- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Pilot-scale Columns Equipped with Aqueous and Solid-phase Sampling Ports Enable Geochemical and Molecular Microbial Investigations of Anoxic Biological Processes
Published: Vol 7, Iss 1, Jan 5, 2017 DOI: 10.21769/BioProtoc.2083 Views: 7846
Reviewed by: Disha SrivastavaAlexander Martin RueckerAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

β-lactamase (Bla) Reporter-based System to Study Flagellar Type 3 Secretion in Salmonella
Fabienne F. V. Chevance and Kelly T. Hughes
Jun 20, 2023 1764 Views
Determination of Poly(3-hydroxybutyrate) Content in Cyanobacterium Synechocystis sp. PCC 6803 Using Acid Hydrolysis Followed by High-performance Liquid Chromatography
Janine Kaewbai-ngam [...] Tanakarn Monshupanee
Aug 20, 2023 1808 Views
An HPLC-based Assay to Study the Activity of Cyclic Diadenosine Monophosphate (C-di-AMP) Synthase DisA from Mycobacterium smegmatis
Avisek Mahapa [...] Dipankar Chatterji
Dec 20, 2024 1779 Views
Abstract
Column studies can be employed to query systems that mimic environmentally relevant flow-through processes in natural and built environments. Sampling these systems spatially throughout operation, while maintaining the integrity of aqueous and solid-phase samples for geochemical and microbial analyses, can be challenging particularly when redox conditions within the column differ from ambient conditions. Here we present a pilot-scale column design and sampling protocol that is optimized for long-term spatial and temporal sampling. We utilized this experimental set-up over approximately 2 years to study a biologically active system designed to precipitate zinc-sulfides during sulfate reducing conditions; however, it can be adapted for the study of many flow-through systems where geochemical and/or molecular microbial analyses are desired. Importantly, these columns utilize retrievable solid-phase bags in conjunction with anoxic microbial techniques to harvest substrate samples while minimally disrupting column operation.
Keywords: Environmental engineeringBackground
The following describes an experimental design and sampling protocol that circumvents the obstacle of vertical coring for temporal and spatial resolution of column systems. The system has the further advantage of minimal disruption to physical, chemical and biological processes. The pilot-scale design incorporates vertically spaced sampling ports for collection of liquid and solid-phase substrate at discrete time points (Figure 1). Spatial sampling of solid-phase substrates that reside within these columns enables researchers to observe biologically relevant processes such as discrete zones of metal immobilization and shifts in microbial biofilm communities. The evolution of these vertical biogeochemical profiles can be tracked and related to performance over time. While optimized for anoxic systems as described below, this experimental design, which surmounts obstacles of more conventional flow-through column systems, could be applied for spatial inquiry into other systems that rely on aqueous and solid-phase interactions.
Of particular interest to our research, Sulfate Reducing Bioreactors (SRBRs) have been employed to mitigate the release of Mining Influenced Water (MIW) for approximately two decades (Wildeman et al., 1994). Due to the anoxic nature of these systems and their spatial heterogeneity, sampling SRBRs without operational disruption presents many challenges. Previously these systems were sampled with limited spatial resolution potentially excluding seminal processes occurring in regions within (Neculita et al., 2008). While sacrificial sampling can surmount this obstacle, it does so at the expense of temporal resolution. Furthermore, spatial inquiry into pilot-scale and larger SRBRs is challenging due to difficulty associated with coring saturated, heterogeneous organic materials (woodchips, sawdust, hay) and disruptions that can result from this form of sample collection. Our design and sampling procedure enabled us to examine the performance of sulfate reducing bioreactors that treat mining influenced water as a function of organic substrate, microbial community structure, water quality, and metal-sulfide precipitation yielding novel insights into the operation of these systems (Drennan et al., 2016).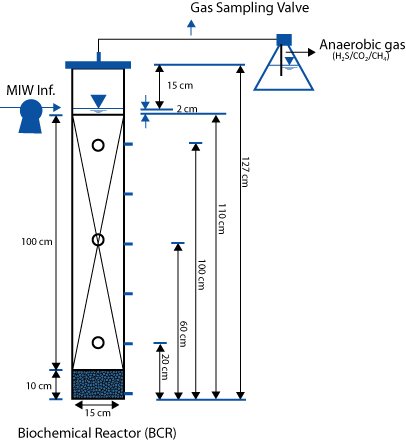
Figure 1. Schematic of vertical down flow biochemical reactor columns. The three ports, indicated by circles on the column, were designed for solid substrate retrieval in conjunction with flow along the z-axis of the columns. The five liquid ports are depicted in blue along the side of the column. For discrete retrieval of solid substrates, columns were temporarily tilted to a horizontal plane using a custom built rack to mitigate water pressure complications and resultant loss (Figures 5 and 6). As labelled, ‘MIW Inf.’ indicates where the mining influenced water is introduced. The effluent from these columns was collected at the bottom liquid port as visualized in Figure 4A.
Materials and Reagents
- Custom column design and construction (materials needed per column)
- Natural organic substrates mixed as described in (Drennan et al., 2016)
- Alfalfa hay (Figure 2A)
- Wood chips (High Desert Investment Company, Phoenix, AZ) (Figure 2B)
- Sawdust (High Desert Investment Company, Phoenix, AZ) (Figure 2C)
- Limestone (Imery’s 3.35-4.95 mm) (Figure 2D)

Figure 2. Examples of solid-phase matrix components deployed within columns. A. Alfalfa hay; B. Woodchips; C. Sawdust; and D. Limestone. Alternative substrates such as sand or other organic solid-phase materials could have relevance to alternative applications such as studying subsurface flow or aquifer recharge.
- A gas collection system linked to the column headspace was constructed using 10% NaOH solution to trap and scrub released sulfide thereby limiting accumulation of this toxin (Figure 3).

Figure 3. Removal of biogenic sulfide. Produced gases traveled from the column headspace to a connected plastic bag. Sulfide was removed by reacting with a 10% NaOH solution.
- Natural organic substrates mixed as described in (Drennan et al., 2016)
- Anoxic columns sampling of aqueous and solid-phase samples
- 10 ml syringes; Luer-Lok® syringes (BD, catalog number: 309604 )
- 15 ml tubes; Falcon® centrifuge tubes (Corning, Falcon®, catalog number: 352196 )
- 0.45 µm filters (EMD Millipore, catalog number: SLHV033RB )
- Plastic funnel (VWR, catalog number: 300009-435 )
- Parafilm (Bemis, catalog number: PM992 )
- Aluminum foil (VWR, catalog number: 89107-724 )
- 20% carbon dioxide balance nitrogen certified standard mixture, size 300 cylinder, CGA-580 (Airgas, catalog number: X02NI80C3003240 )
- Nitric acid; 69.0-70.0% (Avantor Performance Materials, J.T. Baker, catalog number: 9598-00 )
- Ethanol (VWR, catalog number: 200057-586 )
- De-ionized water (DI)
- Ice and coolers (for shipping)
- 10 ml syringes; Luer-Lok® syringes (BD, catalog number: 309604 )
- Separation of solid-phase substrate for geochemical and microbial analysis
- 50 ml tubes; Falcon® centrifuge tubes (Corning, Falcon®, catalog number: 352098 )
- 20% carbon dioxide balance nitrogen certified standard mixture, size 300 cylinder, CGA-580 (Airgas, catalog number: X02NI80C3003240 )
- Ethanol (VWR, catalog number: 200057-586)
- 50 ml tubes; Falcon® centrifuge tubes (Corning, Falcon®, catalog number: 352098 )
Equipment
- Custom column design and construction (materials needed per column)
- Clear PVC pipe with a height of 52’’ (1.32 m) inner diameter (ID) 6” (0.15 m) (Figure 1)
- Nylon mesh bags (Phifer 48 in x 25 ft. BetterVue Screen) (Home Depot Product Authority, catalog number: 3027671 ) (Figure 4E)
- Impulse heat sealer (Packco, Midwest Pacific, model: MP-12 )
- Tygon tubing for influent and effluent; ¼” ID and 3/8”OD (VWR, catalog number: 89403-862 )
- Pump tubing for peristaltic pump (4T [size], Blue and White Industry)
- Glass marbles (~16 mm diameter) filled the bottom 10 cm as an inert porous bed support. Alternatively large glass beads would also suffice
- Fittings (one entry for each type used):
- Column bottom fittings
- 3-way valve for effluent sample (Figure 4A)
- Valve for liquid sampling from side port (Figure 4B) (5 per column)
- Solid sample port (Figures 4C and 4D) (3 per column)

Figure 4. Sample ports for liquid and solid substrate retrieval. A. 3-way valve to sample effluent; B. Intermediate liquid sampling port; C. Position of intermediate solid-phase sampling port in conjunction with liquid sampling ports; D. Side profile of a solid-phase port before substrate was added to columns, bags were lined up in the port adjacent to each other; E. Sacrificial sample bag containing solid-phase substrate utilized in experiments. - PVC sample ports allowing for flow through and sample retrieval (Figure 4D) which can then be packed with sacrificial bags containing solid-phase substrate (Figure 4E)
- Pump (Flex Flow, max feed 2.3 GFD, Blue and White Industry) (one per column)
- Feed tank (250 gal HDPE drum) one tank for all columns.
- Effluent collection tanks (30 gal HDPE drum)
- Custom welded frame (52” height) to hold columns and enable pivoting for sampling (Figures 5A and 4B).

Figure 5. Pilot scale deployment of apparatus detailing. A. Column frame before columns; B. Column frame after columns and plumbing were established. - Column head space gas collection system (to scrub released sulfide to prevent unsafe amounts of sulfide accumulating)
- 250 ml filter flask stopper No. 6 (Kimax Chase Life Science and Research Products, catalog number: 27060 )
- FEP gas bag 6 x 6 on/off (Labpure) (Saint-GoBain, catalog number: D1075002-10 )
- Clear PVC pipe with a height of 52’’ (1.32 m) inner diameter (ID) 6” (0.15 m) (Figure 1)
- Anoxic columns sampling of aqueous and solid-phase samples
- Heavy-Duty Single-Stage Gas Regulator (VWR, catalog number: 55850-277 )
- PVC hose ¾ in ID (VWR, catalog number: 89068-590 )
- Write-On bags (Nasco, Whirl-Pak®, catalog number: B01196WA )
- Vacuum sealer (Manufacturer Rival, model: FSFGSL0150-015 )
- Vacuum bags (Seal-A-Meal [11-Inch by 9-Foot Rolls, 2pk])
- Dissecting forceps; VWR® dissecting forceps, fine tip, curved (VWR, catalog number: 82027-406 )
- Needle nose multi-tool; Multi-Plier® 600 Needlenose Pliers, Gerber® (Gerber Gear, catalog number: 47550N )
- Bic Classic lighters
- Sharpie® permanent ink pen (VWR, catalog number: 500020-888 )
- Spray bottle to sterilize instruments with 70% ethanol (VWR, catalog number: 23609-182 )
- Heavy-Duty Single-Stage Gas Regulator (VWR, catalog number: 55850-277 )
- Separation of solid-phase substrate for geochemical and microbial analysis
- Anaerobic chamber (Sheldon Manufacturing, model: Bactron Anaerobic/Environmental chamber )
- Scale (OHAUS, model: ES 100 L )
- Large weigh boats (VWR, catalog number: 10803-168 )
- Scissors; VWR® dissecting scissors, sharp tip, 4½" (VWR, catalog number: 82027-578 )
- Needle nose multi-tool; Multi-Plier® 600 Needlenose Pliers, Gerber® (Gerber Gear, catalog number: 47550N )
- Dissecting forceps; VWR® dissecting forceps, fine tip, curved (VWR, catalog number: 82027-406)
- Spray bottle (VWR, catalog number: 23609-182)
- Anaerobic chamber (Sheldon Manufacturing, model: Bactron Anaerobic/Environmental chamber )
Procedure
- Sample bag design for non-invasive retrieval of representative solid-phase substrate: Permeable substrate bags were deployed in each of the 3 solid-phase ports (Figure 1 and Figures 4D and 4E) to facilitate spatial and temporal inquiry. After a sample bag was retrieved it was replaced with an identical bag to circumvent any disturbance to column operation between time points.
- Determine the mass of material needed for analyses, and ensure sample bags contain at least that amount. In this case 3 inch by 3 inch bags are sufficient for the 1 g of material used for digests and 0.5 g x 3 for DNA extraction. The remaining material was anaerobically archived in Whirl-Paks in the anaerobic chamber, sealed in a secondary vacuum evacuated bag and then stored at -20 °C.
- Pack material homogenously in porous nylon bags (i.e., mosquito netting or window screen) and ensure the sample material is consistent with the rest of the column material. Four bags are put in each sample port at the beginning of the experiment.
- Record dry mass of each of the sample bags.
- Prepare extra bags for replacing harvested samples during experimental operation in order to minimize resultant flow perturbations.
- Pre-soak bags in experimentally-relevant water (in this case MIW) before packing them into sample ports to account for expansion, record wet mass.
- Determine the mass of material needed for analyses, and ensure sample bags contain at least that amount. In this case 3 inch by 3 inch bags are sufficient for the 1 g of material used for digests and 0.5 g x 3 for DNA extraction. The remaining material was anaerobically archived in Whirl-Paks in the anaerobic chamber, sealed in a secondary vacuum evacuated bag and then stored at -20 °C.
- Liquid sample retrieval for aqueous metal analysis using Inductively Coupled Plasma (ICP-AES) Spectroscopy with a dual view PerkinElmer Optima Model 5300 spectrometer.
- At least 2 people are needed for sample retrieval.
- Ensure pumps are turned off and liquid is flowing freely to avoid pulling a vacuum.
- Harvest liquid samples before substrate to circumvent any flow effects.
- Flush a 10 ml syringe with N2/CO2 gas mix 3 times, letting the gas push the plunger up.
- Attach funnel to tubing to ensure gas is dispersed over the area of concern.
- Continually flush a 15 ml conical with N2/CO2 mix while collecting 10 ml of liquid with a flushed syringe.
- Attach 0.45 µm filter to the syringe containing the sample and dispense the filtered sample into the 15 ml tube while it is being flushed.
- For ICP-AES analysis lower the pH of the sample to 2 with nitric acid (in our case, several drops of 70% nitric acid).
- Screw on the cap and Parafilm over lid for transportation.
- At least 2 people are needed for sample retrieval.
- Liquid collection for geochemical analyses
- Flush a syringe three times with the gas mix.
- Flush 50 ml tubes while collecting liquid with the flushed syringe.
- Fill 50 ml tube allowing no headspace.
- Screw on lid and Parafilm for transportation.
- Wrap in aluminum foil to prevent photooxidation of sulfides if appropriate.
- Tubes contained ~3 ml of gases headspace to prevent cracking during freezing.
- Run these samples as soon as possible (within 48 h).
- Flush a syringe three times with the gas mix.
- Solid substrate retrieval for geochemical and molecular analyses
- Ensure influent water flow is turned off.
- Tilt the column using the hinged rack assembly so that it is parallel to the ground in order to sample substrate bags without substantial fluid loss (Figure 6).

Figure 6. Intact columns and supporting hardware in use during sampling process. One of the columns is being tilted horizontally for sample bag retrieval to enable analysis during the experiment. - Have one person hold the gas line over the sample port before it is open.
- The second person will open the sample port, and using flame sterilized forceps/pliers retrieve the sample bag.
- Forceps, pliers, and other sample handling tools were flame sterilized by being sprayed with 70% ethanol and then lit with a handheld lighter (i.e., Bic) until the alcohol burns off.
- Place the sample bag into a pre-flushed Whirl-Pak®.
- Push out most of the air and ‘whirl’ the bag around the wire per instructions.
- Place Whirl-Pak-ed sample into vacuum bag and seal using vacuum sealer.
- Put the anoxically sealed sample on ice immediately for molecular analyses.
- Replace the harvested sample with a marked extra sample bag as a placeholder.
- Replacement bag should not be collected as a sample, as it would not have been in contact with the reactive substrate as long as the other sample bags.
- Ensure influent water flow is turned off.
- Separation of substrate samples for geochemical and molecular analyses
- Bring defrosted solid-phase samples into the anaerobic chamber (95%:5% N2:H2) with ethanol spray bottle, 50 ml tubes, 15 ml tubes, forceps, pliers, scissors, weigh boats, and scale.
- Loosen 50 ml and 15 ml tube tops before putting them in the evacuation unit so the oxygen inside the tube is also purged during evacuation.
- Use sterilized scissors inside the anaerobic chamber to cut open vacuum seal bags.
- Remove sample bag from Whirl-Pak®, keeping the Whirl-Pak® intact.
- Cut open nylon sample bag with sterilized scissors.
- Put substrate in large disposable weigh boat and homogenize by mixing with forceps.
- Retrieve substrate from sample bag for DNA extraction with sterilized forceps/pliers. Place the substrate inside a pre-tared 50 ml tube, until there is 5 g wet weight in the tube. While only 0.5 g x 3 was used for DNA extraction, collecting 5 g allows for redundancy.
- Set aside DNA extraction tube, and collect 1 g of material for geochemical extractions and place in pre-tared 15 ml tube.
- Seal the 15 ml tube with Parafilm.
- Replace the remaining material back in the Whirl-Pak®.
- Vacuum seal the Whirl-Pak® after coming out of the anaerobic chamber and archive remaining material at -20 °C for extra analyses etc.
- Freeze material for molecular analyses at -20 °C, unless performed immediately.
- Bring defrosted solid-phase samples into the anaerobic chamber (95%:5% N2:H2) with ethanol spray bottle, 50 ml tubes, 15 ml tubes, forceps, pliers, scissors, weigh boats, and scale.
Data analysis
Data collection and analysis encompassing microbial and geochemical properties was performed as described in (Zhou, 1996; Drennan et al., 2016). Reactors were characterized by a temporally stable community structure (as discerned using the 16S rRNA gene) comprised of a rich organoheterotrophic community that maintained a syntrophic relationship with sulfate reducing bacteria. Columns with greater than 10% alfalfa hay were characterized as having better zinc removal than columns dominated with woodchips. These two substrates selected for significantly different bacterial communities from one another (Drennan et al., 2016).
Notes
- Liquid should be sampled before solid substrate is collected to circumvent flow perturbations resulting from pulling reactive material.
- We advocate for an excess of 3 pore volumes of flow between sampling events, as this is a potentially disruptive sampling protocol.
- Genomic DNA harvested from the solid substrate was extracted and amplified in triplicate to minimize complications associated with sample heterogeneity.
- In some cases metal concentrations of interest exceeded the limits of the ICP-AES (particularly for mine water influent). When this occurred, samples were diluted with DI (i.e., 1:10) to bring the sample within detection range. An analogous approach was taken to counter complications with the high sodium concentration from the sodium acetate extraction step on ICP-AES analyses (Carlsson et al., 2002).
Acknowledgments
This material was based in part through work supported by the U.S. National Science Foundation (CBET-1055396) and the Office of Biological and Environmental Research in the U.S. Department of Energy (DE-SC0006997 and DE-SC0016451). Additional financial support was provided by Freeport-McMoRan Inc. and the J. Gust. Richert Memorial Fund (PIAH/12:57). RA was supported through a Marie Curie International Outgoing Fellowship (PIOF-GA-2012-328397) within the 7th European Community Framework Programme and The Carl Trygger Foundation for Scientific Research (CTS 12:11).
References
- Carlsson, E., Thunberg, J., Öhlander, B. and Holmström, H. (2002). Sequential extraction of sulfide-rich tailings remediated by the application of till cover, Kristineberg mine, northern Sweden. Sci Total Environ 299(1-3): 207-226.
- Drennan, D. M., Almstrand, R., Lee, I., Landkamer, L., Figueroa, L. and Sharp, J. O. (2016). Organoheterotrophic bacterial abundance associates with zinc removal in lignocellulose-based sulfate-reducing systems. Environ Sci Technol 50(1): 378-387.
- Neculita, C. M., Zagury, G. J. and Bussière, B. (2008). Effectiveness of sulfate-reducing passive bioreactors for treating highly contaminated acid mine drainage: I. Effect of hydraulic retention time. App Geochem 23(12): 3442-3451.
- Wildeman, T. R., Updegraff, D. M., Reynolds, J. S. and Bolis, J. L. (1994). Passive bioremediation of metals from water using reactors or constructed wetlands. In: Jeffrey, L. M. and Robert, E. H. (Eds.). Emerging Technology for Bioremediation of Metals. Lewis, pp:13-25.
- Zhou, J., Bruns, M. A. and Tiedje, J. M. (1996). DNA recovery from soils of diverse composition. Appl Environ Microbiol 62(2): 316-322.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Drennan, D. M., Almstrand, R., Lee, I., Landkamer, L., Figueroa, L. and Sharp, J. O. (2017). Pilot-scale Columns Equipped with Aqueous and Solid-phase Sampling Ports Enable Geochemical and Molecular Microbial Investigations of Anoxic Biological Processes. Bio-protocol 7(1): e2083. DOI: 10.21769/BioProtoc.2083.
Category
Microbiology > Microbial biochemistry > Other compound
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










