- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Quantification of the Mucilage Detachment from Arabidopsis Seeds
Published: Vol 6, Iss 9, May 5, 2016 DOI: 10.21769/BioProtoc.1802 Views: 8893
Reviewed by: Marisa RosaHarrie van ErpFernanda Salvato

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Enzymatic Starch Quantification in Developing Flower Primordia of Sweet Cherry
Nestor Santolaria [...] Afif Hedhly
Apr 5, 2025 1885 Views
New Approach to Detect and Isolate Rhamnogalacturonan-II in Arabidopsis thaliana Seed Mucilage
Dayan Sanhueza and Susana Saez-Aguayo
Sep 5, 2025 1240 Views
Detailed Method for the Purification of Rhamnogalacturonan-I (RG-I) in Arabidopsis thaliana
Liang Zhang [...] Breeanna R. Urbanowicz
Feb 5, 2026 99 Views
Abstract
The Arabidopsis thaliana seed coat produces large amounts of cell wall polysaccharides, which swell out of the epidermal cells upon hydration of the mature dry seeds. While most mucilage polymers immediately diffuse in the surrounding solution, the remaining fraction tightly adheres to the seed, forming a dense gel-like capsule (Macquet et al., 2007). Recent evidence suggests that the adherence of mucilage is mediated by complex interactions between several cell wall components (Griffiths et al., 2014; Voiniciuc et al., 2015a). Therefore, it is important to evaluate how different cell wall mutants impact this mucilage property. This protocol facilitates the analysis of monosaccharides in sequentially extracted mucilage fractions, and quantifies the detachment of each component from seeds.
Keywords: Mucilage adherenceMaterials and Reagents
- 15 ml Falcon tubes (VWR International, catalog number: 734-0452 )
- 2 ml Eppendorf safe-lock tubes (VWR International, catalog number: 211-2165 )
- 2 ml screw-cap tubes (VWR International, catalog number: 211-0093 )
- Chromatography vials with inserts (VWR International, catalog number: 548-0120 )
- Snap cap for chromatography vials (VWR International, catalog number: 548-1151 )
- Manual pipettes tips
- Arabidopsis thaliana seeds
- 2-Deoxy-D-glucose (2-deoxy-Glc) (Sigma-Aldrich, catalog number: D6134-1 G )
- D-(-)-Ribose (Rib) (Sigma-Aldrich, catalog number: R7500-5 G )
- L-(+)-Arabinose (Ara) (Sigma-Aldrich, catalog number: A3256-25 G )
- L-(-)-Fucose (Fuc) (Sigma-Aldrich, catalog number: F2252-5 G )
- D-(+)-Galactose (Gal) (Sigma-Aldrich, catalog number: G0750-25 G )
- D-(+)-Galacturonic acid monohydrate (GalA) (Sigma-Aldrich, catalog number: 48280-5G-F )
- D-(+)-Glucose (Glc) (Sigma-Aldrich, catalog number: G8270-100 G )
- D-Glucuronic acid (GlcA) (Sigma-Aldrich, catalog number: G5269-10 G )
- D-(+)-Mannose (Man) (Sigma-Aldrich, catalog number: M8574-25 G )
- L-Rhamnose monohydrate (Rha) (Sigma-Aldrich, catalog number: R3875-5 G )
- D-(+)-Xylose (Xyl) (Sigma-Aldrich, catalog number: X3877-25 G )
- Ultrapure water (18.2 MΩ cm at 25 °C)
- Sodium hydroxide (NaOH) (VWR International, catalog number: BAKR3727.2500 )
- Trifluoroacetic acid (TFA) (Carl Roth GmbH + Co., catalog number: 6957.1 )
- Sugar standard stocks (see Recipes)
- 9-Sugar mix (see Recipes)
- 2 M TFA (see Recipes)
- 10 mM NaOH (see Recipes)
- 733 mM NaOH (see Recipes)
Equipment
- Autoclave
- Water purification system (Milli-Q or similar style)
- Manual pipettes (Eppendorf AG, Research plus and Repeater Plus style)
- Analytical balance (Mettler-Toledo, model: XSE205DU )
- Ball mill (Retsch GmbH, catalog number: MM400 )
- Two 24 TissueLyser adapters for ball mill (QIAGEN, catalog number: 69982 )
- Lab coat
- Safety glasses
- Chemical resistant gloves (Honeywell, Dermatril, catalog number: 740 , or similar style)
- Fume hood
- Sample concentrator (Bibby Scientific Limited, Techne, catalog number: FSC400D ), equipped with
- Ice machine
- Ion chromatography (IC): Dionex DX-600 system equipped with
- Vortex mixer (Scientific Industries Inc., model: Vortex-Genie 2 , or similar style)
- Benchtop centrifuge (compatible with 2 ml tubes)
- Racks with lids for 2 ml tubes (VWR International, catalog number: 211-0215 )
- Serological pipettes (Thermo Fisher Scientific, Nunc-type or similar style)
- 2 L volumetric flask
- Helium gas tank
Software
- Chromeleon 6.8 chromatography data system software (Thermo Fisher Scientific, Dionex)
- Microsoft Excel with the Real Statistics Resource Pack (http://www.real-statistics.com/)
Procedure
- Plant growth, seed harvest and storage
Since mucilage phenotypes are influenced by how seeds are produced, harvested and stored (see recent review, Voiniciuc et al., 2015c), it is essential to analyze seeds grown under the same conditions. I recommend the procedures described by Voiniciuc and Günl (2016), which yield consistent seed quality and mucilage chemotypes across multiple generations (Voiniciuc et al., 2015a; Voiniciuc et al., 2015b; Voiniciuc et al., 2015c) - Extraction of non-adherent mucilage
- For each experiment (up to 48 samples), prepare a spreadsheet with the relevant fields (e.g., Sample #; Seed Weight; Seed Bag; Genotype). To simplify labeling, assign a two-digit number to each sample (e.g., 01 to 48).
Note: Two sequential extractions will be performed for each sample. - Pre-label the sides of 2 ml Safe Lock tubes with the sample numbers. Use an analytical balance to add 4-6 mg seeds to each tube and record precise weight in the spreadsheet.
- Prepare the standards listed in Table 1, by adding the shown volumes to 2 ml Screw-Cap Tubes.
Table 1. Dilution series of 9-Sugar mix (10 mg/ml) for monosaccharide quantificationLabel S000 S001 S002 S005 S010 S025 S050 S075 S100 S125 µl 0 1 2 5 10 25 50 75 100 125 µg 0 1 2 5 10 25 50 75 100 125 - Prepare enough 30 µg/ml Rib (Internal Standard #1) solution to add 1 ml to all samples and sugar standards in one experiment. For a typical experiment with 48 samples and 10 standards, add 180 µl of the 10 mg/ml Rib and fill up to 60 ml with ultrapure water.
- Using a repeater pipette, add 1 ml of Internal Standard #1 to all samples and standards.
- Manually invert the rack with the sample tubes to suspend all seeds in solution. Place the samples horizontally in a box (large enough to have a single layer of tubes). Shake for 30 min at 200 rpm using an orbital shaker, at room temperature (~24 °C).
Note: This treatment detaches only the non-adherent mucilage from seeds (Figure 1B).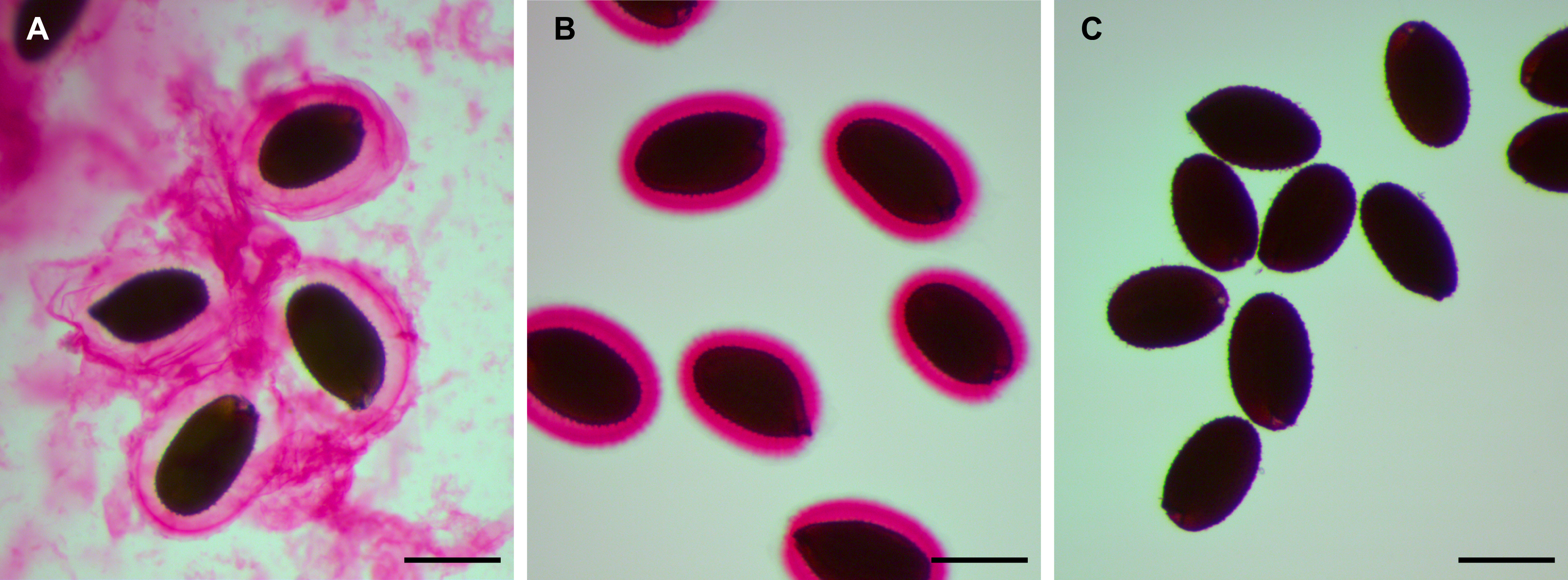
Figure 1. Mucilage staining with ruthenium red (RR) after sequential extractions. Wild-type (Col-0) seeds stained directly in RR, without shaking (A), or after the non-adherent (B) and adherent (C) sequential extractions. Bars = 0.4 mm - Let the seeds settle for at least 30 sec. For each sample, transfer 800 µl of the supernatant to a 2 ml Screw-Cap tube, pre-labeled on its side with the letter N (for non-adherent mucilage extraction) followed by the sample number.
Note: Do not transfer any seeds, and do not cap the tubes at this point. - Evaporate the non-adherent mucilage samples and standards under air flow at 45 °C using the sample concentrator.
- Wash the seeds in the 2 ml Safe Lock tubes with 1 ml water, and discard the solution.
Note: Ensure that no seeds are removed. - Repeat step B9 once. Remove as much of the remaining water as possible, without losing seeds.
Note: The seeds can then be used immediately for the Adherent Mucilage Extraction if a second sample concentrator is available. Alternatively, store the rinsed seeds at -20 °C, and complete the analysis of the non-adherent mucilage samples (drying, hydrolysis, and final elution) before starting the second extraction.
- For each experiment (up to 48 samples), prepare a spreadsheet with the relevant fields (e.g., Sample #; Seed Weight; Seed Bag; Genotype). To simplify labeling, assign a two-digit number to each sample (e.g., 01 to 48).
- Extraction of adherent mucilage
- Prepare a new set of 10 standards in 2 ml Screw-Cap Tubes for the adherent mucilage quantification, by adding the volumes listed in Table 1.
- Prepare enough 30 µg/ml 2-deoxy-Glc solution (Internal Standard #2) to add 1 ml to all samples and sugar standards.
- Using a repeater pipette, add 1 ml of Internal Standard #2 to the new standards, and to the rinsed seeds in Safe Lock tubes (from non-adherent mucilage extraction; completely thawed, if previously frozen).
- Remove the adherent mucilage by essentially performing a total mucilage extraction (Voiniciuc and Günl, 2016). Shake the seed-containing tubes for 15 min at 30 Hz in a ball mill using two 24 TissueLyser Adapters, at room temperature (~24 °C).
- While the seeds are shaking, pre-label the sides of 2 ml Screw-Cap tubes with the letter A (for adherent mucilage extraction) followed by each sample number.
- Rotate block 180° and shake for another 15 min at 30 Hz to finish the mucilage extraction.
Note: This detaches all the adherent mucilage from seeds (Figure 1C). - Let the seeds settle for at least 30 sec. For each sample, transfer 800 µl of the supernatant to the corresponding 2 ml Screw-Cap tube (pre-labeled in step C5).
Note: Do not transfer any seeds, and do not screw the caps on the tubes at this point. - Dry the adherent mucilage samples and the new standards under air flow at 45 °C using the sample concentrator. Process the samples and standards identically from this point onwards.
- Prepare a new set of 10 standards in 2 ml Screw-Cap Tubes for the adherent mucilage quantification, by adding the volumes listed in Table 1.
- Hydrolysis of matrix polysaccharides
Note: The same steps are used for the preparation of both mucilage extracts. Only hydrolyze the two extracts in parallel if there is sufficient space in the Techne Dri-blocks for all samples and standards.- Using a repeater pipette, add 300 µl of 2 M TFA to all tubes.
- Cap each tube tightly, and vortex for 3 sec.
- Transfer tubes to the Techne Dri-block (preheated to 120 °C) and incubate for 60 min.
- Cool heating blocks and tubes on ice. Centrifuge tubes for 30 sec at maximum speed.
- Uncap tubes, and evaporate TFA under air flow at 45 °C using sample concentrator.
Note: Keep caps on a clean paper towel, in the correct order.
- Using a repeater pipette, add 300 µl of 2 M TFA to all tubes.
- Final elution of samples
- Add 600 µl of autoclaved, ultrapure water to all tubes using a repeater pipette. Vortex mix for 3 sec.
Note: If using medium shaking intensity and exercising caution, the tubes do not need to be capped for this step because the solution will not spill over. - Transfer 150 µl from each tube to pre-labeled IC vials with inserts. Seal vials with caps.
- Place tubes in the autosampler of a Dionex DX-600 system in ascending order, but randomize (e.g., using Microsoft Excel) the injection order of the sample and standards.
- Add 600 µl of autoclaved, ultrapure water to all tubes using a repeater pipette. Vortex mix for 3 sec.
- Separation and quantification of monosaccharides
- Perform high-performance anion-exchange chromatography with pulsed amperometric detection (HPAEC-PAD) using CarboPac PA20 Guard and Analytical columns, at 40 °C and a constant flow rate of 0.4 ml/min, essentially as described by Voiniciuc and Günl (2016).
- Ensure that sufficient volumes of the three eluents (ultrapure water, 10 mM NaOH, and 733 mM NaOH) are available to run all the samples and standards for both mucilage extractions.
- Equilibrate the columns for 10 min with 2 mM NaOH (80% water, 20% 10 mM NaOH).
- Inject 10 µl of each sample.
- Separate neutral sugars over 18 min with 2 mM NaOH (80% water, 20% 10 mM NaOH).
- Pump 513 mM NaOH (70% 733 mM NaOH, 30% water) for 7.5 min to separate uronic acids.
- Finally, rinse the column with 733 mM NaOH for 4 min.
Note: Repeat the methods in steps F3-7 for each sample and standard. - Automatically annotate the monosaccharide peaks by configuring the Peak Table and Detection Parameters in the Chromeleon Chromatography Data System software (Dionex) as described by Voiniciuc and Günl (2016).
Note: Minor adjustments are required to annotate the peak of 2-deoxy-Glc (retention time of 2.5 to 3.0 min), and to set it as the internal standard for the adherent mucilage samples and the corresponding standards. - Inspect chromatograms in Chromeleon to ensure that all peaks are correctly labeled.
Note: If necessary, manually correct any peaks that are mislabeled. - All monosaccharide amounts are normalized to the internal standard in each extraction, and quantified as µg based on calibration plots of the standard dilution series (Table 1).
Note: Only trace levels of Rib are detected in the adherent mucilage samples, indicating that two water washes effectively remove carryover of the non-adherent mucilage solution. Therefore, 30 µg Rib could be safely used as the internal standard for the adherent mucilage extracts, eliminating the need to adjust the detection parameters for 2-deoxy-Glc. - Export the amount of monosaccharides in each sample (expressed in µg) to Microsoft Excel for the final calculations.
- Perform high-performance anion-exchange chromatography with pulsed amperometric detection (HPAEC-PAD) using CarboPac PA20 Guard and Analytical columns, at 40 °C and a constant flow rate of 0.4 ml/min, essentially as described by Voiniciuc and Günl (2016).
- Final calculations and statistical analyses
The amounts of monosaccharides detected in the sequential extracts can be expressed as absolute levels (Figure 2A), or as the relative detachment of each sugar from the seed (Figure 2B).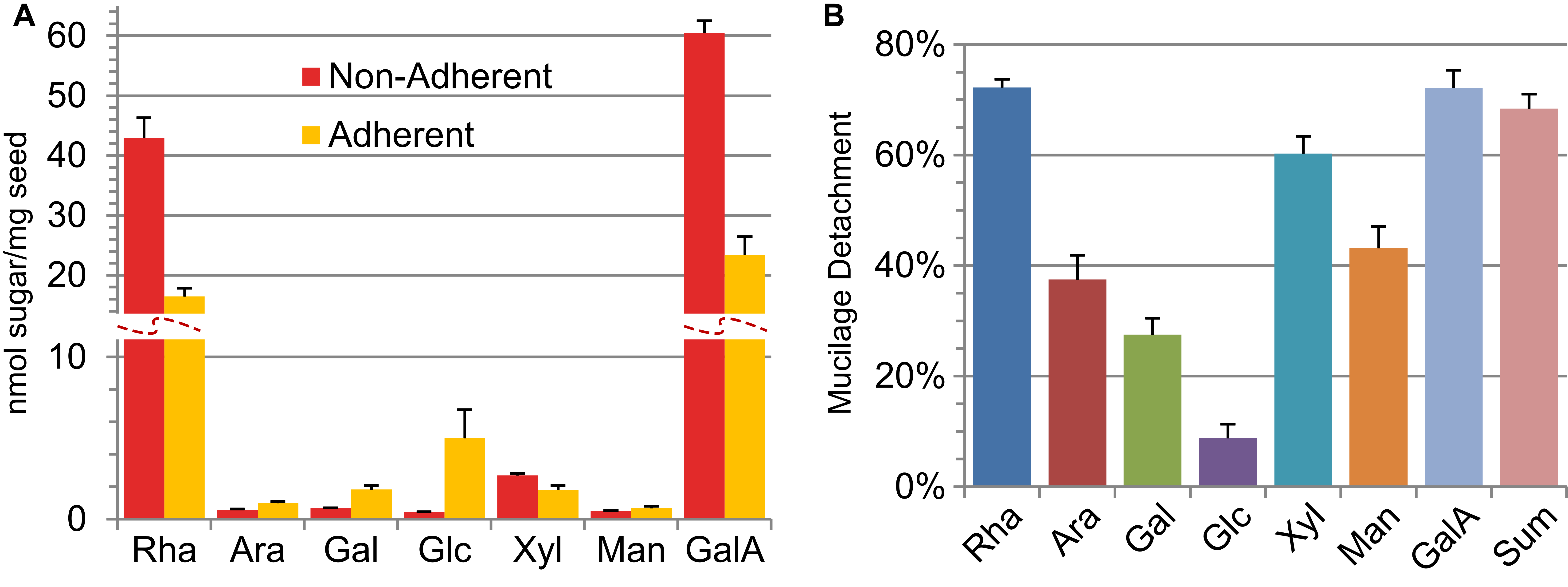
Figure 2. Quantification of mucilage detachment based on sequential extractions. A. Absolute amounts of mucilage sugars in the non-adherent and adherent extracts. B. The detachment of individual mucilage sugars or their combined sum, expressed as a percent of the total amount detected in two sequential extracts. Graphs show means of + SD of five biological replicates for previously analyzed wild type (Col-0) seeds (Voiniciuc et al., 2015b).- Sugar amounts (expressed in µg) can be divided by the amount of seeds (expressed in mg) used for each mucilage extraction, to calculate the absolute composition (µg sugar/mg seed).
Note: Perform this calculation separately for the non-adherent and adherent extractions. - Absolute monosaccharide levels can also be expressed as the number of molecules normalized to the amount of seeds used. To calculate the nmol sugar per mg seed, divide the value obtained in step G1 by the molecular mass of the respective sugar (Table 2), and multiply the result by 1,000.
Note: Perform this calculation separately for each extraction (Figure 2A).
Table 2. Molar mass of the monosaccharides for final calculationsSugar Fuc Rha Ara Gal Glc Xyl Man GalA GlcA Mass 164.16 164.16 150.13 180.16 180.16 150.13 180.16 194.14 194.14 - The relative detachment of each mucilage component (expressed as a percentage) is equal to its non-adherent amount divided by the sum of that particular monomer in the non-adherent and adherent fractions. The overall detachment of seed mucilage is the ratio of non-adherent sugars to the total amount of sugars sequentially extracted (based on values calculated in step G1).
- Statistical analyses to compare the mucilage detachment of wild-type and mutants can be performed in Excel. The absolute amount of particular monosaccharide in a mucilage fraction, or its relative detachment from the seed can be compared between two genotypes with multiple biological replicates using the Excel built-in T.TEST function. When many mutants (and their various sugars components) are compared to wild-type, conditional formatting (e.g., highlight cells with P-value < 0.05) can be used to quickly reveal which components are significantly altered. Two-Factor Analysis of Variance (ANOVA) can also be performed in Excel with the Real Statistics Resource Pack (http://www.real-statistics.com/) to evaluate how mucilage detachment is affected by two independent mutations (via the analysis of wild-type, single and double mutant samples).
Note: In several cell wall mutants, affecting genes with distinct functions, around 90% of Rha and GalA molecules (the most abundant sugars in mucilage) are non-adherent (Voiniciuc et al., 2015a). Although the loss of different genes caused similar increases in mucilage detachment, the precise mechanism that anchors mucilage to the seed surface remains to be elucidated.
- Sugar amounts (expressed in µg) can be divided by the amount of seeds (expressed in mg) used for each mucilage extraction, to calculate the absolute composition (µg sugar/mg seed).
Recipes
- Sugar standard stocks (10 mg/ml)
For each monosaccharide except GalA and Rha, prepare individual stocks by dissolving 100 mg sugar in 10 ml of autoclaved, ultrapure water in a sterile 15 ml Falcon tube.
Since they are sold in monohydrate forms, use 109 mg of GalA and 111 mg of Rha rather than the standard 100 mg.
Aliquot the 2-deoxy-Glc and Rib stocks into 2 ml Safe Lock tubes since they are frequently used.
Store all stocks at -20 °C. - 9-Sugar mix (1 mg/ml)
For a 10 ml solution, mix 1 ml of water with 1 ml of Fuc, Rha, Ara, Gal, Glc, Xyl, Man, GalA, and GlcA (all 10 mg/ml stocks).
Aliquot the 9-Sugar mix into 2 ml Safe Lock tubes and stored at -20 °C. - 2 M Trifluoroacetic acid (TFA)
Prepare 500 ml of a 2 M TFA solution by slowly adding 77 ml of TFA (12.98 M) to 423 ml of ultrapure water
Note: Perform TFA work in a fume hood, with the appropriate personal protective equipment. - 10 mM NaOH (IC eluent)
Use a volumetric flask to transfer 2 L of ultrapure water to the appropriate eluent container
Add 1,040 µl of 50% (w/v) NaOH using a 1 ml serological pipette
Degas eluent using helium for 5 min - 733 mM NaOH (IC eluent)
Use a volumetric flask to transfer 2 L of ultrapure water to the appropriate eluent container
Add a total of 80 ml of 50% (w/v) NaOH using a 50 ml serological pipette
Degas eluent using helium for 5 min
Acknowledgments
The sequential extraction method presented was derived from (Voiniciuc et al., 2015b), and also briefly described by (Voiniciuc et al., 2015a). This work extends the protocol by Voiniciuc and Günl (2016), and was supported by the Natural Sciences and Engineering Research Council of Canada (PGS-D3 grant to C. V.).
References
- Griffiths, J. S., Tsai, A. Y., Xue, H., Voiniciuc, C., Sola, K., Seifert, G. J., Mansfield, S. D. and Haughn, G. W. (2014). SALT-OVERLY SENSITIVE5 mediates Arabidopsis seed coat mucilage adherence and organization through pectins. Plant Physiol 165(3): 991-1004.
- Macquet, A., Ralet, M. C., Kronenberger, J., Marion-Poll, A. and North, H. M. (2007). In situ, chemical and macromolecular study of the composition of Arabidopsis thaliana seed coat mucilage. Plant Cell Physiol 48(7): 984-999.
- Voiniciuc, C. and Günl, M. (2016). Analysis of monosaccharides in total mucilage extractable from Arabidopsis seeds. Bio-protocol 6(9): e1801.
- Voiniciuc, C., Gunl, M., Schmidt, M. H. and Usadel, B. (2015a). Highly branched xylan made by IRREGULAR XYLEM14 and MUCILAGE-RELATED21 links mucilage to Arabidopsis seeds. Plant Physiol 169(4): 2481-2495.
- Voiniciuc, C., Schmidt, M. H., Berger, A., Yang, B., Ebert, B., Scheller, H. V., North, H. M., Usadel, B. and Gunl, M. (2015b). MUCILAGE-RELATED10 produces galactoglucomannan that maintains pectin and cellulose architecture in Arabidopsis seed mucilage. Plant Physiol 169(1): 403-420.
- Voiniciuc, C., Yang, B., Schmidt, M. H., Gunl, M. and Usadel, B. (2015c). Starting to gel: how Arabidopsis seed coat epidermal cells produce specialized secondary cell walls. Int J Mol Sci 16(2): 3452-3473.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Voiniciuc, C. (2016). Quantification of the Mucilage Detachment from Arabidopsis Seeds. Bio-protocol 6(9): e1802. DOI: 10.21769/BioProtoc.1802.
-
Voiniciuc, C., Schmidt, M. H., Berger, A., Yang, B., Ebert, B., Scheller, H. V., North, H. M., Usadel, B. and Gunl, M. (2015b). MUCILAGE-RELATED10 produces galactoglucomannan that maintains pectin and cellulose architecture in Arabidopsis seed mucilage. Plant Physiol 169(1): 403-420.
Category
Plant Science > Plant biochemistry > Carbohydrate
Biochemistry > Carbohydrate > Polysaccharide
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










