- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Isolation of Heterocysts from Anabaena sp. PCC 7120
Published: Vol 5, Iss 8, Apr 20, 2015 DOI: 10.21769/BioProtoc.1456 Views: 15521
Reviewed by: Maria SinetovaRon Saar-DoverAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Induction, Isolation and Counting of Akinetes in Aphanizomenon ovalisporum
Assaf Sukenik [...] Dotan Sela
May 20, 2016 8507 Views
Isolation of Commensal Escherichia coli Strains from Feces of Healthy Laboratory Mice or Rats
Tingting Ju and Benjamin P. Willing
Mar 20, 2018 10906 Views

Detachment Procedure of Bacteria from Atmospheric Particles for Flow-cytometry Counting
Carolina M. Araya [...] Isabel Reche
Jun 20, 2019 5584 Views
Abstract
During combined nitrogen step-down, filaments of cyanobacterium Anabaena sp. PCC 7120 differentiate about 5-10% of vegetative photosynthetic cells into heterocysts, the specialized cells for N2 fixation (Walk, 1996). Heterocysts have a thick cell wall reducing permeation of O2 and consist of two additional layers composed of glycolipids and polysaccharides. The difference in structure and composition of the cell wall between heterocysts and vegetative cells allows separation and isolation of heterocyst. Heterocysts isolated by this protocol can be subjected to protein analysis and activity measurements, which do not require strict anaerobic conditions.
Keywords: HeterocystMaterials and Reagents
- Anabaena sp. PCC 7120
- BG110 growth medium-BG11 medium without addition of combined nitrogen (Rippka et al., 1979)
- 1 M 2-[[1,3-dihydroxy-2-(hydroxymethyl)propan-2-yl]amino]ethanesulfonic acid-KOH (TES) (pH 8.2)
- 1 M 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid-NaOH (Hepes) (pH 7.2)
- 1 M NaCl
- 1 M Na-EDTA
- Sucrose (Merck KGaA, catalog number: K45136551417 )
- Lysozyme (N-acetylmuramide glycanhydrolase) (Roche Diagnostics, catalog number: 10837059001 )
- Extraction buffer (see Recipes)
Equipment
- 50 ml Falcon tubes
- 500 ml centrifuge vials (optional)
- Spectrophotometer (e.g. PerkinElmer, model: Lambda 25 )
- Sorvall GS-3 rotor centrifuge for 500 ml vials (optional)
- SL 16R centrifuge (6 x 50 ml) (e.g. Thermo Fisher Scientific)
- Vortex
- Sonicator (e.g. Labsonic U, B. Braun Melsungen AG)
- Orbital shaker (e.g. Infors HT, Labotron)
- Incubator (e.g. Certomat, B. Braun Melsungen AG)
- Bright-field microscope (40x magnification)
- Water bath or thermostat at 37 °C for Chlorophyll measurements
Procedure
- It is important to cultivate Anabaena sp. PCC 7120 in BG110 medium without combined nitrogen. In our laboratory, cultures are cultivated in BG110 supplemented with 10 mM TES-KOH (pH 8.2) at 30 °C under continuous light intensity of 50 μmol photons m-2 s-1 (PAR) and under agitation (about 120 RPM) to assure continuous gas exchange and kept at high CO2 (air enriched with 3% CO2).
- Harvest filaments during the logarithmic growth phase (OD750 = 1). To obtain 10-30 ml of purified heterocysts at the Chlorophyll (Chl) a concentration of 15-20 μg/ml use about 300-400 ml of cyanobacterial culture.
Note: Chl a quantification, as described in Meeks and Castenholz (1971), is based on the extinction coefficient of 78,74 L/g/cm for Chl a in 90% methanol.

- Centrifuge filaments at 5,000 x g at room temperature for 5-10 min, discard the supernatant. If available, use a centrifuge with the rotor fitting 500 ml tubes or distribute the culture into 50 ml Falcon tubes and perform several rounds of centrifugations. In the end of centrifugation collect all cells in one 50 ml falcon tube and discard as much of growth medium as possible.
- Keep the pellet in falcon tube and add extraction buffer till about 30 ml, vortex thoroughly.

- Add 30 mg of fresh (!) lysozyme into the falcon tube, incubate for 1 h at 37 °C with continuous mixing on orbital shaker (200 RPM).
- Sonicate at 40 W for 1 min in water bath filled with ice-cold water to avoid excessive heating. Lysozyme treatment and sonication will digest vegetative cells, whereas heterocysts will remain intact. After this step all procedures should be performed at 4 °C and on ice.
- Centrifuge cell suspension at 1,000 x g for 5 min (4 °C), discard a supernatant (it can be used as a crude protein extract from vegetative cells).

- Resuspend the pellet in 10-15 ml of ice-cold extraction buffer, vortex thoroughly, centrifuge at 250 x g for 3 min (4 °C) and discard the supernatant. Repeat washing minimum 3 times or until the supernatant is not blue; the residual pellet containing heterocysts should be light-green.

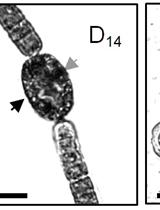
- Check purity of heterocysts by microscope. Take small amount of pellet on a glass slide (optionally resuspend in small amount of extraction buffer) and visualize cells under a microscope (40x magnification). The pellet must be mostly formed by aggregates of intact roundish pale-green heterocysts, in some cells polar granules of cyanophycine might be visible. Alcian blue staining might also be used to confirm the absence of vegetative cells in preparation (Maldener et al., 2003).

Note: Under the microscope vegetative cells and heterocysts can be easily distinguish by the shape of the cells and by the color. Due to the features of the camera, in this photo the color of cells is modified. Small arrows point to the heterocysts in filaments.
Note: If the pellet still contains filaments consisting of several cells, then lysozyme treatment was not successful. In this case repeat lysozyme treatment with a fresh enzyme and continue with sonication.
- Freeze the pellet in liquid nitrogen and store at -80 °C to use later for protein isolation or resuspend in extraction buffer to reach the desirable concentration of Chl a for immediate activity measurements. After freezing heterocysts do not retain the activity.
Recipes
- Extraction buffer
50 mM HEPES-NaOH (pH 7.2)
0.4 M sucrose
10 mM NaCl
10 mM EDTA
Acknowledgments
The protocol was adopted from Ermakova et al. (2014), Fay (1980), Razquin et al. (1996), and Ow et al. (2009). The work was supported by the Academy of Finland (projects 271832) and Kone Foundation.
References
- Ermakova, M., Battchikova, N., Richaud, P., Leino, H., Kosourov, S., Isojarvi, J., Peltier, G., Flores, E., Cournac, L., Allahverdiyeva, Y. and Aro, E. M. (2014). Heterocyst-specific flavodiiron protein Flv3B enables oxic diazotrophic growth of the filamentous cyanobacterium Anabaena sp. PCC 7120. Proc Natl Acad Sci U S A 111(30): 11205-11210.
- Fay, P. (1980). Heterocyst isolation. Methods in Enzymology 69 No. Part C: 801-812.
- Ow, S. Y., Cardona, T., Taton, A., Magnuson, A., Lindblad, P., Stensjo, K. and Wright, P. C. (2008). Quantitative shotgun proteomics of enriched heterocysts from Nostoc sp. PCC 7120 using 8-plex isobaric peptide tags. J Proteome Res 7(4): 1615-1628.
- Razquin, P., Fillat, M. F., Schmitz, S., Stricker, O., Bohme, H., Gomez-Moreno, C. and Peleato, M. L. (1996). Expression of ferredoxin-NADP+ reductase in heterocysts from Anabaena sp. Biochem J 316 (Pt 1): 157-160.
- Rippka, R., Deruelles, J., Waterbury, J. B., Herdman, M. and Stanier, R. Y. (1979). Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111(1): 1-61.
- Maldener, I., Hannus, S. and Kammerer, M. (2003). Description of five mutants of the cyanobacterium Anabaena sp strain PCC 7120 affected in heterocyst differentiation and identification of the transposon-tagged genes. FEMS Microbiol Lett 224(2): 205-213.
- Wolk, C. P. (1996). Heterocyst formation. Annu Rev Genet 30: 59-78.
- Meeks, J. C. and Castenholz, R. W. (1971). Growth and photosynthesis in an extreme thermophile, Synechococcus lividus (Cyanophyta). Arch Mikrobiol 78(1): 25-41.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Ermakova, M. and Allahverdiyeva, Y. (2015). Isolation of Heterocysts from Anabaena sp. PCC 7120. Bio-protocol 5(8): e1456. DOI: 10.21769/BioProtoc.1456.
Category
Microbiology > Microbial cell biology > Cell isolation and culture
Microbiology > Microbial metabolism > Nitrogen fixation
Plant Science > Phycology > Blue-green algae
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









