- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
BODIPY 493/503 Staining of Neutral Lipid Droplets for Microscopy and Quantification by Flow Cytometry
Published: Vol 6, Iss 17, Sep 5, 2016 DOI: 10.21769/BioProtoc.1912 Views: 63389
Reviewed by: Lee-Hwa TaiYong TengVanesa Olivares-Illana

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
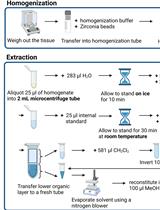
Lipidomics Workflow for Analyzing Lipid Profiles Using Multiple Reaction Monitoring (MRM) in Liver Homogenate of Mice with Non-alcoholic Steatohepatitis (NASH)
Hai Ning Wee [...] Jianhong Ching
Jul 5, 2023 2448 Views
Computational Analysis of Plasma Lipidomics from Mice Fed Standard Chow and Ketogenic Diet
Amy L. Seufert [...] Brooke A. Napier
Sep 20, 2023 2607 Views
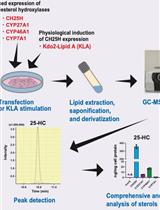
Quantitative Determination of Cholesterol Hydroxylase Specificities by GC–MS/MS in Living Mammalian Cells
Hodaka Saito [...] Yoshio Yamauchi
Jan 20, 2024 2369 Views
Abstract
Lipid droplets (LDs) are ubiquitous, dynamic organelles and function as a storage depot for neutral lipids, including triglycerides and cholesterol esters (Walther and Farese, 2012). The movement of lipid species into and out of LDs impacts a variety of cellular processes, such as energy homeostasis, lipid-based signaling, and membrane homeostasis (Greenberg et al., 2011). For example, neutral lipid storage is enhanced upon increased synthesis or uptake of lipid species. On the other hand, extracellular signals can enhance the release of lipid species packaged within neutral LDs. Thus, the investigation of topics involving lipid metabolism may require the assessment of cellular neutral lipid content. In this protocol, we describe the use of the fluorescent neutral lipid dye 4,4-difluoro-1,3,5,7,8-pentamethyl-4-bora-3a,4a-diaza-s-indacene (BODIPY 493/503) to facilitate quantification of neutral lipid content by flow cytometry and observation of LDs by microscopy.
Keywords: Neutral lipidMaterials and Reagents
- 35 mm cell culture dish/well
- Flow cytometry tube with cell strainer cap (Corning, catalog number: 352235 )
- 15 ml conical tube
- 35 μm filter
- FACS tube
- Glass slides
- Circular cover slip (12 mm #1 circular) (VWR International, catalog number: 101415-528 )
- Cell line of interest
Note: For this protocol, we utilize the A498 clear cell renal cell carcinoma cell line (ATCC, catalog number: A-498 ), but this method can be readily performed with other cell lines. - Oleic acid (OA) (100 mg/ml with 10% BSA in DPBS) (Sigma-Aldrich, catalog number: O3008 )
- Bovine serum albumin (BSA) (10% in DPBS, low endotoxin, fatty acid free, suitable for cell culture, sterile-filtered) (Sigma-Aldrich, catalog number: A1595-50ML )
- 4,4-difluoro-1,3,5,7,8-pentamethyl-4-bora-3a,4a-diaza-s-indacene (BODIPY 493/503) (Thermo Fisher Scientific, Molecular ProbesTM, catalog number: D3922 )
- Dimethyl sulfoxide (DMSO) (Sigma-Aldrich, catalog number: 472301 )
- Phosphate buffered saline (PBS) (Thermo Fisher Scientific, Gibco®, catalog number: 10010031 )
- Trypsin-EDTA (0.25%) with phenol red (Thermo Fisher Scientific, Gibco®, catalog number: 25200-056 )
- HEPES
- NaCl
- CaCl2
- Dulbecco’s modified Eagle’s medium with high glucose and L-glutamine (Thermo Fisher Scientific, Fisher Scientific, catalog number: SH3024301 ) or other medium appropriate for cell line of interest
- Fetal bovine serum (FBS) (Gemini Bio-Products, FoundationTM, catalog number: 900-108 ) or other FBS suitable for cell line of interest
Note: For this protocol, cells were grown in DMEM + 10% FBS. - 4% paraformaldehyde (PFA)
- Prolong® Gold antifade reagent with 4′,6-diamidino-2-phenylindole (DAPI) (Thermo Fisher Scientific, Molecular ProbesTM, catalog number: P36935 )
- Collagen I, rat tail (100 mg) (Corning, catalog number: 354236 )
- 2 μM BODIPY staining solution (see Recipes)
- 10x flow cytometry buffer (see Recipes)
Note: 10x buffer can also be purchased [10x Annexin binding buffer (BD, catalog number: 556454 )].
Equipment
- Forceps
- Flow cytometer equipped with 488 mm laser and filter sets for measuring FITC or GFP (i.e., 533/30)
Note: In this study, a BD Accuri C6 instrument was used.
Procedure
- BODIPY staining for flow cytometry
- Grow cells under culture conditions relevant for the study. A 35 mm dish/well is sufficient for the cell numbers required in this assay. For our assays, 50,000 A498 cells in 35 mm well were sufficient.
- Overnight incubation of cells with 30 μM oleic acid can serve as a positive control for increased neutral lipid content, as oleic acid is a potent inducer of triglyceride synthesis and storage. Fatty acid free BSA serves as a control.
- At the time-point of interest, prepare 2 μM BODIPY staining solution in PBS. The volume of staining solution required for each sample corresponds to the volume of media used for culturing cells.
- Wash cells with a quick rinse using 3 ml PBS to remove media/serum.
- Incubate on BODIPY staining solution in the dark for 15 min at 37 °C. Include an unstained control for flow cytometry.
Note: From this point, protect samples from light as much as possible. - Wash cells with a quick rinse using 3 ml PBS to remove staining solution.
- Trypsinize cells to generate a single cell suspension. For the A498 cell line used in this protocol, cells were incubated with Trypsin-EDTA (0.25%) for 5 min at 37 °C.
- Add 5 ml of PBS and transfer cell suspension to a 15 ml conical tube.
- Pellet cells at 250 x g, 5 min, 4 °C.
- Aspirate supernatant, wash the cell pellet with a quick rinse using 3 ml PBS, and pellet cells at 250 x g, 5 min, 4 °C.
- Carefully aspirate the supernatant and resuspend cells in 300 μl 1x flow cytometry buffer.
- Pass cell suspension through a 35 μm filter into a FACS tube.
- Perform flow cytometry. Obtain a minimum of 10,000 events per condition.
- The investigator can analyze data as mean fluorescence (Figure 1A) or display the data as a histogram (Figure 1B).
- Grow cells under culture conditions relevant for the study. A 35 mm dish/well is sufficient for the cell numbers required in this assay. For our assays, 50,000 A498 cells in 35 mm well were sufficient.
- BODIPY staining for microscopy
- Autoclave coverslips in a glass bottle.
- In the tissue culture hood, place coverslips into 35 mm cell culture dishes.
- Prepare 2 mg/ml collagen solution in PBS.
- Treat the coverslips with collagen to promote cell adherence. Add 3 ml collagen solution to culture dishes and incubate at 37 °C for 30 min.
Note: Use forceps to ensure that coverslips are flush with the bottom of the culture dish, eliminating any air bubbles that may be under the cover slips. - Aspirate the collagen solution.
- Wash with PBS.
- Add PBS to culture dishes and place under UV light in the culture hood to sterilize.
- Plate cells into culture dishes containing the coverslips. The optimal cell number should be determined to achieve confluence of 30-50% at the time of staining to permit proper imaging. For A498 cells used in this protocol, 100,000 cells were plated in 35 mm wells to permit staining at 48 h post plating.
- Incubate under the culture conditions relevant to your experiment.
- For this protocol, A498 cells were incubated in DMEM (high glucose, L-glutamine, sodium pyruvate) supplemented with 10% FBS at 37 °C.
- Overnight incubation of cells with 30 μM oleic acid with BSA can serve as a positive control for increased neutral lipid content, as oleic acid is a potent inducer of triglyceride synthesis and storage. Fatty acid free BSA serves as a control.
- At the time-point of interest, prepare 2 μM BODIPY staining solution in PBS.
- For this protocol, A498 cells were stained 48 h after plating, after an overnight incubation with BSA or BSA + oleic acid.
- Wash cells with 3 ml PBS.
- Incubate on 3 ml staining solution for 15 min at 37 °C.
Note: From this point, protect samples from light as much as possible. - Wash twice in 3 ml PBS.
- Fix cells in 3 ml 4% PFA for 30 min at room temperature.
- Remove 4% PFA.
- Wash samples 3 x 5 min in PBS.
- Use forceps to mount cover slips onto glass slides.
- Add a drop of Prolong® Gold antifade reagent with DAPI onto slide.
- Use forceps to pick up cover slips and place onto the drop of mounting solution, ensuring that the side that side with cells is placed face down onto the mounting solution.
- Allow the mounting solution to cure overnight at room temperature.
- Slides can be stored at 4 °C or imaged immediately (Figure 1C).

Figure 1. Quantification and visualization of intracellular neutral LDs. A. A498 cells were grown overnight in the presence of BSA (0.2%) or 30 μM oleic acid with BSA. The cells were analyzed according to the section titled “BODIPY staining for flow cytometry”. For each sample, mean FL-1 area was normalized to the mean FL-1 area for the BSA treated samples. Note that oleic acid increased FL-1 area. P-value was determined by an unpaired student’s t-test. ***P < 0.001. B. Representative histograms of cells described in (A). C. A498 cells grown on coverslips were treated overnight with BSA (0.2%) or 30 μM oleic acid with BSA. The cells were analyzed according to the section titled “BODIPY staining for microscopy”. Scale bar = 5 μm.
Recipes
- 2 μM BODIPY staining solution
- Prepare 5 mM BODIPY stock solution
Dissolve 1.3 mg BODIPY in 1 ml DMSO and can be stored at -20 °C. - 2 μM BODIPY staining solution can be prepared by diluting stock solution 1:2,500 in PBS.
- Prepare 5 mM BODIPY stock solution
- 10x flow cytometry buffer
0.1 M HEPES (pH 7.4)
1.4 M NaCl
25 mM CaCl2
Notes:- 10x buffer can be stored at 4 °C.
- Dilute to 1x using MilliQ water prior to use.
Acknowledgments
This work was supported by NIH grant 2-P01-CA104838 to M.C.S, and NIH fellowship 5-F30-CA177106 to B.Q.
References
- Greenberg, A. S., Coleman, R. A., Kraemer, F. B., McManaman, J. L., Obin, M. S., Puri, V., Yan, Q. W., Miyoshi, H. and Mashek, D. G. (2011). The role of lipid droplets in metabolic disease in rodents and humans. J Clin Invest 121(6): 2102-2110.
- Walther, T. C. and Farese, R. V., Jr. (2012). Lipid droplets and cellular lipid metabolism. Annu Rev Biochem 81: 687-714.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Qiu, B. and Simon, M. C. (2016). BODIPY 493/503 Staining of Neutral Lipid Droplets for Microscopy and Quantification by Flow Cytometry. Bio-protocol 6(17): e1912. DOI: 10.21769/BioProtoc.1912.
Category
Cell Biology > Cell staining > Lipid
Biochemistry > Lipid > Lipid measurement
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









