- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Mesenchymal Stem Cell (MSC) Aggregate Formation in vivo
Published: Vol 4, Iss 14, Jul 20, 2014 DOI: 10.21769/BioProtoc.1181 Views: 12596
Reviewed by: Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
A Protocol to Induce Brown and Beige Adipocyte Differentiation From Murine and Human Adipose-Derived SVF
Rohit Raj Yadav [...] Narendra Verma
Dec 5, 2025 1641 Views
Optimization of Adipogenic Differentiation Protocol for Murine and Human Cell Culture Models
Junwan Fan [...] Wenyan He
Jan 20, 2026 211 Views
Simple and Rapid Model to Generate Differentiated Endometrial Floating Organoids
Adriana Bajetto [...] Tullio Florio
Feb 5, 2026 92 Views
Abstract
Human mesenchymal stem/progenitor cells (MSCs) isolated from various adult tissues show remarkable therapeutic potential and are being employed in clinical trials for the treatment of numerous diseases (Prockop et al., 2010). While routes of cell administration vary, profound beneficial effects of MSCs in animal models have been observed following intraperitoneal injections of the cells (Roddy et al., 2011). Similar to MSC spheres formed in culture under conditions where attachment to plastic is not permitted (Bartosh et al., 2010), MSCs injected into the peritoneum of mice spontaneously aggregate into 3D sphere-like structures (Bartosh et al., 2013). During the process of sphere assembly and compaction, MSCs upregulate expression of numerous therapeutic anti-inflammatory and immune modulatory factors. Here we describe the method we previously used for the generation of human bone marrow-derived MSC aggregates/spheres in vivo (Bartosh et al., 2013). By tagging the MSCs with green fluorescent protein (GFP), the aggregates formed can be easily visualized, collected and analyzed for changes in cellular properties and interactions with host immune cells.
Keywords: MSCsMaterials and Reagents
- Human bone marrow mesenchymal stem cells expressing green fluorescent protein (GFP-MSCs) from The Center for the Preparation and Distribution of Adult Stem Cells (http://medicine.tamhsc.edu/irm/msc-distribution.html)
- C57BL/6J or BALB/C mice (2-3 months of age) (The Jackson Laboratory)
- Phosphate-buffered saline (PBS) without Ca2+ and Mg2+ (pH 7.4) (Life Technologies, catalog number: 10010 )
- Hank’s Balanced Salt Solution (HBSS) without Ca2+ and Mg2+ (Lonza, catalog number: 04-315Q )
- 0.25% trypsin with 1x EDTA (Life Technologies, catalog number: 25200 )
- Minimum Essential Medium alpha (Life Technologies, catalog number: 12561 )
- Premium select fetal bovine serum (Atlanta Biologicals, catalog number: S11550 )
- Penicillin-streptomycin (Life Technologies, catalog number: 15140 )
- 100x L-glutamine (Life Technologies, catalog number: 25030 )
- Complete culture medium (CCM) for MSC growth (see Recipes)
Equipment
- Stericup-GP 0.22 µm vacuum filtration device (EMD Millipore, catalog number: SCGPU05RE )
- Water bath set to 37 °C
- Centrifuge with swinging-bucket rotor and adaptors for 50 ml conical tubes
- 50 ml sterile conical tube (BD Biosciences, Falcon®, catalog number: 352070 )
- Humidified cell culture incubator set to 37 °C and 5% CO2
- Upright microscope with 4x and 10x objectives and a filter set to visualize GFP
- 29 gauge needle with 1 ml syringe (Terumo Europe N.V., catalog number: 05M2913 )
- Isoflurane anesthesia system with nose cone for mouse
- Sterile dissecting scissors, pins, and curved forceps with a serrated edge
- Rubber or styrofoam platform
- Dissecting microscope with optional camera and monitor (Figure 1)
- Illumatool Bright Lights Systems LT 9900 with epi-fluorescence attachment (Lightools Research) and GFP filter set (Figure 1)

Figure 1. Equipment required to visualize and collect GFP-MSC aggregates/spheres from the mouse peritoneum. GFP-MSC aggregates/spheres can be visualized in the mouse peritoneum using a dissecting microscope with an epi-fluorescence attachment and GFP filter set. High quality images can be acquired with an appropriate camera mounted to the dissecting scope (a camera is not a requirement for collecting the aggregates).
Procedure
- Determine the number of animals that will be injected with the GFP-MSCs and the number of cells that will be required.
Note: We have successfully injected 1-3 x 106 GFP-MSCs per animal to achieve the desired results.
- Expand the GFP-MSCs in CCM from low-density seeding (100-300 cells/cm2) for 6-7 days. Upon reaching 70% confluence, harvest the GFP-MSCs using trypsin/EDTA then collect the cells by centrifugation at 450 x g for 5-7 min.
Note: Proper MSC culture method is detailed in references below. It is important to prevent MSC cultures from becoming confluent. If necessary, the MSCs can be passaged to avoid the formation of confluent cultures.
- Aspirate the supernatant and suspend the GFP-MSC pellet in a low volume of CCM for cell counts.
Note: A viability dye such as trypan blue is recommended to discriminate between live and dead cells. GFP-MSCs are typically greater than 95% viable.
- After counting the cells, add up to 45 ml HBSS to the cells and centrifuge at 450 x g for 5-7 min.
- Aspirate the supernatant and suspend the cells in HBSS at a concentration of 5,000-10,000 viable cells per µl.
- With the mouse under isoflurane anesthesia, inject 200-300 µl of GFP-MSCs (1-3 x 106 cells) from the cell suspension into the peritoneal cavity using a 29 gauge needle.
Notes:- For optimal MSC viability, do not mix the cell suspension with the needle. Instead, mix cells using a pipet or simply by flicking the vial.
- Intraperitoneal injections should be performed following procedures set forth by your Institutional Animal Care and Use Committee or equivalent.
- For optimal MSC viability, do not mix the cell suspension with the needle. Instead, mix cells using a pipet or simply by flicking the vial.
- Immediately following GFP-MSC injection, disperse the cells within the peritoneum by gently massaging the abdomen for 5-10 sec.
Note: To massage the abdomen, gently press on the abdomen with 1 or 2 fingers. Start from an area in close proximity to the injection site and progress distally. Do not press forcefully to avoid causing trauma to the internal organs.
- Repeat procedure with additional mice.
- Accommodate the mice for 4-72 h on a standard 12 h light/dark cycle and with standard nutrition.
Notes:- Aggregates collected at 72 h are compact and often adhere to the tissues making them more difficult to obtain. Aggregates collected at earlier time points are less compact and more abundant.
- Aggregates collected from the peritoneum will vary in size and number. MSC aggregates typically range in size from several hundreds of microns to 2-3 mm in diameter although smaller aggregates composed of fewer cells can be obtained. The number of aggregates that can be collected from the peritoneum gradually decreases with time. Following injection of 1-3 x 106 GFP-MSCs, 15-30 aggregates can be obtained at 4 h, 10-15 aggregates at 24 h, and less than 10 aggregates at 72 h.
- Aggregates collected at 72 h are compact and often adhere to the tissues making them more difficult to obtain. Aggregates collected at earlier time points are less compact and more abundant.
- After 4-72 h, anesthetize the mouse by isoflurane inhalation then euthanize the animal by cervical dislocation.
Note: Other methods of euthanasia can be performed however it is not recommended to use a method that introduces fluid into the peritoneum.
- Using dissecting pins stabilize the euthanized mouse on a rubber or styrofoam platform (ventral side up).
- Expose the peritoneal cavity using dissecting forceps and scissors (Figure 2). Avoid disturbing the internal organs or cell aggregates.
- Visualize the GFP signal using a dissecting microscope with an epi-fluorescence attachment and appropriate filter set (Figure 2).
Note: The localization of the aggregates can be variable between animals. Under some circumstances, visualization of aggregates requires careful repositioning of the internal organs.
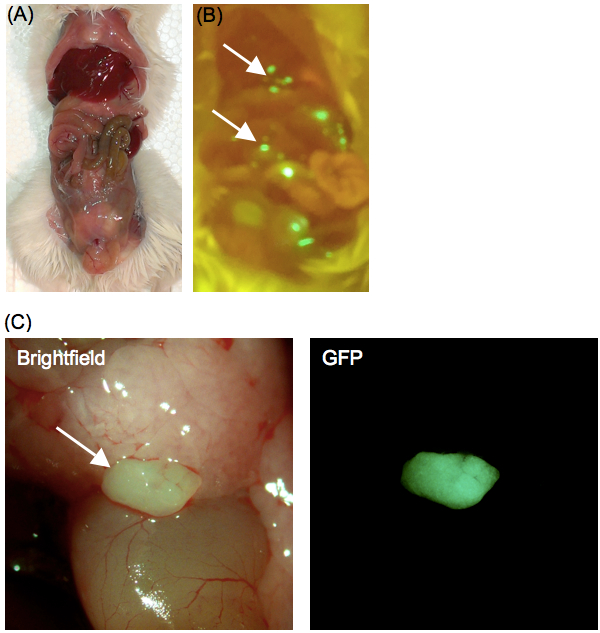
Figure 2. Formation of GFP-MSC aggregates in the peritoneal cavity. Images of the mouse peritoneal cavity uncovered to reveal the (A) internal organs and (B) GFP-MSC aggregates. (C) Magnified view of a GFP-MSC aggregate (arrow) in the peritoneum of a BALB/c mouse 4 h after intraperitoneal injection of 2 x 106 GFP-MSCs.
- Collect the aggregates using forceps and transfer to a 50 ml tube containing ice cold HBSS.
Notes:- Due to surface tension, the aggregates adhere readily to the forceps without the need to squeeze the forceps around the cells.
- Aggregate formation by MSCs in the peritoneal cavity is not the only fate of the cells. MSCs will adhere to certain structures of the peritoneum. Free floating aggregates can be distinguished from adherent cells with careful examination. Moreover, the peritoneum can be washed repeatedly with 1-2 ml HBSS to distinguish the free floating aggregates that often reposition during the wash from adherent cells that will not move.
- Due to surface tension, the aggregates adhere readily to the forceps without the need to squeeze the forceps around the cells.
- After collecting the GFP-MSC aggregates of interest, make certain that most of the cells descended to the bottom of the tube.
Note: Aggregates connected to fatty tissue will remain in suspension and can be removed by careful aspiration. Aggregates that are void of fatty tissue will rapidly fall to the bottom of the tube.
- Aspirate the HBSS supernatant to remove cell aggregates containing fatty tissue while avoiding contact with the cell pellet.
- Wash the cells in HBSS and collect aggregates by centrifugation at 400 x g for 5 min.
- Aspirate the supernatant. The aggregates are now prepared for downstream applications and analysis. For analyzing changes in gene and protein expression, the cell pellet can by lysed in appropriate lysis solution. Alternatively, aggregates can be transferred to a cell culture dish (Figure 3) or to a cryomold and frozen for immunofluorescence applications. MSC aggregates can also be dissociated using trypsin/EDTA and analyzed by microscopy (Figure 4) or flow cytometry. Other applications have not been tested.
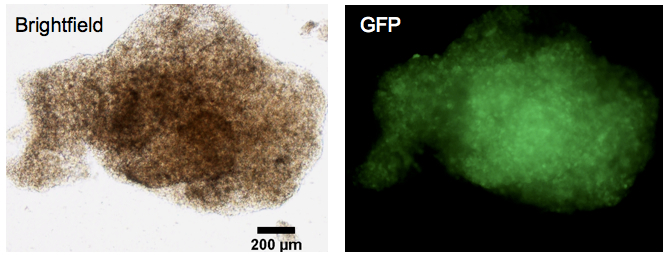
Figure 3. Transfer of GFP-MSC aggregates in vitro. Images of a GFP-MSC aggregate 1 h after transfer from the peritnoneum of a BALB/c mouse to a cell culture dish. The aggregates form sturdy micro-tissue structures. Scale bar = 200 µm

Figure 4. Microscopic examination of GFP-MSCs derived from peritoneal aggregates. Images of the cells obtained 12 h following dissociation of aggregates harvested from the peritoneum. GFP-MSCs appear as large flat cells that readily attach to a cell culture dish. Small round cells (GFP negative) are also obtained from peritoneal aggregates. The majority of the small round cells are host immune cells. Scale bar = 100 µm
Notes
- All animal procedures were approved by the Animal Care and Use Committe of Texas A&M Health Science Center and in accordance with guidelines set forth by the National Institutes of Health.
- The protocol was performed using MSCs obtained from human bone marrow aspirates. The cells were transduced with a lentiviral reporter harboring GFP. Other types of cells have not been thoroughly tested in this model.
- Aggregates/spheres will show a range in size from several hundreds of microns to 2-3 mm in diameter with each aggregate containing thousands of cells. The size and number of aggregates can be augmented by increasing the number of cells injected into the peritoneum. We typically inject 1-3 x 106 GFP-MSCs per animal and readily collect 10-30 aggregates/spheres of variable size.
- Aggregates/spheres collected from the peritoneum generally consist of both the GFP-MSCs that were administered and resident peritoneal immune cells.
Recipes
- Complete Culture Medium (CCM)
Minimum Essential Medium alpha, 1 L
Premium select fetal bovine serum, 200 ml
Penicillin-streptomycin, 12 ml
100x L-glutamine, 12 ml
Filter sterilize (0.22 µm)
Stored at 4 °C for up to 1 month
Pre-warm to 37 °C in water bath prior to use
Acknowledgments
This protocol was adapted from our previous work (Bartosh et al., 2013) and was supported by an NIH grant (P40RR17447) to Darwin J. Prockop, Institute for Regenerative Medicine, Texas A&M University Health Science Center, and a grant from the Cancer Prevention and Research Institute of Texas (RP110553-P1).
References
- Bartosh, T. J., Ylostalo, J. H., Bazhanov, N., Kuhlman, J. and Prockop, D. J. (2013). Dynamic compaction of human mesenchymal stem/precursor cells into spheres self-activates caspase-dependent IL1 signaling to enhance secretion of modulators of inflammation and immunity (PGE2, TSG6, and STC1). Stem Cells 31(11): 2443-2456.
- Bartosh, T. J., Ylostalo, J. H., Mohammadipoor, A., Bazhanov, N., Coble, K., Claypool, K., Lee, R. H., Choi, H. and Prockop, D. J. (2010). Aggregation of human mesenchymal stromal cells (MSCs) into 3D spheroids enhances their antiinflammatory properties. Proc Natl Acad Sci U S A 107(31): 13724-13729.
- Prockop, D. J., Kota, D. J., Bazhanov, N. and Reger, R. L. (2010). Evolving paradigms for repair of tissues by adult stem/progenitor cells (MSCs). J Cell Mol Med 14(9): 2190-2199.
- Roddy, G. W., Oh, J. Y., Lee, R. H., Bartosh, T. J., Ylostalo, J., Coble, K., Rosa, R. H., Jr. and Prockop, D. J. (2011). Action at a distance: systemically administered adult stem/progenitor cells (MSCs) reduce inflammatory damage to the cornea without engraftment and primarily by secretion of TNF-alpha stimulated gene/protein 6. Stem Cells 29(10): 1572-1579.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Bartosh, T. J. and Ylostalo, J. H. (2014). Mesenchymal Stem Cell (MSC) Aggregate Formation in vivo. Bio-protocol 4(14): e1181. DOI: 10.21769/BioProtoc.1181.
Category
Stem Cell > Adult stem cell > Mesenchymal stem cell
Stem Cell > Adult stem cell > Maintenance and differentiation
Cell Biology > Cell isolation and culture > 3D cell culture
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











