and Saint, R. (1999). A His2AvDGFP fusion gene complements a lethal His2AvD mutant allele and provides an in vivo marker for Drosophila chromosome behavior. DNA Cell Biol 18(6): 457-462.
- Giansanti, M. G., Sechi, S., Frappaolo, A., Belloni, G. and Piergentili, R. (2012). Cytokinesis in Drosophila male meiosis. Spermatogenesis 2(3): 185-196.
- Hayashi, D., Tanabe, K., Katsube, H. and Inoue, Y. H. (2016). B-type nuclear lamin and the nuclear pore complex Nup107-160 influences maintenance of the spindle envelope required for cytokinesis in Drosophila male meiosis. Biol Open 5(8): 1011-1021.
- Inoue, Y. H., Miyauchi, C., Ogata, T. and Kitazawa, D. (2012). Dynamics of cellular components in meiotic and premeiotic divisions in Drosophila males. In: Swan, A. (Ed.). InTech. Open Access Publisher 67-86.
- Inoue, Y. H., Savoian, M. S., Suzuki, T., Mathe, E., Yamamoto, M. T. and Glover, D. M. (2004). Mutations in orbit/mast reveal that the central spindle is comprised of two microtubule populations, those that initiate cleavage and those that propagate furrow ingression. J Cell Biol 166(1): 49-60.
- Kitazawa, D., Matsuo, T., Kaizuka, K., Miyauchi, C., Hayashi, D. and Inoue, Y. H. (2014). Orbit/CLASP is required for myosin accumulation at the cleavage furrow in Drosophila male meiosis. PLoS one 9: e93669.
- Kitazawa, D., Yamaguchi, M., Mori, H. and Inoue, Y. H. (2012). COPI-mediated membrane trafficking is required for cytokinesis in Drosophila male meiotic divisions. J Cell Sci 125(Pt 15): 3649-3660.
- Miyauchi, C., Kitazawa, D., Ando, I., Hayashi, D. and Inoue, Y. H. (2013). Orbit/CLASPis required for germline cyst formation through its developmental control of fusomes and ring canals in Drosophila males. PLoS one 8(3): e58220.
- Rebollo, E. and Gonzalez, C. (2004). Time-lapse imaging of male meiosis by phase-contrast and fluorescence microscopy. Methods Mol Biol 247: 77-87.
- Savoian, M. S. (2015). Using photobleaching to measure spindle microtubule dynamics in primary cultures of dividing Drosophila meiotic spermatocytes. J Biomol Tech 26(2): 66-73.
- Savoian, M. S., Goldberg, M. L. and Rieder, C. L. (2000). The rate of poleward chromosome motion is attenuated in Drosophila zw10 and rod mutants. Nat Cell Biol 2(12): 948-952.
- Ueishi, S., Shimizu, H. and Inoue, Y. H. (2009). Male germline stem cell division and spermatocyte growth required insulin signaling in Drosophila. Cell Struct Funct 34: 61-69.
- White-Cooper, H. (2012). Tissue, cell type and stage-specific ectopic gene expression and RNAi induction in Drosophila testis. Spermatogenesis 2(1):11-12.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Tanabe, K., Okazaki, R., Kaizuka, K. and Inoue, Y. H. (2017). Time-lapse Observation of Chromosomes, Cytoskeletons and Cell Organelles during Male Meiotic Divisions in Drosophila. Bio-protocol 7(8): e2225. DOI: 10.21769/BioProtoc.2225.
Category
Cell Biology > Cell imaging > Live-cell imaging
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









