- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Cued Rat Gambling Task
Published: Vol 7, Iss 3, Feb 5, 2017 DOI: 10.21769/BioProtoc.2118 Views: 13267
Reviewed by: Soyun KimAdler R. DillmanAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Pupillometry: A Simple and Automatic Way to Explore Implicit Cognitive Processing
Tian Yuan [...] Yi Jiang
Apr 5, 2025 1294 Views
The Mouse Social Frailty Index (mSFI): A Standardized Protocol
Charles W. Collinge [...] Alessandro Bartolomucci
Apr 20, 2025 1809 Views
Training Mice to Perform Attentional Set-Shifting Under Head Restraint
Katarina Kalajzic [...] Timothy Spellman
Sep 5, 2025 1470 Views
Abstract
The ability of salient cues to serve as powerful motivators has long been recognized in models of drug addiction, but little has been done to investigate their effects on complex decision making. The Cued rat Gambling Task (CrGT) is an operant behavioural task which pairs salient, audiovisual cues with the delivery of sucrose pellet rewards on complex schedules of reinforcement that involve both sugar pellet ‘wins’ and timeout penalty ‘losses’. The task was designed with the intention of providing insight into the influence of such cues on decision making in a manner that models human gambling.
Keywords: Decision makingBackground
Although numerous rodent behavioural paradigms that capture different facets of gambling-like behaviour have recently been developed, the motivational power of cues in biasing individuals towards risky choice has so far received little attention despite the central role played by drug-paired cues in successful laboratory models of chemical dependency. Here, we describe the cued rat Gambling Task (CrGT) - a cued version of the rGT analogue of the Iowa Gambling Task (Zeeb et al., 2009). In these tasks, animals chose between four options associated with different magnitudes and frequencies of reward and punishing time-out periods. As in the Iowa Gambling Task, favoring options associated with smaller per-trial rewards but smaller losses, and avoiding the tempting ‘high-risk, high-reward’ options, maximized gains on the task. Crucially, in the CrGT, salient, audiovisual cues were paired with the delivery of sucrose pellet rewards. These cues increase in complexity with the size of the ‘win’, similar to human gambling scenarios. Recent data indicate that the addition of these reward-concurrent cues drastically increases choice of the maladaptive, risky options, thereby biasing choice against the animals’ best interests.
Materials and Reagents
- Male Long-Evans rats (Charles Rivers Laboratories) weighing 250-275 g upon arrival at the animal facility
Notes: - Initially animals should be gradually food restricted to 85% of free-feeding weight (fed ~14 g of standard rat chow per day).
- Water is available ad libitum in home cages.
- Animals are housed in pairs/trios and maintained in a climate-controlled colony room on a 12 h reverse light cycle (lights off at 8:00 AM). Temperature is kept between 19-23 °C and humidity ranges from 40%-70%.
- Bio-Serv Dustless Precision Pellets, 45 mg, sucrose (Bio-Serv, catalog number: F06233 )
Equipment
- Chambers (Med Associates) (Depicted in Figure 1)
- Extra tall modular test chamber (Med Associates, catalog number: ENV-007-VP )
- Extra tall MDF sound attenuating cubicle (Med Associates, catalog number: ENV-018MD )
- 5 unit curved nose poke wall (Med Associates, catalog number: ENV-115A )
- Stainless steel grid floor (Med Associates, catalog number: ENV-005 )
- Hooded house light (Med Associates, catalog number: ENV-215M )
- Pedestal mount pellet dispenser for rat (Med Associates, catalog number: ENV-203-45 )
- Pellet receptacle (Med Associates, catalog number: ENV-200R2M-6.0 )
- Receptacle light (Med Associates, catalog number: ENV-200RL )
- Head entry detector for rat (Med Associates, catalog number: ENV-254-CB )
- Multiple tone generator (Med Associates, catalog number: ENV-223 )
- Cage speaker (Med Associates, catalog number: ENV-224AM )
- Large tabletop cabinet and power supply (Med Associates, catalog number: SG-6510D )

Figure 1. Testing chamber. ‘A’ right side of chamber, ‘B’ left side.
- Extra tall modular test chamber (Med Associates, catalog number: ENV-007-VP )
- IBM-compatible computer running Med PC software
- CrGT code and training programs (5CSRT and CrGT forced choice) (freely available from Dr. Catharine Winstanley upon request)
- Personal protective equipment (PPE): May include (but is not limited to) scrubs, a laboratory coat, gloves, bouffant cap, ventilation mask, and shoe covers, depending on the requirements of the unit in which the work is taking place
Software
- Med PC software
Procedure
Notes:
- Figure 2 depicts the time course of a typical experiment.
- Prior to bringing the animals into the testing room, prepare the operant chambers (ensure cleanliness, connectivity of wires and fill pellet dispensers), and the computer software.
- Five min prior to testing, animals are moved from the colony room to the testing room in their home cages.
- Each animal is subsequently moved into a chamber and undergoes testing according to their respective testing phase.
- Each animal always undergoes testing in the same chamber.
- Animals should be tested within the same 1 h daily window between 8 AM and 8 PM, during their dark cycle. Animals should be tested 5 days a week.
- There is a maximum of one session per day, with each session comprised of an unlimited number of trials.
- Each test session lasts 30 min. Animals usually take 30-40 sessions to achieve a stable, asymptotic baseline on the CrGT, depending on the animal’s cognitive abilities. Ensure that you understand how long it will take to train your animals, and that you have the time in your schedule to complete the experiment.
- Animals should be fed after the training session each day, in order to maximize the motivational value of food deprivation.
- The size of each cohort typically ranges from 8 to 24 animals per experimental condition.
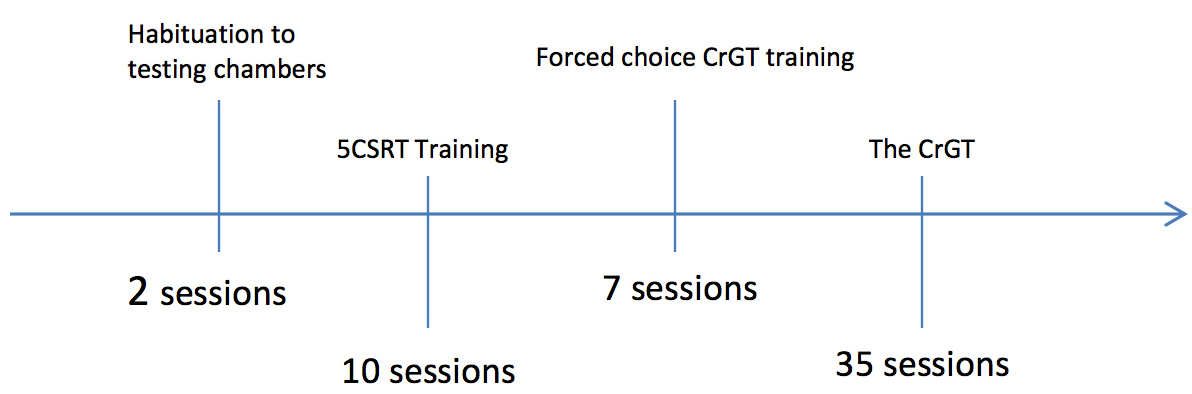
Figure 2. The time course of a typical CrGT experiment
- Training stage 1: Habituation to testing chamber
- In each chamber, place ten reward pellets in the food well and two pellets in each of the five response holes.
- The house light, the food well light and all five stimulus lights (located within the response holes) should stay turned off throughout the session.
- The animal is allowed to freely explore the chamber for 30 min, with the pellets serving as an incentive to nose-poke the response holes and the food tray.
- Following the session, the rat is then returned to its home cage and is fed.
- Repeat steps A1-A4 until the rats reliably consume all the pellets provided within the time allotted (usually 2 sessions).
- In each chamber, place ten reward pellets in the food well and two pellets in each of the five response holes.
- Stage 2: 5-choice serial reaction time (5CSRT) training to encourage rats to nose poke at illuminated lights within the array
- Upon initialization of the computer program, the food tray light illuminates to signal that the first trial is available.
- The animal nose-pokes the food tray to initiate the trial, and the food tray light extinguishes.
- A 5-sec inter-trial interval (ITI) follows the nose poke in the food tray.
- If the animal nose-pokes one of the five response holes during the ITI, the house light illuminates and the trial is recorded as a premature response; after a five sec ‘time out’ punishment, the house light then extinguishes, the food tray light illuminates, and the chamber resets to allow subsequent trials. These premature responses provide an index of impulsive action.
- If the animal nose-pokes one of the five response holes during the ITI, the house light illuminates and the trial is recorded as a premature response; after a five sec ‘time out’ punishment, the house light then extinguishes, the food tray light illuminates, and the chamber resets to allow subsequent trials. These premature responses provide an index of impulsive action.
- Following the ITI, one of the five response hole lights illuminates, chosen pseudo randomly as determined by the MedPC algorithm.
- The animal then has the opportunity to respond to the illuminated stimulus.
- Correct response: the animal nose-pokes the illuminated response hole.
- Incorrect response: the animal nose-pokes in one of the four response holes that are not illuminated.
- Omission: the animal takes no action.
- Correct response: the animal nose-pokes the illuminated response hole.
- The animal is then rewarded or punished for its behaviour.
- A correct response is rewarded with the delivery of one sugar pellet into the food tray, and the food tray light illuminates to signal that the next trial is available.
- Incorrect responses or omissions result in the illumination of the house light and a 5-sec ‘time out’ punishment, wherein the next trial is not available; following the punishment, the house light extinguishes and the food tray light illuminates, signalling that the next trial is available.
- A correct response is rewarded with the delivery of one sugar pellet into the food tray, and the food tray light illuminates to signal that the next trial is available.
- The session expires after 100 trials or 30 min, whichever comes first.
- If the animal registers 30 or more correct responses in a given session, the animal is considered to have passed that level of training and can move on to the next. For each subsequent training level, the task parameters are adjusted to make the task slightly more difficult, until the target parameters are attained (see Table 1 for training stages). These target parameters can usually be reached within 10 sessions.
- Animals progress to forced choice CrGT training when all animals pass training level 3.
Table 1. 5CSRT training stages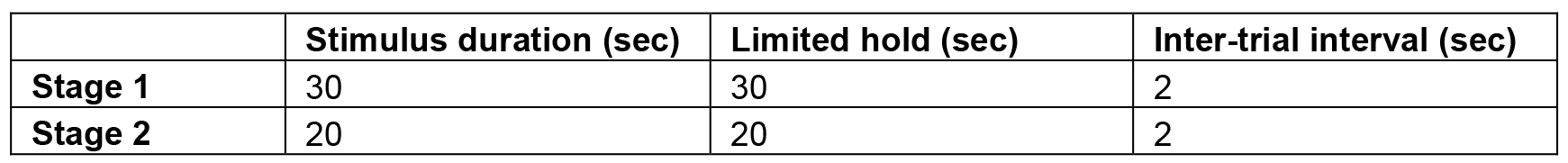
- Upon initialization of the computer program, the food tray light illuminates to signal that the first trial is available.
- Stage 3: Forced choice CrGT training
This training stage is identical to the CrGT task (see below) but only one option is presented on each trial. Roughly equal numbers of each trial type are presented each session, thereby ensuring that the rats adequately sample all four options. Rats receive 7 sessions of forced choice training before moving to the CrGT. - Stage 4: The CrGT
- In the CrGT, animals are presented with four options (named P1-P4) they must select between. Each option is associated with a different schedule of sucrose pellet reward, audiovisual cues and time-out punishment. The physical order in which these options are presented is counter-balanced across each cohort of rats; CrGT version A presents them from left to right as P1, P4, P2, P3 while version B presents them as P4, P1, P3, P2. The contingencies associated with each option are presented in Table 2.
Table 2. Rewards and punishments on the CrGT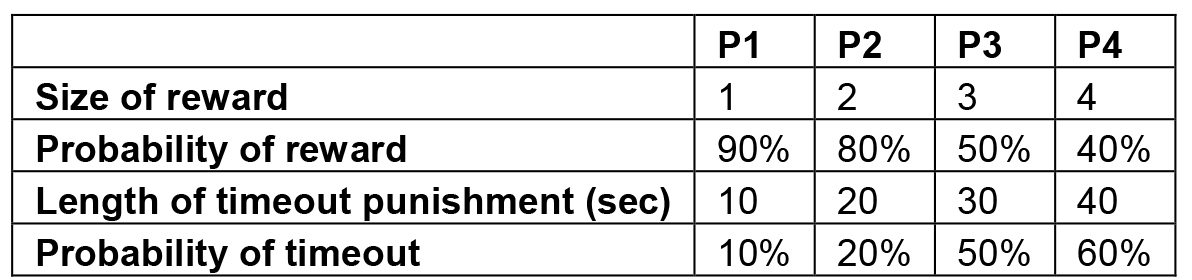
- A task schematic is provided as Figure 3.
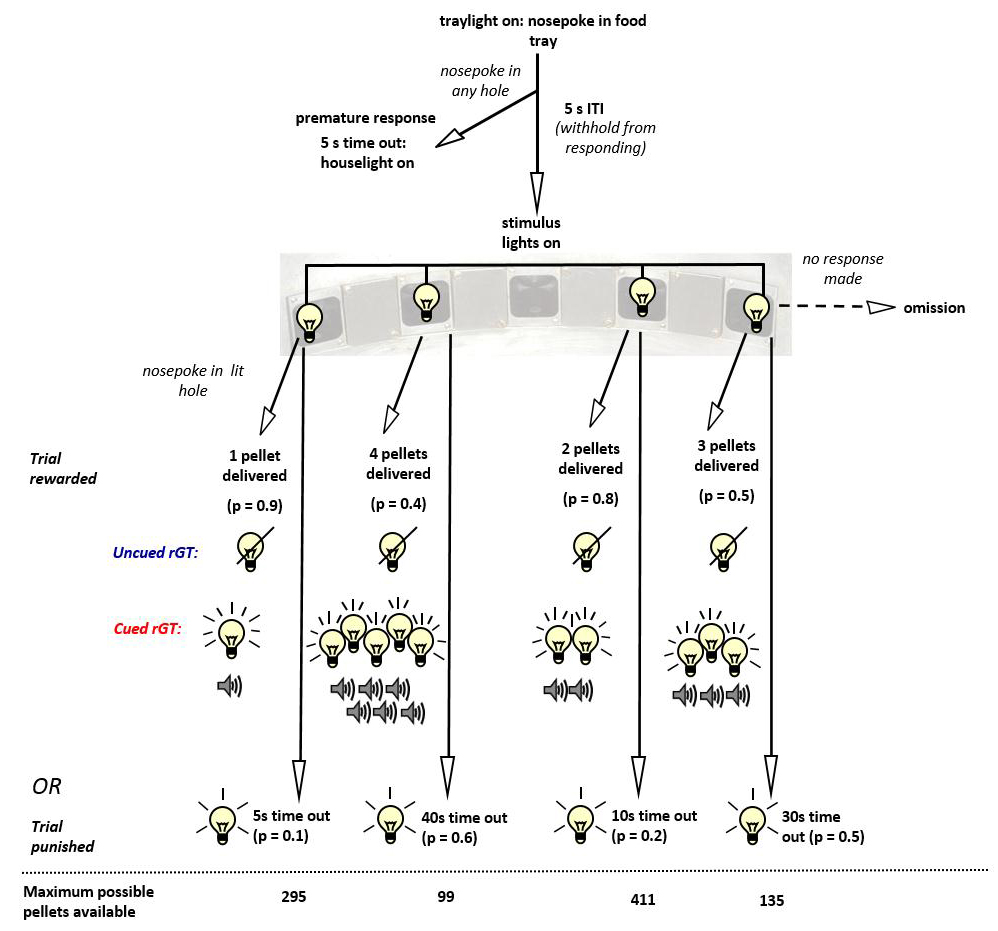
Figure 3. Schematic of the CrGT, showing the sequence of events for each trial and the contingencies associated with each aperture - Animals initiate each trial by making a nosepoke response at the food tray. This triggers the start of a 5 sec inter-trial interval (ITI) before the stimulus lights are turned on in the active holes (holes 1, 2, 4 and 5, running form left to right across the array).
- A response at one of the illuminated holes results in all the stimulus lights being turned off and either delivery of the amount of reward or the start of the time-out ‘punishment’ period associated with that response outcome, according to the reinforcement schedules.
- If the animal is rewarded on any trial, food delivery is signaled by onset of the traylight, and this traylight remains illuminated until the animal collects its reward. Responding at the food tray initiates the start of the next trial.
- If the animal is punished, a time-out period is initiated during which the stimulus light within the hole chosen on that trial flashes at a frequency of 0.5 Hz. This cues the animal to form an association between responding at that particular aperture and the ensuing period of non-reward or frustration. The traylight remains off until the end of the time-out period, whereupon the traylight is turned on to signal that the animal can initiate the next trial.
- If the animal fails to make a response within 10 sec of the illumination of the four stimulus lights, the stimulus lights are turned off, the trial is scored as an omission and the traylight is illuminated to signal the beginning of the next trial. Animals are not punished for omitting trials.
- Premature or impulsive responses made at the array during the ITI are punished by a 5 sec time-out period. During these timeout periods, the houselight is illuminated, all other lights are turned off, and the animal is unable to register any responses. After the time-out period, the traylight is illuminated and the animal can begin a new trial. A series of different trials is depicted in Video 1.
- Training continues until a stable pattern of choice between the four options is observed (typically achieved within 30-40 sessions). A typical pattern of choice amongst the four options is shown in Figure 4.Video 1. Example trials on the CrGT. This video depicts wins and losses on each of the trial types on the CrGT.
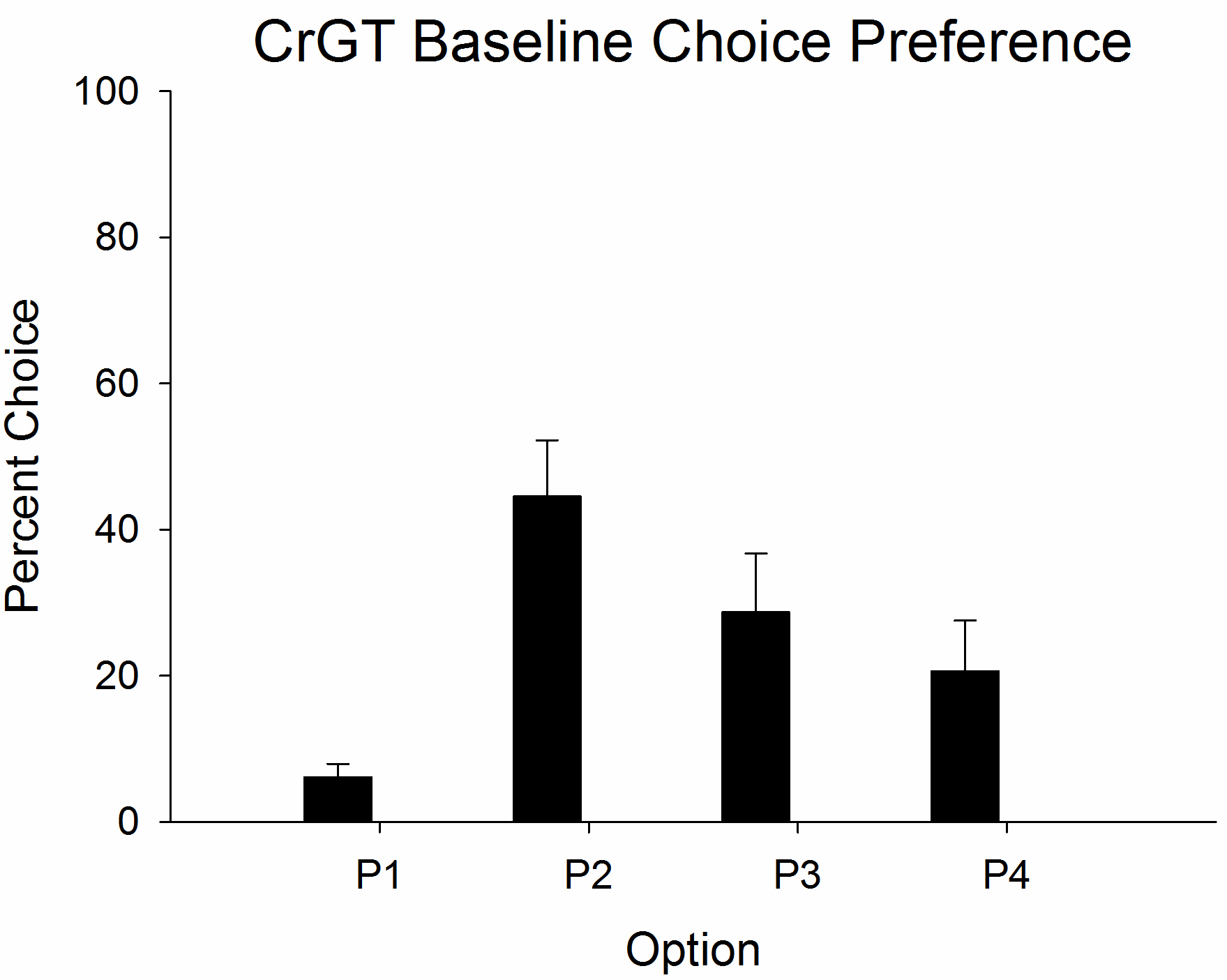
Figure 4. Depiction of typical baseline choice preference on the CrGT. Data derived from Barrus and Winstanley, 2016.
- In the CrGT, animals are presented with four options (named P1-P4) they must select between. Each option is associated with a different schedule of sucrose pellet reward, audiovisual cues and time-out punishment. The physical order in which these options are presented is counter-balanced across each cohort of rats; CrGT version A presents them from left to right as P1, P4, P2, P3 while version B presents them as P4, P1, P3, P2. The contingencies associated with each option are presented in Table 2.
Data analysis
- Percentage variables are arcsine transformed to minimize artificial ceiling effects.
- Significance is set at the P ≤ 0.05 level for all data analyses.
- Repeated-measures ANOVAs are used to analyze data, with choice (four levels, P1-P4), session, and (if relevant) drug dose (four levels, vehicle + three doses of drug) as within-subjects factors and group as a between-subjects factor. Typically, choice, premature responses, choice latency, collection latency, trials and omissions are analysed using the latest version of SPSS (IBM), with the primary dependent variable being choice of the different options.
- Animals are excluded if they fail to achieve behavioral stability (defined as a null effect of session on a repeated measures ANOVA in which session + choice are within-subjects factors analysed over the most recent 3-5 sessions). Animals are also excluded if they fail to complete a sufficient number of trials per session (~20) to allow for meaningful analysis. Both of these scenarios are rare, as long as appropriate training procedures are followed.
Notes
The original uncued version of the task (the rGT) has been in continuous use since 2008, and behaviour on that version has been consistent both within the Winstanley Lab and beyond (i.e., Baarendse et al., 2013; Aleksandrova et al., 2013). All rGT data collected within the Winstanley lab between 2008 and 2012 were included in a meta-analysis that paid particular attention to the relationship between premature responding and choice behaviour on the task (Barrus et al., 2015), which should be consulted for an overview of prototypical rGT behaviour. While the CrGT is still relatively new, we expect that choice behavior will be similarly consistent between cohorts (albeit riskier, on average i.e., Barrus and Winstanley, 2016).
Acknowledgments
The rat Gambling Task, the predecessor to the CrGT, was co-developed by Dr. Fiona Zeeb (first published as Zeeb et al., 2009). The funding for the development of the CrGT was provided in the form of a Canadian Institute of Health Research (CIHR) operating grant awarded to CAW.
References
- Aleksandrova, L. R., Creed, M. C., Fletcher, P. J., Lobo, D. S., Hamani, C. and Nobrega, J. N. (2013). Deep brain stimulation of the subthalamic nucleus increases premature responding in a rat gambling task. Behav Brain Res 245: 76-82.
- Baarendse, P. J., Winstanley, C. A. and Vanderschuren, L. J. (2013). Simultaneous blockade of dopamine and noradrenaline reuptake promotes disadvantageous decision making in a rat gambling task. Psychopharmacology (Berl) 225(3): 719-731.
- Barrus, M. M., Hosking, J. G., Zeeb, F. D., Tremblay, M. and Winstanley, C. A. (2015). Disadvantageous decision-making on a rodent gambling task is associated with increased motor impulsivity in a population of male rats. J Psychiatry Neurosci 40(2): 108-117.
- Barrus, M. M. and Winstanley, C. A. (2016). Dopamine D3 receptors modulate the ability of win-paired cues to increase risky choice in a rat gambling task. J Neurosci 36(3): 785-794.
- Zeeb, F. D., Robbins, T. W. and Winstanley, C. A. (2009). Serotonergic and dopaminergic modulation of gambling behavior as assessed using a novel rat gambling task. Neuropsychopharmacology 34(10): 2329-2343.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Barrus, M. M. and Winstanley, C. A. (2017). Cued Rat Gambling Task. Bio-protocol 7(3): e2118. DOI: 10.21769/BioProtoc.2118.
-
Barrus, M. M. and Winstanley, C. A. (2016). Dopamine D3 receptors modulate the ability of win-paired cues to increase risky choice in a rat gambling task. J Neurosci 36(3): 785-794.
Category
Neuroscience > Behavioral neuroscience > Cognition
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











