- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Olfactory Habituation in Fasted Mice
Published: Vol 4, Iss 20, Oct 20, 2014 DOI: 10.21769/BioProtoc.1267 Views: 9770
Reviewed by: Soyun KimAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
The Mouse Social Frailty Index (mSFI): A Standardized Protocol
Charles W. Collinge [...] Alessandro Bartolomucci
Apr 20, 2025 1809 Views
Training Mice to Perform Attentional Set-Shifting Under Head Restraint
Katarina Kalajzic [...] Timothy Spellman
Sep 5, 2025 1470 Views

A Protocol to Assess Time-of-Day-Dependent Learning and Memory in Mice Using the Novel Object Recognition Test
Jordan Mar [...] Isabella Farhy-Tselnicker
Sep 20, 2025 2571 Views
Abstract
Sensory perception is tightly modulated by the individual’s internal states. In particular, it has been shown that olfactory processes are constantly influenced by metabolic signals reflecting the energy status of the body. Thus, it is important to implement novel approaches to evaluate the impact of body energy changes on olfactory performance. Here, we describe a behavioral protocol to accurately evaluate olfactory habituation in fasted mice (Soria-Gomez et al., 2014) using basic equipment that mice are familiar with. Briefly, the mouse is placed in a test cage where it is presented first, an odorless solvent (the control), then an odor A (twice) and finally an odor B. This test relies on the fact that animals present an attenuation of the behavioral response after several presentations of the same olfactory stimulus.
Keywords: Food intakeMaterials and Reagents
- Animals: C57/BL6N male mice (Janvier Labs)
- Mineral oil (Sigma-Aldrich, catalog number: M-5904 )
- Isoamyl acetate (banana odor) (Sigma-Aldrich, catalog number: W205508 )
- Benzaldehyde (almond odor) (Sigma-Aldrich, catalog number: 418099 )
- 20% ethanol
Note: Mineral oil was used as a solvent, because of its odorless property as reported in the literature (Linster et al., 2009; Qiu et al., 2014; Slotnick and Restrepo, 2005; Tan et al., 2010). Almond and banana odors were used as novel, yet neutral odors(Yang et al., 2013; Yang and Crawley, 2009). However, other odors are also suitable to test in our conditions, such as hexanal.
Equipment
- Standard individual plexiglass cage for mice (see Figure 1A)
- Water bottle cap with sipper tube made in stainless steel (from now called Odor holder, see Figure 1B) (Tecniplast)
- Filter paper (Thermo Fisher Scientific, see Figure 1C and Figure 2)
- Opaque eppendorfs (1 ml volume) (Eppendorf, see Figure 1C)
- Scissors
- 10 µl pipet
- 10 µl pipet tips
- Tissue paper
- Standard table to place the test cage
- CCTV camera for an aerial view of the test
- Standard computer (PC or Mac)

Figure 1. Basic material to perform the olfactory habituation test. A) Test cage made in plexiglass (12 cm width x 30 cm length x 13 cm height), with a circular perforation (5 cm diameter) in one of the extremes. B) Odor holder made in stainless steel (5 cm height from the base to the top of the sipper tube; base of 5 cm diameter; the hole of the sipper tube of 3 mm diameter). C) Standard opaque Eppendorf (left) containing the odor solution and the filter paper (3 cm height x 1 cm width) used to administer the odor. This filter paper is introduced in the sipper tube of B).
Software
- Video recording software (GrabBee, grabbee.software.informer.com)
- Software to analyze exploratory behavior (Behav_Scor_v3.0_beta)
Procedure
- Test cage
- A circular perforation (5 cm diameter) is made in the floor in one of the extremes of the plexiglass cage (see Figure 1A). This modification allows the odor holder to be placed between the table and the test cage, keeping it stable and resistant to any pressure made by the mouse.
- A circular perforation (5 cm diameter) is made in the floor in one of the extremes of the plexiglass cage (see Figure 1A). This modification allows the odor holder to be placed between the table and the test cage, keeping it stable and resistant to any pressure made by the mouse.
- Preparation of the odor solution
- A stock solution is prepared by adding 1 µl of the odor, either almond or banana, to 1 ml of mineral oil (0.1% concentration) which is then aliqouted into 50 µl lots/batches in Eppendorf tube.
- All the tubes were sealed using parafilm to prevent evaporation from the tubes and were then stored at room temperature in a cardboard box protected from the light.
- A stock solution is prepared by adding 1 µl of the odor, either almond or banana, to 1 ml of mineral oil (0.1% concentration) which is then aliqouted into 50 µl lots/batches in Eppendorf tube.
- Preparation of the scented filter paper
- The filter paper is cut into small pieces (1 cm x 3 cm, Figure 1C).
- For the olfactory habituation test, 5 µl of the diluted odorant (0.1%) or mineral oil is applied using a pipet to one edge of the pre-cut filter paper, which is then placed inside of the odor holder, introducing only two thirds of the scented filter paper (see Figure 2).
Note: It is important not to introduce completely the filter paper in the holder in order to avoid physical contact (i.e. with the tongue) from the mouse. When this happens there is high probability that the mouse will bite the odor source, making it difficult to score the olfactory exploration. - Three odor holders were used for the test, one dedicated to the mineral oil, another to the banana odor and the last one to the almond odor. The holders were cleaned with 20% ethanol after every use.

Figure 2. Procedure to place the scented filter paper inside the odor holder. The filter paper is bent along its longitudinal axis, and is then introduced (two thirds) into the tube of the odor holder. Before placing the odor holder into the test cage, verify that the filter paper does not change position. - The filter paper is cut into small pieces (1 cm x 3 cm, Figure 1C).
- Olfactory habituation test
- The test is performed during the light cycle with a standard temperature in an experimental room separate from the housing room.
- On day 0 the animals are food deprived for 24 h, starting 4 h after the onset of the light cycle.
- On day 1 (the test day) 30 min before the task all the necessary material (see above) is prepared. The test cage is placed on the table and cleaned with 20% ethanol. Then, all the parameters concerning the video recording and scoring software are established, such as camera focus and contrast, position of the test cage in the right camera view field, etc.
- Once everything is set up, the first holder containing the mineral oil is placed in the test cage and the video recording is started. The mouse is placed in one of the extremes of the test cage opposite to the odor holder. The task consists of 5 min exposure to mineral oil or odor separated by 5 min inter-trial intervals; during this time the mouse remains in the test cage.
- In a given session, mice are first presented with only the holder containing the mineral oil. They are then presented with the first odorant during two successive trials (habituation). Subsequently, they are presented the second odorant. The Figure 3 schematizes the different steps of the protocol.
Note: The presentation of the second odor is reflecting whether the animals can discriminate between odors, which are based on animal’s tendency to explore novel smell, and to control for a potential disinterest in the test odor.
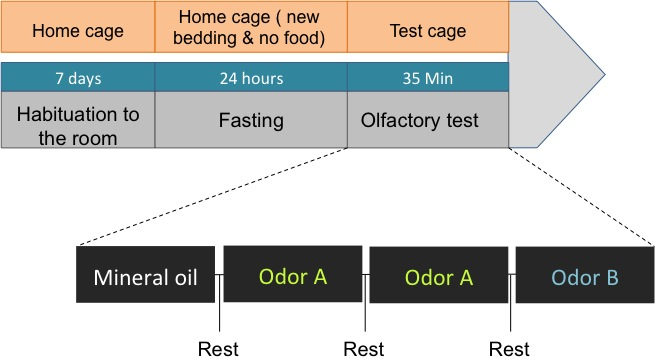
Figure 3. Olfactory habituation protocol. Immediately after arrival, mice are placed in individual cages for one week, where the body weight and food intake are monitored daily. On the test day, food deprived (24 h) animals are placed in the test cage and submitted to the presentation of first, the mineral oil, then, twice the odor A (almond or banana) and finally the odor B (almond or banana). Each presentation is followed by a rest time in which no odor or oil is presented. The time of exploration of the odor source is scored. - The test is performed during the light cycle with a standard temperature in an experimental room separate from the housing room.
Representative data
- A representative habituation/ dishabituation curve is presented in Figure 4B. Exploration of the odor source was defined as directing the nose at a distance <1 cm from the tip of the holder, with the vibrissae moving, and/or touching it with the nose. The exploration was not scored when the mouse was chewing the object.
- The test cage was cleaned with ethanol 20% between the sessions. The two odorants used were presented in accordance with a counterbalanced schedule.

Figure 4. Olfactory habituation/dishabituation in fasted mice. A) Odor source exploration by a fasted mouse. Exploration of the odor source was defined as directing the nose at a distance <1 cm from the tip of the holder, with the vibrissae moving and/or touching it with the nose. B) Representative habituation/dishabituation curve of a group of a 24 h fasted wild type mice. Note the decrease in the exploration (habituation) after the second presentation of a given odor (almond or banana). The exploration is recovered (dishabituation) after the presentation of a different odor (almond or banana).
Notes
- All animals are housed in individual cages for at least one week before the experiment.
- Bedding is changed at the start of food deprivation to prevent remnants of food from remaining in the cage
- This test is suitable to evaluate olfactory exploration in fasted mice (C57/BL6N). Non-fasted mice present low levels of exploration at the tested odorant concentration (0.1%).
- The olfactory habituation task was tested in several mice lines. We found more variability in the C57/BL6N mice than in the animals produced in our own institute. This could be due to a potential higher level of stress, associated with transport or housing conditions.
Acknowledgments
We would like to thank Samantha James and Annelot Van Esbroeck (Neurocentre Magendie) for the critical reading of this manuscript. This protocol was adapted according to McNamara et al. (2008), Fadool et al. (2004) and Ferguson et al. (2000). This work was supported by INSERM (G.M.), EU–Fp7 (REPROBESITY, HEALTH–F2–2008–223713, G.M.), European Research Council (ENDOFOOD, ERC–2010–StG–260515, G.M.), Fondation pour la Recherche Medicale (G.M.), Region Aquitaine (G.M.), LABEX BRAIN (ANR-10-LABX-43), Fyssen Foundation (E.S.–G.), We thank to Alexandre Desprez for its valuable technical assistance.
References
- Soria-Gomez, E., Bellocchio, L., Reguero, L., Lepousez, G., Martin, C., Bendahmane, M., Ruehle, S., Remmers, F., Desprez, T., Matias, I., Wiesner, T., Cannich, A., Nissant, A., Wadleigh, A., Pape, H. C., Chiarlone, A. P., Quarta, C., Verrier, D., Vincent, P., Massa, F., Lutz, B., Guzman, M., Gurden, H., Ferreira, G., Lledo, P. M., Grandes, P. and Marsicano, G. (2014). The endocannabinoid system controls food intake via olfactory processes. Nat Neurosci 17(3): 407-415.
- McNamara, A. M., Magidson, P. D., Linster, C., Wilson, D. A. and Cleland, T. A. (2008). Distinct neural mechanisms mediate olfactory memory formation at different timescales. Learn Mem 15(3): 117-125.
- Fadool, D. A., Tucker, K., Perkins, R., Fasciani, G., Thompson, R. N., Parsons, A. D., Overton, J. M., Koni, P. A., Flavell, R. A. and Kaczmarek, L. K. (2004). Kv1.3 channel gene-targeted deletion produces "Super-Smeller Mice" with altered glomeruli, interacting scaffolding proteins, and biophysics. Neuron 41(3): 389-404.
- Ferguson, J. N., Young, L. J., Hearn, E. F., Matzuk, M. M., Insel, T. R. and Winslow, J. T. (2000). Social amnesia in mice lacking the oxytocin gene. Nat Genet 25(3): 284-288.
- Linster, C., Menon, A. V., Singh, C. Y. and Wilson, D. A. (2009). Odor-specific habituation arises from interaction of afferent synaptic adaptation and intrinsic synaptic potentiation in olfactory cortex. Learn Mem 16(7): 452-459.
- Qiu, Q., Scott, A., Scheerer, H., Sapkota, N., Lee, D. K., Ma, L. and Yu, C. R. (2014). Automated analyses of innate olfactory behaviors in rodents. PLoS One 9(4): e93468.
- Slotnick, B. and Restrepo, D. (2005). Olfactometry with mice. Curr Protoc Neurosci: 8.20. 21-28.20. 24.
- Tan, J., Savigner, A., Ma, M. and Luo, M. (2010). Odor information processing by the olfactory bulb analyzed in gene-targeted mice. Neuron 65(6): 912-926.
- Yang, L., Zou, B., Xiong, X., Pascual, C., Xie, J., Malik, A., Xie, J., Sakurai, T. and Xie, X. S. (2013). Hypocretin/orexin neurons contribute to hippocampus-dependent social memory and synaptic plasticity in mice. J Neurosci 33(12): 5275-5284.
- Yang, M. and Crawley, J. N. (2009). Simple behavioral assessment of mouse olfaction. Curr Protoc Neurosci Chapter 8: Unit 8 24.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Desprez, T., Marsicano, G. and Soria-Gómez, E. (2014). Olfactory Habituation in Fasted Mice. Bio-protocol 4(20): e1267. DOI: 10.21769/BioProtoc.1267.
Category
Neuroscience > Behavioral neuroscience > Cognition
Neuroscience > Behavioral neuroscience > Learning and memory
Neuroscience > Sensory and motor systems
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










