- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Cryopreservation Protocol for Chlamydomonas reinhardtii
Published: Vol 6, Iss 22, Nov 20, 2016 DOI: 10.21769/BioProtoc.2024 Views: 10229
Reviewed by: Maria SinetovaAgnieszka ZienkiewiczAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Determination of Reduced and Total Glutathione Content in Extremophilic Microalga Galdieria phlegrea
Giovanna Salbitani [...] Simona Carfagna
Jul 5, 2017 22472 Views

Production and Bioassay of a Diffusible Factor that Induces Gametophyte-to-Sporophyte Developmental Reprogramming in the Brown Alga Ectocarpus
Haiqin Yao [...] J. Mark Cock
Sep 20, 2020 4638 Views
Flow Cytometry-based Measurement of Reactive Oxygen Species in Cyanobacteria
Soumila Mondal and Shailendra P. Singh
May 20, 2022 3836 Views
Abstract
Cryopreservation is commonly used for storing viable cells, tissues, organs or organisms at ultralow temperatures, and usually involves immersion in liquid nitrogen at -196 °C. Here we provide a detailed cryopreservation protocol for C. reinhardtii based on Crutchfield’s work (Crutchfield et al., 1999), with minor changes (Yang and Li, 2016). In this study, we compared the cryoprotection effect of two common cryopreservation agents (CPAs), methanol and DMSO. Furthermore, the two-step cryopreservation process was divided into five stages to study the factors affecting the survival rate at each stage. We found that the use of methanol as the CPA, combined with the cooling process outlined here (cooling from 25 °C to -55 °C at a rate of 1 °C/min), were indispensable for cell survival after cryopreservation. The thawing process described here (thawing at 35 °C for 5 min) was also important for increasing the survival rate.
Background
Nowadays, cryopreservation is used frequently for the storage of transgenic lines or mutation lines of C. reinhardtii, and for experimental needs involving this organism. Morris et al. (1979) discussed the effects of different CPAs in cooling, the relationships between temperature and survival rate with or without the CPAs, and the cooling rate. They found that with the addition of methanol the half-lethal temperature was the lowest of all the CPAs tested (-14.4 °C), while that of DMSO was -4.9 °C (Morris et al., 1979).
Through storage in 7% (v/v) DMSO overnight at room temperature, followed by storage at -70 °C, Johnson and Dutcher (1993) gained the highest viabilities, nearly 10% in C. reinhardtii cultures. However, the survival rate may be restricted in C. reinhardtii cell lines, especially in the cell line CC-125 we used, with viabilities of only 0.34%, although the authors claim was caused by liquid culturing. Nevertheless, this method was time consuming and resulted in low cell viabilities. Crutchfield et al. (1999) reported a two-step cooling procedure for the cryopreservation of C. reinhardtii using 5% methanol as the CPA, which retained relatively high viability (> 40%).
Three different methods for improving survival rate were compared in this study. We followed the protocols mentioned above, and made detailed analyses at each stage of the cryopreservation process. The different effects of methanol and DMSO are also discussed, the results agreeing with the work of previous authors.
Materials and Reagents
- 1.5 ml and 4 ml Eppendorf tubes
- 50 ml flask
- Cryovials (VWR, Nalgene®, catalog number: 5000 )
- Filter membrane (Filter pore size: 0.2-0.3 μm) for tissue culture
- 0.22 μm membrane filters (EMD Millipore, model: SLGV033RB )
- Chlamydomonas reinhardtii CC125 was purchased from the Chlamydomonas Resource Center at the University of Minnesota (http://www.chlamycollection.org/cart/)
- TAP medium (Gorman and Levine, 1965, http://www.chlamycollection.org/methods/media-recipes/tap-and-tris-minimal/)
- Isopropanol, purity ≥ 99.5% (Sangon Biotech, catalog number: A507048 )
- Liquid nitrogen
- Trypan blue (Sigma-Aldrich, catalog number: T6146 )
- Methanol (MeOH) (AR), purity ≥ 99% (Chongqing Chuandong Chemical, catalog number: methanol)
- Dimethyl sulfoxide (DMSO) (Sangon Biotech, catalog number: A503039 )
- Iodine (AR) (Sangon Biotech, catalog number: A500538 )
- Potassium iodide (AR) (Sangon Biotech, catalog number: A100512 )
- Lugol’s iodine solution (see Recipes)
- CPA stock solution (see Recipes)
Equipment
- Thermostatic rocking incubator (Shanghai Shipping, model: SPH-211B )
- Hemocytometer (Qiujing)
- Freezing container (Cryo 1 °C freezing container) (VWR, Nalgene®, model: 5100 )
- Autoclave
- Laminar flow hood
- Microscope (OLYMPUS, model: CX31 )
- Water bath (Amersham Bioscience)
- Portable liquid nitrogen tank
- Centrifuge (Hettich, model: D-78532 Tuttlingen)
Note: This equipment has been discontinued, other types with the same centrifugal force can be used as substitutions. - -80 °C freezer (Thermo Fisher Scientific, model: 8607 )
Software
- Microsoft Office Excel
Procedure
- Cell culture conditions
- Cell culture is grown using 20 ml of TAP medium in a 50 ml flask. 1 ml of stationary-phase cell culture is transferred into 20 ml fresh medium every week. The culture conditions are adjusted to a speed of 120 rpm shaking at 25 °C using a Thermostatic rocking incubator, and the culture is illuminated continuously with white fluorescent light (PAR = 30 μmol photons m-2 s-1). Cultures at a cell density of approximately 0.9-2.0 x 107 ml-1 (3-5 days after transfer, late linear phase) are used for the experiment.
- Cell culture is grown using 20 ml of TAP medium in a 50 ml flask. 1 ml of stationary-phase cell culture is transferred into 20 ml fresh medium every week. The culture conditions are adjusted to a speed of 120 rpm shaking at 25 °C using a Thermostatic rocking incubator, and the culture is illuminated continuously with white fluorescent light (PAR = 30 μmol photons m-2 s-1). Cultures at a cell density of approximately 0.9-2.0 x 107 ml-1 (3-5 days after transfer, late linear phase) are used for the experiment.
- Calculating the cell density of C. reinhardtii and volume of cell culture needed in a 2 ml cryovial
- 3-5 d after transfer, take 1 ml cell culture out of the 50 ml flask for cell density estimating.
- Measure cell density using a hemocytometer: mix 10% volume (10 μl) of Lugol’s iodine solution with 90 μl cell culture (diluted if necessary) and add 8 μl of the mixture to each chamber.
- Count five small squares in each chamber as a means of calculating the mean number of cells per chamber. The cell number per microliter = mean numbers of cells in five small squares x 5 x 104 x dilution factor (10/9, if not further diluted). Optimally, the number of cells should be approximately 0.9-2.0 x 107 cells per ml.
Note: Steps B2 and B3 can be substituted by other cell counting methods. - Calculate the volume of cell culture needed in a 2 ml cryovial and confirm that the final density in the cryovial is no more than 3.3 x 106 cells per ml (it was exactly 3.3 x 106 in our experiment). It has been reported that when the final cell density in a cryovial is more than 3.3 x 106 cells per ml, the survival rate after cryopreservation decreases significantly (Piasecki et al., 2009). Cell culture volume in 2 ml cryovial = 3.3 x 106 cells per ml x 1.8 ml/the number of cells per ml. Supplement with TAP medium at: volume of TAP medium = 0.9 ml-cell culture volume.
- 3-5 d after transfer, take 1 ml cell culture out of the 50 ml flask for cell density estimating.
- Preparation and conditions for the cryopreservation process
- Precool the CPA stock solution to 4 °C.
- Put 250 ml isopropanol in the lower department of the two-compartment freezing container, precool it to 4 °C (chill for more than 30 min).
- Sterilize all the equipment and tubes used by ultraviolet sterilization and autoclave.
- Carry out all further operations for the further cultivation of C. reinhardtii cells in a clean and sterile environment (we suggest using a laminar flow hood) to avoid contamination.
- Avoid exposing the CPA (or culture containing CPA) to bright light, because methanol becomes toxic to cells after illumination (Crutchfield et al., 1999).
- Precool the CPA stock solution to 4 °C.
- The cryopreservation process
- The cryopreservation process was based on Crutchfield’s work (Crutchfield et al., 1999), with minor changes. We divided the process into five stages, shown in Figure 1. The treatment 4 CC’LTR in Figure 1 is a typical cryopreservation process in this field.
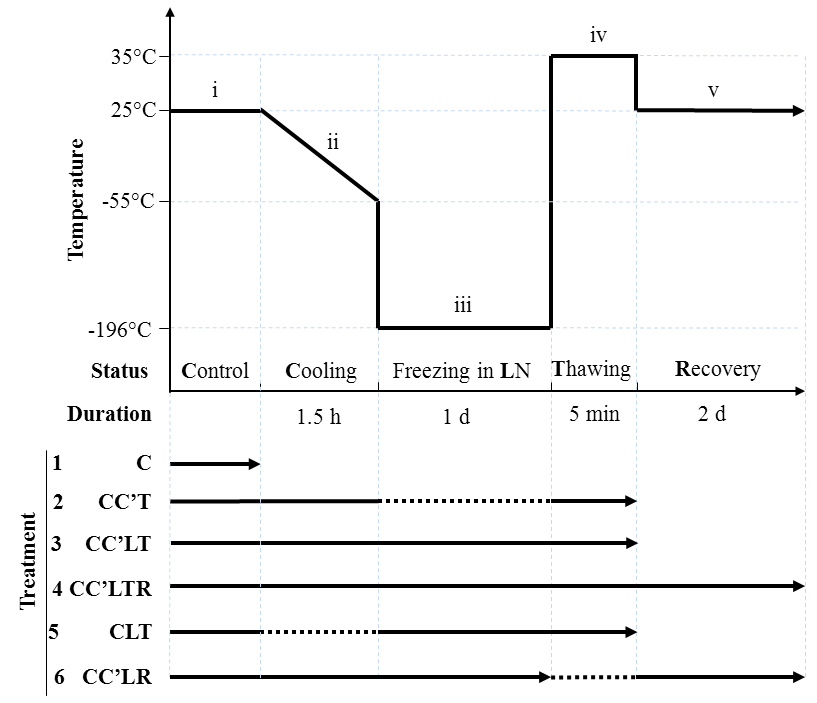
Figure 1. Cryogenic treatments and cryopreservation processes of C. reinhardtii. The process was divided into five stages: (i) Control (normal growth) (C); (ii) Cooling from 25 °C to -55 °C at a rate of 1 °C/min (C’); (iii) Freezing in liquid nitrogen (L); (iv) Thawing at 35 °C for 5 min (T); and (v) Recovery growth for 2 days under normal growth conditions (R). The processes that the organism underwent in each treatment is indicated by a solid line; for an omitted step, a dotted line is used. There were six different treatment combinations: (1) C: Control (normal growth) only; (2) CC’T: Control, Cooling and Thawing; (3) CC’LT: Control, Cooling, Freezing in liquid nitrogen (LN) and Thawing; (4) CC’LTR: Control, Cooling, Freezing in LN, Thawing and Recovery growth; (5) CLT: Control, Freezing in LN and Thawing; (6) CC’LR: Control, Cooling, Freezing in LN, Incubation at room temperature for 30 min and Recovery growth. (Yang and Li, 2016) - Put 0.9 ml CPA stock solution (we used two other CPA controls to compare with methanol in our experiment, Table 1) and a corresponding volume (see step B4) of cell culture and TAP into a 2 ml cryovial, close the cap, mix gently. The total liquid volume in a 2 ml cryovial should be 1.8 ml and the final cell density 6.6 x 106 cells per ml.
- Put the cryovials into the upper compartment of the freezing container. Close the container and place it in a freezer at -80 °C, leaving it undisturbed for 1.5 h (the ‘Cooling’ phase in Figure 1).
- Remove the cryovials from the freezing container, put them into liquid nitrogen immediately, store for 1 day (‘Freezing in LN’ in Figure 1).
Note: In our cryopreservation practice, C. reinhardtii survived in storage for at least 6 months. - Remove the cryovials from the liquid nitrogen, then quickly transfer them to a water bath at 35 °C for 5 min (‘Thawing’ in Figure 1).
- Centrifuge the cryovials at 1,000 x g for 2 min. Then discard the liquid supernatant, add 1 ml of fresh TAP to the cryovials for culturing (preventing contamination). Loosen the cap of the cryovials, mix their contents twice a day. The culture conditions should be the same as in the Control phase, but without shaking (‘Recovery’ in Figure 1).
- The cryopreservation process was based on Crutchfield’s work (Crutchfield et al., 1999), with minor changes. We divided the process into five stages, shown in Figure 1. The treatment 4 CC’LTR in Figure 1 is a typical cryopreservation process in this field.
- Calculation of survival rate
To assess the integrity of the cell membranes, the Evans blue dye test was used. The viability of cultures after cryopreservation is reflected by the percentage of cells that excluded the Evans blue dye. The method follows Crutchfield et al. (1999) with minor changes.- Mix equal volumes of Evans blue dye (0.1% w/v in water) and cell culture (diluted if needed), leave undisturbed in an Eppendorf tube for 5 min.
- Add about 8 μl mixture to each chamber of a hemocytometer to enable counting under a microscope. At least 200 cells were examined at 400x magnification in a chamber, both chambers were counted independently to determine whether the cells had taken up the Evans blue dye.
- Calculate the survival rate as follows: the ratio of the number of cells that retained their original green color to the total number of cells. Each of the treatments outlined above was performed five times. The survival rate for each treatment (Figure 1) in our experiment is shown in Table 1.
- Mix equal volumes of Evans blue dye (0.1% w/v in water) and cell culture (diluted if needed), leave undisturbed in an Eppendorf tube for 5 min.
Table 1. Survival rate of C. reinhardtii in culture medium, culture medium plus 5% DMSO, and culture medium plus 5% MeOH after cryogenic treatments (Yang and Li, 2016)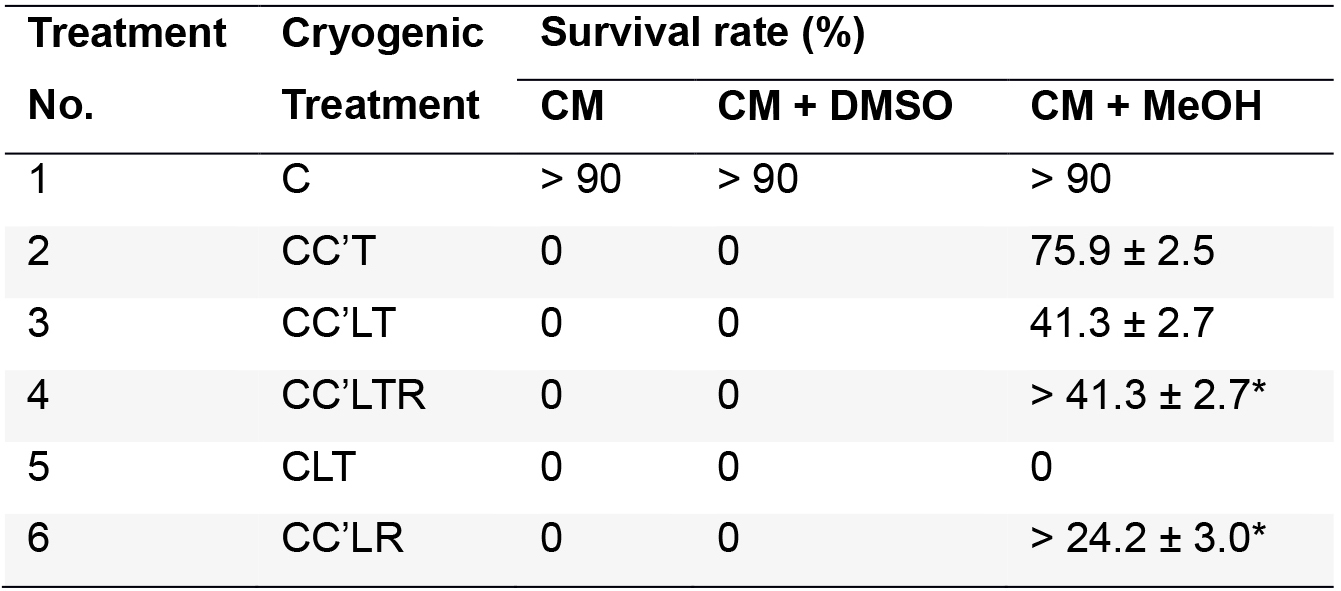
CM, culture medium; CM + DMSO, culture medium plus 5% DMSO; CM + MeOH, culture medium plus 5% MeOH. Values are means ± standard deviation (n = 5). ‘*’ represents those samples in which the survival rate was calculated immediately after thawing without further culturing.
Data analysis
The survival of C. reinhardtii after cryopreservation was assessed by Evan’s blue method (see Procedure E) (Crutchfield et al., 1999). For each treatment (2-6) in Table 1, five samples were counted. The mean and standard deviation was calculated by functions ‘AVERAGE’ and ‘STDEV’ respectively in Microsoft Office Excel.
Notes
- These experiments were repeated three times on different dates (2014.7, 2015.5 and 2015.6), the survival rate in CC’LT treatment being 55.38%, 34.6%, 41.3%, respectively. The second time, the CPAs used had been stored at 4 °C for more than three months, so if fresh CPAs are used, the viability seems to rise. Estimation by eye under a light microscope of whether or not a cell was stained with Evan’s blue will introduce some random errors.
- Additional fluorescent lighting will be needed in the Thermostatic rocking incubator to reach the required illumination intensity.
- Make sure the C. reinhardtii cells are well cultured: the liquid should be yellow-green or green and uniform, running under the microscope, and mainly comprising single cells, with no aggregates. We used the cells 3-5 days after transfer from liquid culture. If cultured from cryopreservation, 2 or more liquid transfers may be necessary.
- Avoid light in the steps with CPAs. The CPAs stocks should be stored at 4 °C, for no longer than 1 month.
- Please handle liquid nitrogen and the -80 °C freezer with care to avoid injury.
Recipes
- Lugol’s iodine solution
1 g I2, 2 g KI, with distilled water added to total volume of 20 ml
Dilute 10-fold when used - CPA stock solution
CPA stock solution: 10% MeOH in TAP medium
Control: TAP only or 10% DMSO in TAP medium
Acknowledgments
This protocol was adapted from previously published study of Crutchfield et al. (1999). Our thanks also go to Matt Laudon, curator of the Chlamydomonas Resource Center, who provided the cell lines. This work was performed by Yang and Li (2016). The author thanks Dr. Jane Marczewski, Zheng Guowei, Chen Hongying for critical reading the protocol. This research was supported by grants from the National Natural Science Foundation of China (31070262), Kunming Institute of Botany (KSCX2-EW-J-24), the Germplasm Bank of Wild Species and the CAS Innovation Program of Kunming Institute (540806321211), as well as the 100-Talents Program of CAS.
References
- Crutchfield, A. L. M., Diller, K. R. and Brand, J. J. (1999). Cryopreservation of Chlamydomonas reinhardtii (Chlorophyta). Eur J Phys 34(1): 43-52.
- Gorman, D. S. and Levine, R. P. (1965). Cytochrome f and plastocyanin: their sequence in the photosynthetic electron transport chain of Chlamydomonas reinhardi. Proc Natl Acad Sci U S A 54(6): 1665-1669.
- Johnson, D. E. and Dutcher, S. K. (1993). A simple, reliable method for prolonged frozen storage of Chlamydomonas. Trends Genet 9(6): 194-195.
- Morris, G. J., Coulson, G. and Clarke, A. (1979). The cryopreservation of Chlamydomonas. Cryobiology 16(4): 401-410.
- Piasecki, B. P., Diller, K. R. and Brand, J. J. (2009). Cryopreservation of Chlamydomonas reinhardtii: a cause of low viability at high cell density. Cryobiology 58(1): 103-109.
- Yang, D. and Li, W. (2016). Methanol-promoted lipid remodelling during cooling sustains cryopreservation survival of Chlamydomonas reinhardtii. PLoS One 11(1): e0146255.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Yang, D. and Li, W. (2016). Cryopreservation Protocol for Chlamydomonas reinhardtii. Bio-protocol 6(22): e2024. DOI: 10.21769/BioProtoc.2024.
- Yang, D. and Li, W. (2016). Methanol-promoted lipid remodelling during cooling sustains cryopreservation survival of Chlamydomonas reinhardtii. PLoS One 11(1): e0146255.
Category
Plant Science > Phycology > Physiology
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










