- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Rat Model of Intracerebral Hemorrhage Induced by Collagenase IV
Published: Vol 5, Iss 14, Jul 20, 2015 DOI: 10.21769/BioProtoc.1541 Views: 11808
Reviewed by: Soyun KimManuel D. GaheteShai Berlin

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Conditioned Lick Suppression: Assessing Contextual, Cued, and Context-cue Compound Fear Responses Independently of Locomotor Activity in Mice
Youcef Bouchekioua [...] Yu Ohmura
Dec 5, 2022 1641 Views
In situ Microinflammation Detection Using Gold Nanoclusters and a Tissue-clearing Method
Fayrouz Naim [...] Masaaki Murakami
Apr 5, 2023 2670 Views
A One-Step Mouse Model of Parkinson’s Disease Combining rAAV-α-Synuclein and Preformed Fibrils of α-Synuclein
Santhosh Kumar Subramanya [...] Poonam Thakur
Dec 5, 2025 1644 Views
Abstract
Stroke, the second leading cause of death worldwide (Ingall, 2004), is one of the major causes of morbidity and mortality. Intracerebral hemorrhage (ICH), a lethal type of stroke, accounts for 20% of all strokes (Qureshi et al., 2001), and occurs in about 50-60% of Asians (Inagawa, 2002). In order to understand the disease process, three animal model of ICH have been used to study the pathophysiology and treatment of ICH, including the microballoon model, the bacterial collagenase injection model (Rosenberg et al., 1990) and the autologous blood injection model (Andaluz et al., 2002; Belayev et al., 2003). In the collagenase injection model, the hemorrhage size is controllable which was induced by small vessel breakdown. This model also can mimic the onset of spontaneous intraparenchymal bleeding and the expansion of continuous bleeding in ICH patients (Kazui et al., 1996; MacLellan et al., 2008; James et al., 2008). In the past several years, our previous studies have proven that our modified collagenase IV injection model is a reliable and reproducible model of ICH in rat (Lu et al., 2014; Gao et al., 2014; Wang et al., 2011). We hereby introduce our model of ICH as following.
Keywords: Intracerebral hemorrhageMaterials and Reagents
- About 2 months old male Sprague-Dawley rats(280~320 g)
- Isoflurane
- Collagenase IV (Sigma-Aldrich, catalog number: C5138 )
- 4-0 monocryl
Equipment
- Sterile iodophor wipes
- Sterile bone wax
- Induction chamber
- Stereotactic apparatus (KOPF®, model: 900 Small Animal Stereotaxic Instrument)
- Stereotaxic drill (KOPF®, model: 1474 High Speed Stereotaxic Drill)
- The animal gas anesthesia system (RDW Life Science)
- Temperature controller and temperature sensor system (RDW Life Science, model: 69001)
- Microinjection pump (KD Scientific, model: KDS310 )
- Hamilton syringe 26 G (5 μl)
- Magnetic Resonance Imaging (MRI)
- Brain matrix (RDW Life Science, catalog number: 68710 )
Procedure
- The stereotactic apparatus are set up and the weight of rats are recorded before operation (Figure 1).
- The rats are deeply anesthetized with 3% isoflurane in an induction chamber.
- To ensure the depth of anesthesia, the rats are tested by toe pinch.
- After loss of consciousness, the rats are fixed on the stereotactic frame using a nose clamp and two ear bars (Figure 1).
- A temperature sensor is inserted into the rats’ anus, and the temperature of rats is maintained at 37 °C by a temperature controller.
- The rats are kept anesthetized with 2% isoflurane by gas mask.
- After shaving the fur, the superior surface of rat head is prepared by sterile iodophor wipes as surgical field.
- A longitudinal incision is made on the surface of head to expose the bregma.
- Using the stereotactic apparatus, a left point 3 mm lateral to the bregma is marked.
- A burr hole through the skull is drilled at the marked point by a high speed stereotaxic drill.
Note: The Depth of the drilling depended on the experience of the operator. We can assess the damage by Immunohistochemistry.
- 1 μl collagenase type IV (0.25 IU/μl) is injected into corpus striatum (5 mm below the skull) by a 5 μl Hamilton syringe 26 G at a slow rate of 0.2 μl/min.
- The syringe is remained at the place about 7 min after the injection is completed.
- For about 7 min later, the syringe is removed slowly.
- Once the syringe is removed, sterile bone wax is used to plug the hole quickly.
- The skin on the surface of head is closed by using 4-0 monocryl.
- The rats are removed from the stereotactic apparatus and are allowed to recover in a warmed cage with free access to food and water.
Note: The recovery period is about 20-30 min.
- To evaluate the rat Model of ICH induced by collagenase IV, the rats are sacrificed and the brain is cut into slices after operation to assess the volume of hematoma (see Note 3) (Figure 2A and 2B). Furthermore, the neurobehavior (Bederson test) of ICH rats is assessed (Figure 2C-2F). At last, the volume of hematoma also is analysed by MRI (Figure 2G-2H).

Figure 1. A picture representing a set of the stereotactic apparatus system and a rat mounted in the stereotactic frame. A. Stereotactic frame; B. Infusion pump; C. Stereotaxic drill; D. Nose clamp; E. Ear bar; F. Hamilton syringe 26 G.
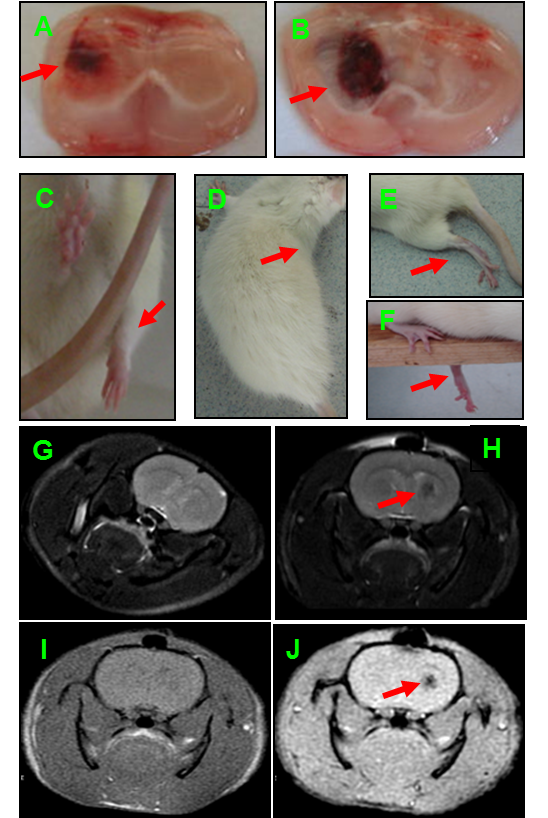
Figure 2. Evaluation of ICH rat induced by collagenase IV. A. The hematoma at 3 h after operation. B. The hematoma at 3 days after operation. C-F. Neurobehavioral test at 3 days after operation, such as, failure to extend contralateral forelimb fully (C), failure to flex the hind legs (E), circling-walking (D), and failure to keep balance on a beam. G-J. The volume of hematoma tested by MRI at 3 days after operation.
Notes
- The concentration of collagenase IV should be exactly 0.25 IU/μl, because a small change in concentration of collagenase IV may cause a different in the volume of the hematoma.
- The stereospecific place collagenase IV injected is 3 mm lateral to the bregma and 5 mm below to skull.
- The brains are sliced by the brain matrix, which can slice the brains about 1 mm thickness. And before sliced, the brains should be freezed at -40 °C about 20 min.
- Bederson test (Bederson et al., 1986) is an evaluation for neurologic status of stroke rats. Rats will be suspended about 1m high by holding the tail, the status of the forelimb flexion will be observed and recorded. Rats that extend both of their forelimbs will be considered as normal or having no observable deficit, standing for grade 0. If any consistent flexion of forelimb occurs, such as only wrist flexion, or shoulder adduction with elbow extension, or even full flexion of wrist and elbow with adduction and internal rotation of the shoulder contralateral to the infarct side, rats will be assigned at least grade 1. If further, rats could not resist the lateral push toward the paretic side after gently providing a lateral pressure behind the shoulder, will be grade 2. Then rats are allowed to move freely. Whether the rats will circle toward the paretic side is the indicator for grade 3. Generally, rats that display circling behavior, being grade 3, have greater neurologic deficit.
Acknowledgments
The study was supported by the grant from the National Natural Science foundation of China (No. 81171088).
References
- Andaluz, N., Zuccarello, M. and Wagner, K. R. (2002). Experimental animal models of intracerebral hemorrhage. Neurosurg Clin N Am 13(3): 385-393.
- Belayev, L., Saul, I., Curbelo, K., Busto, R., Belayev, A., Zhang, Y., Riyamongkol, P., Zhao, W. and Ginsberg, M. D. (2003). Experimental intracerebral hemorrhage in the mouse: histological, behavioral, and hemodynamic characterization of a double-injection model. Stroke 34(9): 2221-2227.
- Bederson, J. B., Pitts, L. H., Tsuji, M., Nishimura, M. C., Davis, R. L. and Bartkowski, H. (1986). Rat middle cerebral artery occlusion: evaluation of the model and development of a neurologic examination. Stroke 17(3): 472-476.
- Gao, L., Lu, Q., Huang, L. J., Ruan, L. H., Yang, J. J., Huang, W. L., ZhuGe, W. S., Zhang, Y. L., Fu, B., Jin, K. L. and ZhuGe, Q. C. (2014). Transplanted neural stem cells modulate regulatory T, gammadelta T cells and corresponding cytokines after intracerebral hemorrhage in rats. Int J Mol Sci 15(3): 4431-4441.
- Inagawa, T. (2002). What are the actual incidence and mortality rates of intracerebral hemorrhage? Neurosurg Rev 25(4): 237-246.
- Ingall, T. (2004). Stroke--incidence, mortality, morbidity and risk. J Insur Med 36(2): 143-152.
- James, M. L., Warner, D. S. and Laskowitz, D. T. (2008). Preclinical models of intracerebral hemorrhage: a translational perspective. Neurocrit Care 9(1): 139-152.
- Kazui, S., Naritomi, H., Yamamoto, H., Sawada, T. and Yamaguchi, T. (1996). Enlargement of spontaneous intracerebral hemorrhage. Incidence and time course. Stroke 27(10): 1783-1787.
- Lu, Q., Gao, L., Huang, L., Ruan, L., Yang, J., Huang, W., Li, Z., Zhang, Y., Jin, K. and Zhuge, Q. (2014). Inhibition of mammalian target of rapamycin improves neurobehavioral deficit and modulates immune response after intracerebral hemorrhage in rat. J Neuroinflammation 11: 44.
- MacLellan, C. L., Silasi, G., Poon, C. C., Edmundson, C. L., Buist, R., Peeling, J. and Colbourne, F. (2008). Intracerebral hemorrhage models in rat: comparing collagenase to blood infusion. J Cereb Blood Flow Metab 28(3): 516-525.
- Qureshi, A. I., Tuhrim, S., Broderick, J. P., Batjer, H. H., Hondo, H. and Hanley, D. F. (2001). Spontaneous intracerebral hemorrhage. N Engl J Med 344(19): 1450-1460.
- Rosenberg, G. A., Mun-Bryce, S., Wesley, M. and Kornfeld, M. (1990). Collagenase-induced intracerebral hemorrhage in rats. Stroke 21(5): 801-807.
- Wang, Z., Cui, C., Li, Q., Zhou, S., Fu, J., Wang, X. and Zhuge, Q. (2011). Intracerebral transplantation of foetal neural stem cells improves brain dysfunction induced by intracerebral haemorrhage stroke in mice. J Cell Mol Med 15(12): 2624-2633.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Lu, Q., Huang, L. and ZhuGe, Q. (2015). A Rat Model of Intracerebral Hemorrhage Induced by Collagenase IV. Bio-protocol 5(14): e1541. DOI: 10.21769/BioProtoc.1541.
Category
Neuroscience > Nervous system disorders > Animal model
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link